Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
GTPase KRas [1-169,G12C,C118A]
Ligand
BDBM544351
Substrate
n/a
Meas. Tech.
Coupled Nucleotide Exchange Assay
IC50
31.0±n/a nM
Citation
 Allen, JG; Lanman, BA; Chen, J; Reed, AB; Cee, VJ; Liu, L; Lopez, P; Wurz, RP; Nguyen, TT; Booker, S; Allen, JR; Chu-Moyer, M; Amegadzie, A; Chen, N; Goodman, C; Low, JD; Ma, VV; Minatti, AE; Nishimura, N; Pickrell, AJ; Wang, H; Shin, Y; Siegmund, AC; Yang, KC; Tamayo, NA; Walton, M; Xue, Q Substituted piperazines as KRAS G12C inhibitors US Patent US11285156 Publication Date 3/29/2022
Allen, JG; Lanman, BA; Chen, J; Reed, AB; Cee, VJ; Liu, L; Lopez, P; Wurz, RP; Nguyen, TT; Booker, S; Allen, JR; Chu-Moyer, M; Amegadzie, A; Chen, N; Goodman, C; Low, JD; Ma, VV; Minatti, AE; Nishimura, N; Pickrell, AJ; Wang, H; Shin, Y; Siegmund, AC; Yang, KC; Tamayo, NA; Walton, M; Xue, Q Substituted piperazines as KRAS G12C inhibitors US Patent US11285156 Publication Date 3/29/2022 More Info.:
Target
Name:
GTPase KRas [1-169,G12C,C118A]
Synonyms:
GTPase KRas | KRAS | KRAS2 | RASK2 | RASK_HUMAN
Type:
PROTEIN
Mol. Mass.:
19407.24
Organism:
Human
Description:
P01116[1-169,G12C,C118A]
Residue:
169
Sequence:
MTEYKLVVVGACGVGKSALTIQLIQNHFVDEYDPTIEDSYRKQVVIDGETCLLDILDTAGQEEYSAMRDQYMRTGEGFLCVFAINNTKSFEDIHHYREQIKRVKDSEDVPMVLVGNKADLPSRTVDTKQAQDLARSYGIPFIETSAKTRQRVEDAFYTLVREIRQYRLK
Inhibitor
Name:
BDBM544351
Synonyms:
US11285156, Ex.# 177-1
Type:
Small organic molecule
Emp. Form.:
C33H34ClFN6O2
Mol. Mass.:
601.113
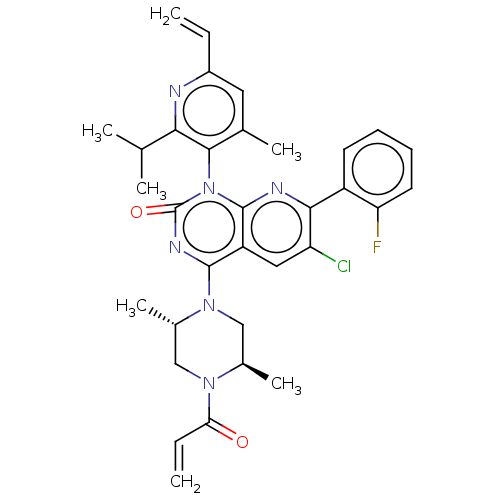
SMILES:
CC(C)c1nc(C=C)cc(C)c1-n1c2nc(c(Cl)cc2c(nc1=O)N1C[C@@H](C)N(C[C@@H]1C)C(=O)C=C)-c1ccccc1F |r,wU:30.34,wD:26.29,(.61,-27.08,;1.38,-25.75,;-.16,-25.75,;2.92,-25.75,;3.69,-27.08,;5.23,-27.08,;6,-28.42,;7.54,-28.42,;6,-25.75,;5.23,-24.42,;6,-23.08,;3.69,-24.42,;2.92,-23.08,;3.69,-21.75,;5.23,-21.75,;6,-20.41,;5.23,-19.08,;6,-17.75,;3.69,-19.08,;2.92,-20.41,;1.38,-20.41,;.61,-21.75,;1.38,-23.08,;.61,-24.42,;.61,-19.08,;1.38,-17.75,;.61,-16.41,;1.38,-15.08,;-.93,-16.41,;-1.7,-17.75,;-.93,-19.08,;-1.7,-20.41,;-1.7,-15.08,;-.93,-13.75,;-3.24,-15.08,;-4.01,-13.75,;7.54,-20.41,;8.31,-21.75,;9.85,-21.75,;10.62,-20.41,;9.85,-19.08,;8.31,-19.08,;7.54,-17.75,)|