Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2A6
Ligand
BDBM21358
Substrate
BDBM12342
Meas. Tech.
Cytochrome P450 Inhibition
pH
7.4±n/a
Temperature
310.15±n/a K
IC50
41000±n/a nM
Citation
 Cole, DC; Stock, JR; Lennox, WJ; Bernotas, RC; Ellingboe, JW; Boikess, S; Coupet, J; Smith, DL; Leung, L; Zhang, GM; Feng, X; Kelly, MF; Galante, R; Huang, P; Dawson, LA; Marquis, K; Rosenzweig-Lipson, S; Beyer, CE; Schechter, LE Discovery of N1-(6-chloroimidazo[2,1-b][1,3]thiazole-5-sulfonyl)tryptamine as a potent, selective, and orally active 5-HT(6) receptor agonist. J Med Chem 50:5535-8 (2007) [PubMed] Article
Cole, DC; Stock, JR; Lennox, WJ; Bernotas, RC; Ellingboe, JW; Boikess, S; Coupet, J; Smith, DL; Leung, L; Zhang, GM; Feng, X; Kelly, MF; Galante, R; Huang, P; Dawson, LA; Marquis, K; Rosenzweig-Lipson, S; Beyer, CE; Schechter, LE Discovery of N1-(6-chloroimidazo[2,1-b][1,3]thiazole-5-sulfonyl)tryptamine as a potent, selective, and orally active 5-HT(6) receptor agonist. J Med Chem 50:5535-8 (2007) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2A6
Synonyms:
1,4-cineole 2-exo-monooxygenase | 1.14.13.- | CP2A6_HUMAN | CYP2A3 | CYP2A6 | CYPIIA6 | Coumarin 7-hydroxylase | Cytochrome P450 2A6 | Cytochrome P450 IIA3 | Cytochrome P450(I)
Type:
Protein
Mol. Mass.:
56514.34
Organism:
Homo sapiens (Human)
Description:
P11509
Residue:
494
Sequence:
MLASGMLLVALLVCLTVMVLMSVWQQRKSKGKLPPGPTPLPFIGNYLQLNTEQMYNSLMKISERYGPVFTIHLGPRRVVVLCGHDAVREALVDQAEEFSGRGEQATFDWVFKGYGVVFSNGERAKQLRRFSIATLRDFGVGKRGIEERIQEEAGFLIDALRGTGGANIDPTFFLSRTVSNVISSIVFGDRFDYKDKEFLSLLRMMLGIFQFTSTSTGQLYEMFSSVMKHLPGPQQQAFQLLQGLEDFIAKKVEHNQRTLDPNSPRDFIDSFLIRMQEEEKNPNTEFYLKNLVMTTLNLFIGGTETVSTTLRYGFLLLMKHPEVEAKVHEEIDRVIGKNRQPKFEDRAKMPYMEAVIHEIQRFGDVIPMSLARRVKKDTKFRDFFLPKGTEVYPMLGSVLRDPSFFSNPQDFNPQHFLNEKGQFKKSDAFVPFSIGKRNCFGEGLARMELFLFFTTVMQNFRLKSSQSPKDIDVSPKHVGFATIPRNYTMSFLPR
Inhibitor
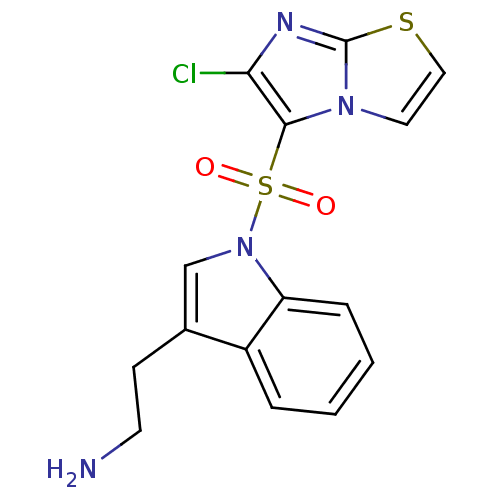
Name:
BDBM21358
Synonyms:
2-[1-({6-chloroimidazo[2,1-b][1,3]thiazole-5-}sulfonyl)-1H-indol-3-yl]ethan-1-amine | N1-(6-chloroimidazo[2,1-b][1,3]thiazole-5-sulfonyl)tryptamine | SAX-187 | WAY-181187
Type:
Small organic molecule
Emp. Form.:
C15H13ClN4O2S2
Mol. Mass.:
380.872
SMILES:
NCCc1cn(c2ccccc12)S(=O)(=O)c1c(Cl)nc2sccn12