Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cholecystokinin receptor type A
Ligand
BDBM50329185
Substrate
n/a
Meas. Tech.
ChEMBL_674626 (CHEMBL1273773)
EC50
481.2±n/a nM
Citation
 Elliott, RL; Cameron, KO; Chin, JE; Bartlett, JA; Beretta, EE; Chen, Y; Jardine, Pda S; Dubins, JS; Gillaspy, ML; Hargrove, DM; Kalgutkar, AS; LaFlamme, JA; Lame, ME; Martin, KA; Maurer, TS; Nardone, NA; Oliver, RM; Scott, DO; Sun, D; Swick, AG; Trebino, CE; Zhang, Y Discovery of N-benzyl-2-[(4S)-4-(1H-indol-3-ylmethyl)-5-oxo-1-phenyl-4,5-dihydro-6H-[1,2,4]triazolo[4,3-a][1,5]benzodiazepin-6-yl]-N-isopropylacetamide, an orally active, gut-selective CCK1 receptor agonist for the potential treatment of obesity. Bioorg Med Chem Lett 20:6797-801 (2010) [PubMed] Article
Elliott, RL; Cameron, KO; Chin, JE; Bartlett, JA; Beretta, EE; Chen, Y; Jardine, Pda S; Dubins, JS; Gillaspy, ML; Hargrove, DM; Kalgutkar, AS; LaFlamme, JA; Lame, ME; Martin, KA; Maurer, TS; Nardone, NA; Oliver, RM; Scott, DO; Sun, D; Swick, AG; Trebino, CE; Zhang, Y Discovery of N-benzyl-2-[(4S)-4-(1H-indol-3-ylmethyl)-5-oxo-1-phenyl-4,5-dihydro-6H-[1,2,4]triazolo[4,3-a][1,5]benzodiazepin-6-yl]-N-isopropylacetamide, an orally active, gut-selective CCK1 receptor agonist for the potential treatment of obesity. Bioorg Med Chem Lett 20:6797-801 (2010) [PubMed] Article More Info.:
Target
Name:
Cholecystokinin receptor type A
Synonyms:
CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A
Type:
Enzyme Catalytic Domain
Mol. Mass.:
49676.37
Organism:
Rat
Description:
Cholecystokinin central 0 RAT::P30551
Residue:
444
Sequence:
MSHSPARQHLVESSRMDVVDSLLMNGSNITPPCELGLENETLFCLDQPQPSKEWQSALQILLYSIIFLLSVLGNTLVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLKDFIFGSAVCKTTTYFMGTSVSVSTFNLVAISLERYGAICRPLQSRVWQTKSHALKVIAATWCLSFTIMTPYPIYSNLVPFTKNNNQTANMCRFLLPSDAMQQSWQTFLLLILFLLPGIVMVVAYGLISLELYQGIKFDASQKKSAKEKKPSTGSSTRYEDSDGCYLQKSRPPRKLELQQLSSGSGGSRLNRIRSSSSAANLIAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTVSAEKHLSGTPISFILLLSYTSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGVRGEVGEEEDGRTIRALLSRYSYSHMSTSAPPP
Inhibitor
Name:
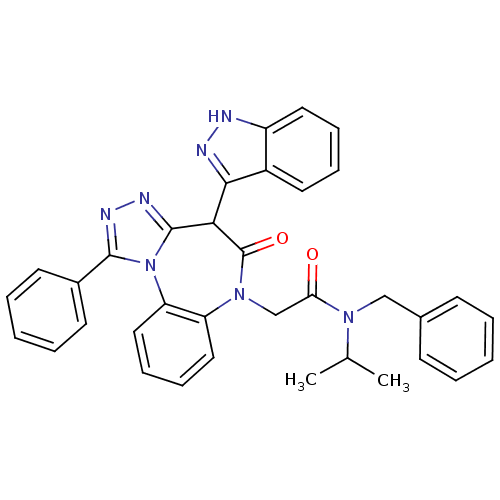
BDBM50329185
Synonyms:
2-(4-(1H-indazol-3-yl)-5-oxo-1-phenyl-4H-benzo[b][1,2,4]triazolo[4,3-d][1,4]diazepin-6(5H)-yl)-N-benzyl-N-isopropylacetamide | CHEMBL1269263
Type:
Small organic molecule
Emp. Form.:
C35H31N7O2
Mol. Mass.:
581.6663
SMILES:
CC(C)N(Cc1ccccc1)C(=O)CN1c2ccccc2-n2c(nnc2-c2ccccc2)C(c2n[nH]c3ccccc23)C1=O