Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Estrogen receptor
Ligand
BDBM263681
Substrate
n/a
Meas. Tech.
ChEMBL_1824582 (CHEMBL4324346)
EC50
0.900000±n/a nM
Citation
 El-Ahmad, Y; Tabart, M; Halley, F; Certal, V; Thompson, F; Filoche-Romm�, B; Gruss-Leleu, F; Muller, C; Brollo, M; Fabien, L; Loyau, V; Bertin, L; Richepin, P; Pilorge, F; Desmazeau, P; Girardet, C; Beccari, S; Louboutin, A; Lebourg, G; Le-Roux, J; Terrier, C; Vall�e, F; Steier, V; Mathieu, M; Rak, A; Abecassis, PY; Vicat, P; Benard, T; Bouaboula, M; Sun, F; Shomali, M; Hebert, A; Levit, M; Cheng, H; Courjaud, A; Ginesty, C; Perrault, C; Garcia-Echeverria, C; McCort, G; Schio, L Discovery of 6-(2,4-Dichlorophenyl)-5-[4-[(3 J Med Chem 63:512-528 (2020) [PubMed] Article
El-Ahmad, Y; Tabart, M; Halley, F; Certal, V; Thompson, F; Filoche-Romm�, B; Gruss-Leleu, F; Muller, C; Brollo, M; Fabien, L; Loyau, V; Bertin, L; Richepin, P; Pilorge, F; Desmazeau, P; Girardet, C; Beccari, S; Louboutin, A; Lebourg, G; Le-Roux, J; Terrier, C; Vall�e, F; Steier, V; Mathieu, M; Rak, A; Abecassis, PY; Vicat, P; Benard, T; Bouaboula, M; Sun, F; Shomali, M; Hebert, A; Levit, M; Cheng, H; Courjaud, A; Ginesty, C; Perrault, C; Garcia-Echeverria, C; McCort, G; Schio, L Discovery of 6-(2,4-Dichlorophenyl)-5-[4-[(3 J Med Chem 63:512-528 (2020) [PubMed] Article More Info.:
Target
Name:
Estrogen receptor
Synonyms:
ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1
Type:
Protein
Mol. Mass.:
66230.44
Organism:
Human
Description:
P03372
Residue:
595
Sequence:
MTMTLHTKASGMALLHQIQGNELEPLNRPQLKIPLERPLGEVYLDSSKPAVYNYPEGAAYEFNAAAAANAQVYGQTGLPYGPGSEAAAFGSNGLGGFPPLNSVSPSPLMLLHPPPQLSPFLQPHGQQVPYYLENEPSGYTVREAGPPAFYRPNSDNRRQGGRERLASTNDKGSMAMESAKETRYCAVCNDYASGYHYGVWSCEGCKAFFKRSIQGHNDYMCPATNQCTIDKNRRKSCQACRLRKCYEVGMMKGGIRKDRRGGRMLKHKRQRDDGEGRGEVGSAGDMRAANLWPSPLMIKRSKKNSLALSLTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMINWAKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLLLDRNQGKCVEGMVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVYTFLSSTLKSLEEKDHIHRVLDKITDTLIHLMAKAGLTLQQQHQRLAQLLLILSHIRHMSNKGMEHLYSMKCKNVVPLYDLLLEMLDAHRLHAPTSRGGASVEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV
Inhibitor
Name:
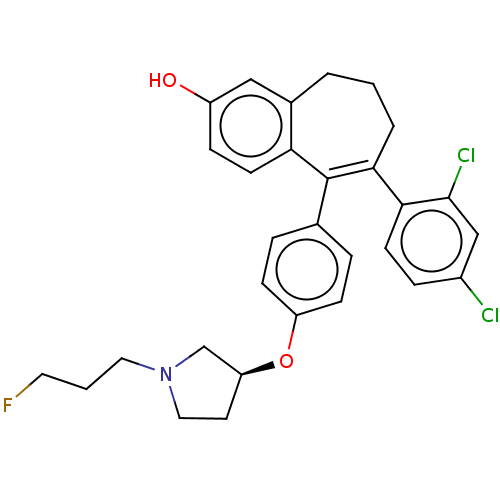
BDBM263681
Synonyms:
6-(2,4- dichloro- phenyl)-5-[4- [(3S)-1-(3- fluoropropyl)- pyrrolidin-3- yl]oxy- phenyl]-8,9- dihydro- 7H- benzo[7]- annulen-2-ol | US9714221, Example 18
Type:
Small organic molecule
Emp. Form.:
C30H30Cl2FNO2
Mol. Mass.:
526.469
SMILES:
Oc1ccc2c(CCCC(c3ccc(Cl)cc3Cl)=C2c2ccc(O[C@H]3CCN(CCCF)C3)cc2)c1 |r,c:18|