Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Sodium channel protein type 8 subunit alpha
Ligand
BDBM444497
Substrate
n/a
Meas. Tech.
In Vitro Assay
IC50
402±n/a nM
Citation
 Burford, KN; Chowdhury, S; Dehnhardt, CM; Focken, T; Grimwood, ME; Hasan, SA Benzenesulfonamide compounds and their use as therapeutic agents US Patent US10815229 Publication Date 10/27/2020
Burford, KN; Chowdhury, S; Dehnhardt, CM; Focken, T; Grimwood, ME; Hasan, SA Benzenesulfonamide compounds and their use as therapeutic agents US Patent US10815229 Publication Date 10/27/2020 More Info.:
Target
Name:
Sodium channel protein type 8 subunit alpha
Synonyms:
MED | SCN8A | SCN8A_HUMAN | Sodium channel protein type 8 alpha subunit | Sodium channel protein type 8 subunit alpha | Sodium channel protein type VIII alpha subunit | Voltage-gated sodium channel subunit alpha Nav1.6 | ep nav1.6
Type:
PROTEIN
Mol. Mass.:
225271.59
Organism:
Homo sapiens (Human)
Description:
ChEMBL_1516902
Residue:
1980
Sequence:
MAARLLAPPGPDSFKPFTPESLANIERRIAESKLKKPPKADGSHREDDEDSKPKPNSDLEAGKSLPFIYGDIPQGLVAVPLEDFDPYYLTQKTFVVLNRGKTLFRFSATPALYILSPFNLIRRIAIKILIHSVFSMIIMCTILTNCVFMTFSNPPDWSKNVEYTFTGIYTFESLVKIIARGFCIDGFTFLRDPWNWLDFSVIMMAYITEFVNLGNVSALRTFRVLRALKTISVIPGLKTIVGALIQSVKKLSDVMILTVFCLSVFALIGLQLFMGNLRNKCVVWPINFNESYLENGTKGFDWEEYINNKTNFYTVPGMLEPLLCGNSSDAGQCPEGYQCMKAGRNPNYGYTSFDTFSWAFLALFRLMTQDYWENLYQLTLRAAGKTYMIFFVLVIFVGSFYLVNLILAVVAMAYEEQNQATLEEAEQKEAEFKAMLEQLKKQQEEAQAAAMATSAGTVSEDAIEEEGEEGGGSPRSSSEISKLSSKSAKERRNRRKKRKQKELSEGEEKGDPEKVFKSESEDGMRRKAFRLPDNRIGRKFSIMNQSLLSIPGSPFLSRHNSKSSIFSFRGPGRFRDPGSENEFADDEHSTVEESEGRRDSLFIPIRARERRSSYSGYSGYSQGSRSSRIFPSLRRSVKRNSTVDCNGVVSLIGGPGSHIGGRLLPEATTEVEIKKKGPGSLLVSMDQLASYGRKDRINSIMSVVTNTLVEELEESQRKCPPCWYKFANTFLIWECHPYWIKLKEIVNLIVMDPFVDLAITICIVLNTLFMAMEHHPMTPQFEHVLAVGNLVFTGIFTAEMFLKLIAMDPYYYFQEGWNIFDGFIVSLSLMELSLADVEGLSVLRSFRLLRVFKLAKSWPTLNMLIKIIGNSVGALGNLTLVLAIIVFIFAVVGMQLFGKSYKECVCKINQDCELPRWHMHDFFHSFLIVFRVLCGEWIETMWDCMEVAGQAMCLIVFMMVMVIGNLVVLNLFLALLLSSFSADNLAATDDDGEMNNLQISVIRIKKGVAWTKLKVHAFMQAHFKQREADEVKPLDELYEKKANCIANHTGADIHRNGDFQKNGNGTTSGIGSSVEKYIIDEDHMSFINNPNLTVRVPIAVGESDFENLNTEDVSSESDPEGSKDKLDDTSSSEGSTIDIKPEVEEVPVEQPEEYLDPDACFTEGCVQRFKCCQVNIEEGLGKSWWILRKTCFLIVEHNWFETFIIFMILLSSGALAFEDIYIEQRKTIRTILEYADKVFTYIFILEMLLKWTAYGFVKFFTNAWCWLDFLIVAVSLVSLIANALGYSELGAIKSLRTLRALRPLRALSRFEGMRVVVNALVGAIPSIMNVLLVCLIFWLIFSIMGVNLFAGKYHYCFNETSEIRFEIEDVNNKTECEKLMEGNNTEIRWKNVKINFDNVGAGYLALLQVATFKGWMDIMYAAVDSRKPDEQPKYEDNIYMYIYFVIFIIFGSFFTLNLFIGVIIDNFNQQKKKFGGQDIFMTEEQKKYYNAMKKLGSKKPQKPIPRPLNKIQGIVFDFVTQQAFDIVIMMLICLNMVTMMVETDTQSKQMENILYWINLVFVIFFTCECVLKMFALRHYYFTIGWNIFDFVVVILSIVGMFLADIIEKYFVSPTLFRVIRLARIGRILRLIKGAKGIRTLLFALMMSLPALFNIGLLLFLVMFIFSIFGMSNFAYVKHEAGIDDMFNFETFGNSMICLFQITTSAGWDGLLLPILNRPPDCSLDKEHPGSGFKGDCGNPSVGIFFFVSYIIISFLIVVNMYIAIILENFSVATEESADPLSEDDFETFYEIWEKFDPDATQFIEYCKLADFADALEHPLRVPKPNTIELIAMDLPMVSGDRIHCLDILFAFTKRVLGDSGELDILRQQMEERFVASNPSKVSYEPITTTLRRKQEEVSAVVLQRAYRGHLARRGFICKKTTSNKLENGGTHREKKESTPSTASLPSYDSVTKPEKEKQQRAEEGRRERAKRQKEVRESKC
Inhibitor
Name:
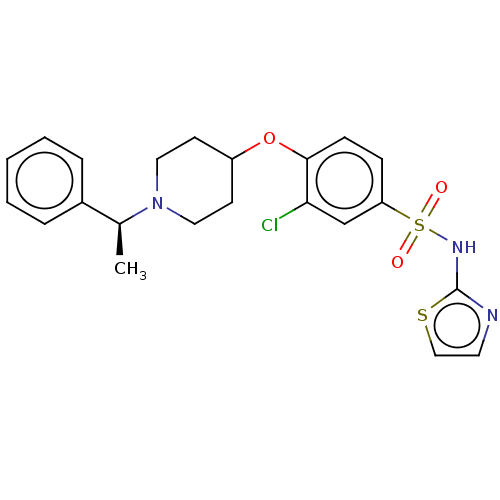
BDBM444497
Synonyms:
(S)-3-chloro-4-((1-(1-phenylethyl)piperidin-4-yl)oxy)-N-(thiazol-2-yl)benzenesulfonamide 2,2,2-trifluoroacetate | US10662184, Example 58 | US10815229, Example 58 | US11299490, Example 58
Type:
Small organic molecule
Emp. Form.:
C22H24ClN3O3S2
Mol. Mass.:
478.027
SMILES:
C[C@H](N1CCC(CC1)Oc1ccc(cc1Cl)S(=O)(=O)Nc1nccs1)c1ccccc1 |r|