Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
E3 ubiquitin-protein ligase Mdm2 [17-125]
Ligand
BDBM224323
Substrate
n/a
Meas. Tech.
FRET Assay
Temperature
298.15±n/a K
IC50
0.670±n/a nM
Comments
extracted
Citation
 Cammarano, CM; Christopher, MP; Dinsmore, C; Doll, RJ; Fradera Llinas, FX; Li, C; Machacek, M; Martinez, M; Nair, LG; Pan, W; Reutershan, MH; Shizuka, M; Steinhuebel, DP; Sun, B; Thompson, CF; Trotter, BW; Wang, Y; Yang, L; Bogen, SL; Voss, ME; Panda, J; Kurissery, AT 2,6,7,8 substituted purines as HDM2 inhibitors US Patent US9540377 Publication Date 1/10/2017
Cammarano, CM; Christopher, MP; Dinsmore, C; Doll, RJ; Fradera Llinas, FX; Li, C; Machacek, M; Martinez, M; Nair, LG; Pan, W; Reutershan, MH; Shizuka, M; Steinhuebel, DP; Sun, B; Thompson, CF; Trotter, BW; Wang, Y; Yang, L; Bogen, SL; Voss, ME; Panda, J; Kurissery, AT 2,6,7,8 substituted purines as HDM2 inhibitors US Patent US9540377 Publication Date 1/10/2017 More Info.:
Target
Name:
E3 ubiquitin-protein ligase Mdm2 [17-125]
Synonyms:
Double minute 2 protein | E3 ubiquitin-protein ligase Mdm2 | MDM2 | MDM2_HUMAN | p53-binding protein Mdm2
Type:
Enzyme Catalytic Domain
Mol. Mass.:
12522.24
Organism:
Homo sapiens (Human)
Description:
Residue 17 to 125
Residue:
109
Sequence:
SQIPASEQETLVRPKPLLLKLLKSVGAQKDTYTMKEVLFYLGQYIMTKRLYDEKQQHIVYCSNDLLGDLFGVPSFSVKEHRKIYTMIYRNLVVVNQQESSDSGTSVSEN
Inhibitor
Name:
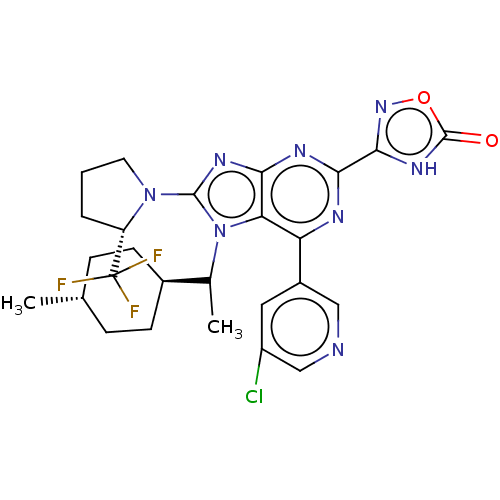
BDBM224323
Synonyms:
3-{6-(5-chloropyridin-3-yl)-7-[1- (trans-4-methylcyclohexyl)ethyl]-8- [(2s)-2-(trifluoromethyl)pyrrolidin- 1-yl]-7h-purin-2-yl}-1,2,4- oxadiazol-5(4h)-one (mixture of diastereomers) | US9540377, 18.49
Type:
Small organic molecule
Emp. Form.:
C26H28ClF3N8O2
Mol. Mass.:
577.001
SMILES:
CC([C@H]1CC[C@H](C)CC1)n1c(nc2nc(nc(-c3cncc(Cl)c3)c12)-c1noc(=O)[nH]1)N1CCC[C@H]1C(F)(F)F |r,wU:5.5,wD:2.1,35.41,(.82,-2.85,;1.91,-1.76,;3.4,-2.16,;3.8,-3.65,;5.29,-4.05,;6.37,-2.96,;7.86,-3.36,;5.98,-1.47,;4.49,-1.07,;1.51,-.28,;2.42,.97,;1.51,2.22,;.05,1.74,;-1.28,2.51,;-2.62,1.74,;-2.62,.2,;-1.28,-.57,;-1.28,-2.11,;.05,-2.88,;.05,-4.42,;-1.28,-5.19,;-2.62,-4.42,;-3.95,-5.19,;-2.62,-2.88,;.05,.2,;-3.95,2.51,;-3.95,4.05,;-5.42,4.53,;-6.32,3.28,;-7.86,3.28,;-5.42,2.03,;3.96,.97,;4.86,-.28,;6.33,.2,;6.33,1.74,;4.86,2.22,;4.47,3.7,;5.55,4.79,;4.07,5.19,;2.98,4.1,)|