Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Adenylate cyclase type 1
Ligand
BDBM50052123
Substrate
n/a
Meas. Tech.
ChEMBL_31878 (CHEMBL645668)
IC50
36±n/a nM
Citation
 Robbins, JD; Boring, DL; Tang, WJ; Shank, R; Seamon, KB Forskolin carbamates: binding and activation studies with type I adenylyl cyclase. J Med Chem 39:2745-52 (1996) [PubMed] Article
Robbins, JD; Boring, DL; Tang, WJ; Shank, R; Seamon, KB Forskolin carbamates: binding and activation studies with type I adenylyl cyclase. J Med Chem 39:2745-52 (1996) [PubMed] Article More Info.:
Target
Name:
Adenylate cyclase type 1
Synonyms:
ADCY1 | ADCY1_HUMAN | Adenylate cyclase | Brain adenylate cyclase 1
Type:
PROTEIN
Mol. Mass.:
123469.95
Organism:
Homo sapiens (Human)
Description:
ChEMBL_31877
Residue:
1119
Sequence:
MAGAPRGGGGGGGGAGEPGGAERAAGTSRRRGLRACDEEFACPELEALFRGYTLRLEQAATLKALAVLSLLAGALALAELLGAPGPAPGLAKGSHPVHCVLFLALLVVTNVRSLQVPQLQQVGQLALLFSLTFALLCCPFALGGPARGSAGAAGGPATAEQGVWQLLLVTFVSYALLPVRSLLAIGFGLVVAASHLLVTATLVPAKRPRLWRTLGANALLFVGVNMYGVFVRILTERSQRKAFLQARSCIEDRLRLEDENEKQERLLMSLLPRNVAMEMKEDFLKPPERIFHKIYIQRHDNVSILFADIVGFTGLASQCTAQELVKLLNELFGKFDELATENHCRRIKILGDCYYCVSGLTQPKTDHAHCCVEMGLDMIDTITSVAEATEVDLNMRVGLHTGRVLCGVLGLRKWQYDVWSNDVTLANVMEAAGLPGKVHITKTTLACLNGDYEVEPGYGHERNSFLKTHNIETFFIVPSHRRKIFPGLILSDIKPAKRMKFKTVCYLLVQLMHCRKMFKAEIPFSNVMTCEDDDKRRALRTASEKLRNRSSFSTNVVYTTPGTRVNRYISRLLEARQTELEMADLNFFTLKYKHVEREQKYHQLQDEYFTSAVVLTLILAALFGLVYLLIFPQSVVVLLLLVFCICFLVACVLYLHITRVQCFPGCLTIQIRTVLCIFIVVLIYSVAQGCVVGCLPWAWSSKPNSSLVVLSSGGQRTALPTLPCESTHHALLCCLVGTLPLAIFFRVSSLPKMILLSGLTTSYILVLELSGYTRTGGGAVSGRSYEPIVAILLFSCALALHARQVDIRLRLDYLWAAQAEEEREDMEKVKLDNRRILFNLLPAHVAQHFLMSNPRNMDLYYQSYSQVGVMFASIPNFNDFYIELDGNNMGVECLRLLNEIIADFDELMEKDFYKDIEKIKTIGSTYMAAVGLAPTSGTKAKKSISSHLSTLADFAIEMFDVLDEINYQSYNDFVLRVGINVGPVVAGVIGARRPQYDIWGNTVNVASRMDSTGVQGRIQVTEEVHRLLRRCPYHFVCRGKVSVKGKGEMLTYFLEGRTDGNGSQIRSLGLDRKMCPFGRAGLQGRRPPVCPMPGVSVRAGLPPHSPGQYLPSAAAGKEA
Inhibitor
Name:
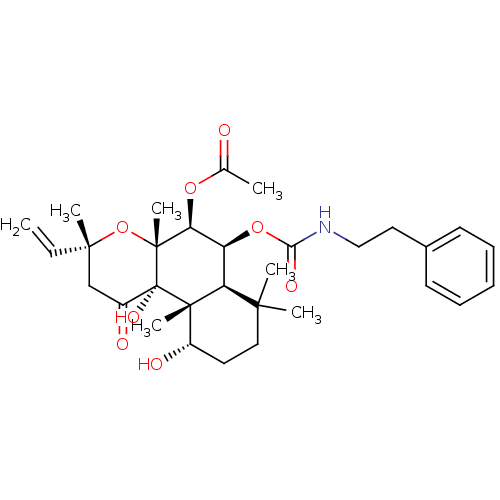
BDBM50052123
Synonyms:
Acetic acid (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-10,10b-dihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-6-phenethylcarbamoyloxy-3-vinyl-dodecahydro-benzo[f]chromen-5-yl ester | CHEMBL329247
Type:
Small organic molecule
Emp. Form.:
C31H43NO8
Mol. Mass.:
557.675
SMILES:
CC(=O)O[C@H]1[C@@H](OC(=O)NCCc2ccccc2)[C@H]2C(C)(C)CC[C@H](O)[C@]2(C)[C@@]2(O)C(=O)C[C@@](C)(O[C@]12C)C=C