Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50123479
Substrate
n/a
Meas. Tech.
ChEMBL_210607 (CHEMBL816574)
Ki
0.54±n/a nM
Citation
 Burgey, CS; Robinson, KA; Lyle, TA; Sanderson, PE; Lewis, SD; Lucas, BJ; Krueger, JA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Lyle, EA; Williams, PD; Coburn, CA; Dorsey, BD; Barrow, JC; Stranieri, MT; Holahan, MA; Sitko, GR; Cook, JJ; McMasters, DR; McDonough, CM; Sanders, WM; Wallace, AA; Clayton, FC; Bohn, D; Leonard, YM; Detwiler, TJ; Lynch, JJ; Yan, Y; Chen, Z; Kuo, L; Gardell, SJ; Shafer, JA; Vacca, JP Metabolism-directed optimization of 3-aminopyrazinone acetamide thrombin inhibitors. Development of an orally bioavailable series containing P1 and P3 pyridines. J Med Chem 46:461-73 (2003) [PubMed] Article
Burgey, CS; Robinson, KA; Lyle, TA; Sanderson, PE; Lewis, SD; Lucas, BJ; Krueger, JA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Lyle, EA; Williams, PD; Coburn, CA; Dorsey, BD; Barrow, JC; Stranieri, MT; Holahan, MA; Sitko, GR; Cook, JJ; McMasters, DR; McDonough, CM; Sanders, WM; Wallace, AA; Clayton, FC; Bohn, D; Leonard, YM; Detwiler, TJ; Lynch, JJ; Yan, Y; Chen, Z; Kuo, L; Gardell, SJ; Shafer, JA; Vacca, JP Metabolism-directed optimization of 3-aminopyrazinone acetamide thrombin inhibitors. Development of an orally bioavailable series containing P1 and P3 pyridines. J Med Chem 46:461-73 (2003) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Human
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
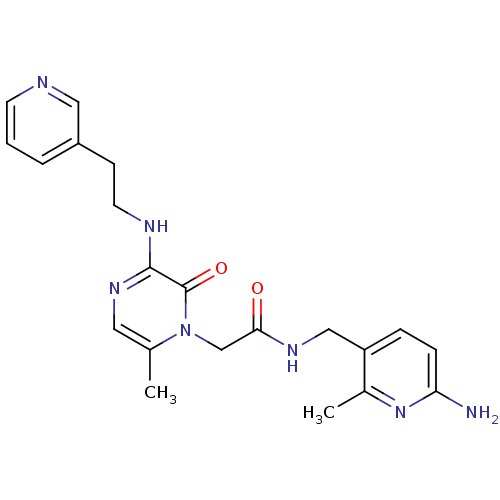
Inhibitor
Name:
BDBM50123479
Synonyms:
CHEMBL143008 | N-(6-Amino-2-methyl-pyridin-3-ylmethyl)-2-[6-methyl-2-oxo-3-(2-pyridin-3-yl-ethylamino)-2H-pyrazin-1-yl]-acetamide
Type:
Small organic molecule
Emp. Form.:
C21H25N7O2
Mol. Mass.:
407.4689
SMILES:
Cc1nc(N)ccc1CNC(=O)Cn1c(C)cnc(NCCc2cccnc2)c1=O