Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Serine protease 1
Ligand
BDBM14140
Substrate
BDBM14141
Meas. Tech.
Enzyme Inhibition Assay
IC50
5.8±n/a nM
Citation
 Costanzo, MJ; Almond, HR; Hecker, LR; Schott, MR; Yabut, SC; Zhang, HC; Andrade-Gordon, P; Corcoran, TW; Giardino, EC; Kauffman, JA; Lewis, JM; de Garavilla, L; Haertlein, BJ; Maryanoff, BE In-depth study of tripeptide-based alpha-ketoheterocycles as inhibitors of thrombin. Effective utilization of the S1' subsite and its implications to structure-based drug design. J Med Chem 48:1984-2008 (2005) [PubMed] Article
Costanzo, MJ; Almond, HR; Hecker, LR; Schott, MR; Yabut, SC; Zhang, HC; Andrade-Gordon, P; Corcoran, TW; Giardino, EC; Kauffman, JA; Lewis, JM; de Garavilla, L; Haertlein, BJ; Maryanoff, BE In-depth study of tripeptide-based alpha-ketoheterocycles as inhibitors of thrombin. Effective utilization of the S1' subsite and its implications to structure-based drug design. J Med Chem 48:1984-2008 (2005) [PubMed] Article More Info.:
Target
Name:
Serine protease 1
Synonyms:
Beta-Trypsin | Cationic trypsin | PRSS1 | TRP1 | TRY1 | TRY1_BOVIN | TRYP1 | Trypsin | Trypsin I
Type:
Enzyme
Mol. Mass.:
25790.52
Organism:
Bovine
Description:
P00760
Residue:
246
Sequence:
MKTFIFLALLGAAVAFPVDDDDKIVGGYTCGANTVPYQVSLNSGYHFCGGSLINSQWVVSAAHCYKSGIQVRLGEDNINVVEGNEQFISASKSIVHPSYNSNTLNNDIMLIKLKSAASLNSRVASISLPTSCASAGTQCLISGWGNTKSSGTSYPDVLKCLKAPILSDSSCKSAYPGQITSNMFCAGYLEGGKDSCQGDSGGPVVCSGKLQGIVSWGSGCAQKNKPGVYTKVCNYVSWIKQTIASN
Inhibitor
Name:
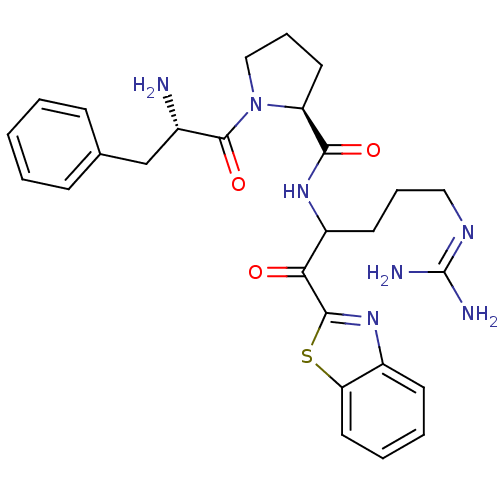
BDBM14140
Synonyms:
(2S)-1-[(2S)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamido-1-oxopentan-2-yl]pyrrolidine-2-carboxamide | 2-ketobenzothiazole 78
Type:
Small organic molecule
Emp. Form.:
C27H33N7O3S
Mol. Mass.:
535.661
SMILES:
[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r|
Substrate
Name:
BDBM14141
Synonyms:
Cbo-Gly-D-Ala-Arg-pNA.AcOH | Spectrozyme TRY | benzyl N-({[(1R)-1-{[(1S)-4-carbamimidamido-1-[(4-nitrophenyl)carbamoyl]butyl]carbamoyl}ethyl]carbamoyl}methyl)carbamate; acetic acid | carbobenzoxy-glycyl-D-alanine-p-nitroanilide-acetate
Type:
n/a
Emp. Form.:
n/a
Mol. Mass.:
n/a
SMILES:
n/a