Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prolyl endopeptidase
Ligand
BDBM50155838
Substrate
n/a
Meas. Tech.
ChEMBL_2116616 (CHEMBL4825557)
IC50
0.200000±n/a nM
Citation
 P�tsi, HT; Kilpel�inen, TP; Auno, S; Dillemuth, PMJ; Arja, K; Lahtela-Kakkonen, MK; My�h�nen, TT; Wall�n, EAA 2-Imidazole as a Substitute for the Electrophilic Group Gives Highly Potent Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett 12:1578-1584 (2021) [PubMed] Article
P�tsi, HT; Kilpel�inen, TP; Auno, S; Dillemuth, PMJ; Arja, K; Lahtela-Kakkonen, MK; My�h�nen, TT; Wall�n, EAA 2-Imidazole as a Substitute for the Electrophilic Group Gives Highly Potent Prolyl Oligopeptidase Inhibitors. ACS Med Chem Lett 12:1578-1584 (2021) [PubMed] Article More Info.:
Target
Name:
Prolyl endopeptidase
Synonyms:
3.4.21.26 | PE | PPCE_PIG | PREP | Post-proline cleaving enzyme
Type:
n/a
Mol. Mass.:
80758.04
Organism:
Sus scrofa
Description:
n/a
Residue:
710
Sequence:
MLSFQYPDVYRDETAIQDYHGHKVCDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIRGLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILSDDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTHDGKGMFYNAYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAELSDDGRYVLLSIREGCDPVNRLWYCDLQQESNGITGILKWVKLIDNFEGEYDYVTNEGTVFTFKTNRHSPNYRLINIDFTDPEESKWKVLVPEHEKDVLEWVACVRSNFLVLCYLHDVKNTLQLHDLATGALLKIFPLEVGSVVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPRVFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISITPNYSVSRLIFVRHMGGVLAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKEGYTSPKRLTINGGSNGGLLVATCANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYGCSDSKQHFEWLIKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIVGRSRKQNNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNIDWIP
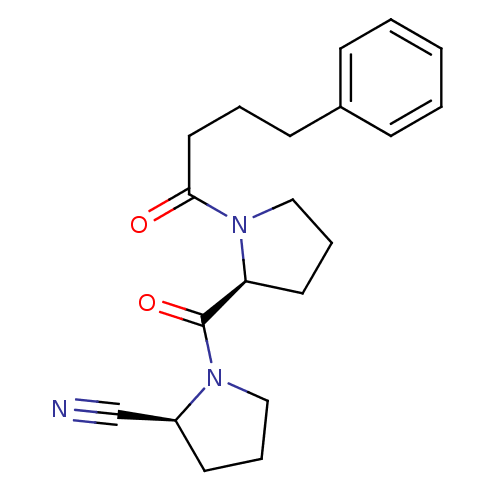
Inhibitor
Name:
BDBM50155838
Synonyms:
(S)-1-((S)-1-(4-phenylbutanoyl)pyrrolidine-2-carbonyl)pyrrolidine-2-carbonitrile | (S)-1-[(S)-1-(4-Phenyl-butyryl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonitrile | CHEMBL189620
Type:
Small organic molecule
Emp. Form.:
C20H25N3O2
Mol. Mass.:
339.4314
SMILES:
O=C(CCCc1ccccc1)N1CCC[C@H]1C(=O)N1CCC[C@H]1C#N