Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Phospho-N-acetylmuramoyl-pentapeptide-transferase
Ligand
BDBM50318260
Substrate
n/a
Meas. Tech.
ChEMBL_629295 (CHEMBL1103975)
IC50
63000±n/a nM
Citation
 Ii, K; Ichikawa, S; Al-Dabbagh, B; Bouhss, A; Matsuda, A Function-oriented synthesis of simplified caprazamycins: discovery of oxazolidine-containing uridine derivatives as antibacterial agents against drug-resistant bacteria. J Med Chem 53:3793-813 (2010) [PubMed] Article
Ii, K; Ichikawa, S; Al-Dabbagh, B; Bouhss, A; Matsuda, A Function-oriented synthesis of simplified caprazamycins: discovery of oxazolidine-containing uridine derivatives as antibacterial agents against drug-resistant bacteria. J Med Chem 53:3793-813 (2010) [PubMed] Article More Info.:
Target
Name:
Phospho-N-acetylmuramoyl-pentapeptide-transferase
Synonyms:
MRAY_BACSU | mraY
Type:
PROTEIN
Mol. Mass.:
35531.14
Organism:
Bacillus subtilis
Description:
ChEMBL_796247
Residue:
324
Sequence:
MLEQVILFTILMGFLISVLLSPILIPFLRRLKFGQSIREEGPKSHQKKSGTPTMGGVMIILSIIVTTIVMTQKFSEISPEMVLLLFVTLGYGLLGFLDDYIKVVMKRNLGLTSKQKLIGQIIIAVVFYAVYHYYNFATDIRIPGTDLSFDLGWAYFILVLFMLVGGSNAVNLTDGLDGLLSGTAAIAFGAFAILAWNQSQYDVAIFSVAVVGAVLGFLVFNAHPAKVFMGDTGSLALGGAIVTIAILTKLEILLVIIGGVFVIETLSVILQVISFKTTGKRIFKMSPLHHHYELVGWSEWRVVVTFWAAGLLLAVLGIYIEVWL
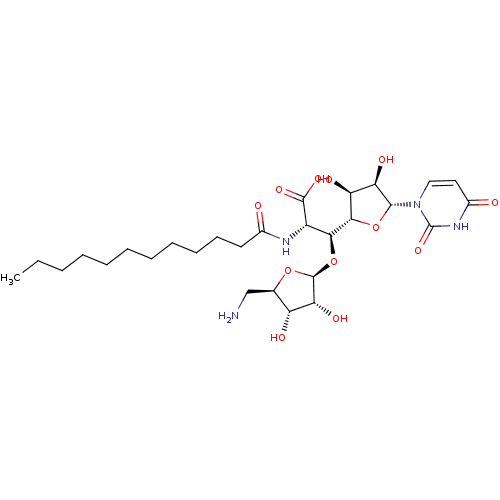
Inhibitor
Name:
BDBM50318260
Synonyms:
5-O-(5-Amino-5-deoxy-beta-D-ribofuranosyl)-6-deoxy-6-dodecanoylamino-1-(uracil-1-yl)-beta-D-glycero-L-talo-heptofuranuronicAcid Trifluoroacetic Salt | CHEMBL1095317
Type:
Small organic molecule
Emp. Form.:
C28H46N4O12
Mol. Mass.:
630.6844
SMILES:
CCCCCCCCCCCC(=O)N[C@@H]([C@H](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)C(O)=O |r|