Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50066641
Substrate
n/a
Meas. Tech.
ChEMBL_208533 (CHEMBL813627)
Ki
250±n/a nM
Citation
 Phillips, GB; Buckman, BO; Davey, DD; Eagen, KA; Guilford, WJ; Hinchman, J; Ho, E; Koovakkat, S; Liang, A; Light, DR; Mohan, R; Ng, HP; Post, JM; Shaw, KJ; Smith, D; Subramanyam, B; Sullivan, ME; Trinh, L; Vergona, R; Walters, J; White, K; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Discovery of N-[2-[5-[Amino(imino)methyl]-2-hydroxyphenoxy]-3, 5-difluoro-6-[3-(4, 5-dihydro-1-methyl-1H-imidazol-2-yl)phenoxy]pyridin-4-yl]-N-methylgl y cine (ZK-807834): a potent, selective, and orally active inhibitor of the blood coagulation enzyme factor Xa. J Med Chem 41:3557-62 (1998) [PubMed] Article
Phillips, GB; Buckman, BO; Davey, DD; Eagen, KA; Guilford, WJ; Hinchman, J; Ho, E; Koovakkat, S; Liang, A; Light, DR; Mohan, R; Ng, HP; Post, JM; Shaw, KJ; Smith, D; Subramanyam, B; Sullivan, ME; Trinh, L; Vergona, R; Walters, J; White, K; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Discovery of N-[2-[5-[Amino(imino)methyl]-2-hydroxyphenoxy]-3, 5-difluoro-6-[3-(4, 5-dihydro-1-methyl-1H-imidazol-2-yl)phenoxy]pyridin-4-yl]-N-methylgl y cine (ZK-807834): a potent, selective, and orally active inhibitor of the blood coagulation enzyme factor Xa. J Med Chem 41:3557-62 (1998) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
Inhibitor
Name:
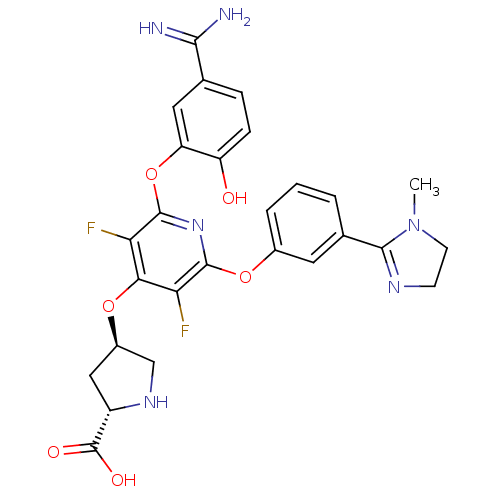
BDBM50066641
Synonyms:
(2S,4R)-4-{2-(5-Carbamimidoyl-2-hydroxy-phenoxy)-3,5-difluoro-6-[3-(1-methyl-4,5-dihydro-1H-imidazol-2-yl)-phenoxy]-pyridin-4-yloxy}-pyrrolidine-2-carboxylic acid | CHEMBL332189
Type:
Small organic molecule
Emp. Form.:
C27H26F2N6O6
Mol. Mass.:
568.5287
SMILES:
CN1CCN=C1c1cccc(Oc2nc(Oc3cc(ccc3O)C(N)=N)c(F)c(O[C@H]3CN[C@@H](C3)C(O)=O)c2F)c1 |c:4|