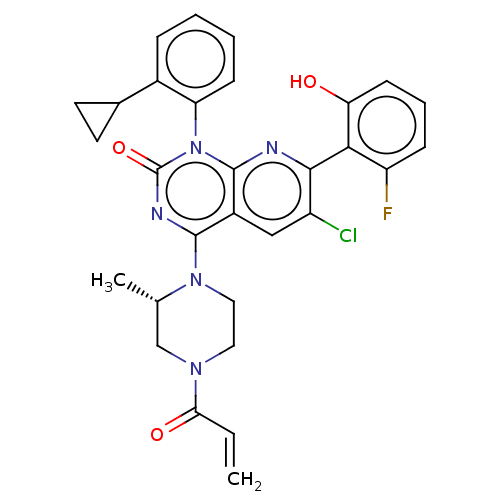
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
GTPase KRas
Ligand
BDBM50514401
Substrate
n/a
Meas. Tech.
ChEMBL_1856529 (CHEMBL4357258)
IC50
225±n/a nM
Citation
 Lanman, BA; Allen, JR; Allen, JG; Amegadzie, AK; Ashton, KS; Booker, SK; Chen, JJ; Chen, N; Frohn, MJ; Goodman, G; Kopecky, DJ; Liu, L; Lopez, P; Low, JD; Ma, V; Minatti, AE; Nguyen, TT; Nishimura, N; Pickrell, AJ; Reed, AB; Shin, Y; Siegmund, AC; Tamayo, NA; Tegley, CM; Walton, MC; Wang, HL; Wurz, RP; Xue, M; Yang, KC; Achanta, P; Bartberger, MD; Canon, J; Hollis, LS; McCarter, JD; Mohr, C; Rex, K; Saiki, AY; San Miguel, T; Volak, LP; Wang, KH; Whittington, DA; Zech, SG; Lipford, JR; Cee, VJ Discovery of a Covalent Inhibitor of KRAS J Med Chem 63:52-65 (2020) [PubMed] Article
Lanman, BA; Allen, JR; Allen, JG; Amegadzie, AK; Ashton, KS; Booker, SK; Chen, JJ; Chen, N; Frohn, MJ; Goodman, G; Kopecky, DJ; Liu, L; Lopez, P; Low, JD; Ma, V; Minatti, AE; Nguyen, TT; Nishimura, N; Pickrell, AJ; Reed, AB; Shin, Y; Siegmund, AC; Tamayo, NA; Tegley, CM; Walton, MC; Wang, HL; Wurz, RP; Xue, M; Yang, KC; Achanta, P; Bartberger, MD; Canon, J; Hollis, LS; McCarter, JD; Mohr, C; Rex, K; Saiki, AY; San Miguel, T; Volak, LP; Wang, KH; Whittington, DA; Zech, SG; Lipford, JR; Cee, VJ Discovery of a Covalent Inhibitor of KRAS J Med Chem 63:52-65 (2020) [PubMed] Article More Info.:
Target
Name:
GTPase KRas
Synonyms:
GTPase KRas, N-terminally processed | K-Ras 2 | KRAS | KRAS2 | Ki-Ras | RASK2 | RASK_HUMAN | c-K-ras | c-Ki-ras
Type:
PROTEIN
Mol. Mass.:
21656.10
Organism:
Homo sapiens (Human)
Description:
ChEMBL_1476955
Residue:
189
Sequence:
MTEYKLVVVGAGGVGKSALTIQLIQNHFVDEYDPTIEDSYRKQVVIDGETCLLDILDTAGQEEYSAMRDQYMRTGEGFLCVFAINNTKSFEDIHHYREQIKRVKDSEDVPMVLVGNKCDLPSRTVDTKQAQDLARSYGIPFIETSAKTRQRVEDAFYTLVREIRQYRLKKISKEEKTPGCVKIKKCIIM
Inhibitor
Name:
BDBM50514401
Synonyms:
CHEMBL4549665
Type:
Small organic molecule
Emp. Form.:
C30H27ClFN5O3
Mol. Mass.:
560.018
SMILES:
C[C@H]1CN(CCN1c1nc(=O)n(-c2ccccc2C2CC2)c2nc(c(Cl)cc12)-c1c(O)cccc1F)C(=O)C=C |r,wU:1.0,(10.67,-11.53,;10.68,-10,;9.35,-9.21,;9.35,-7.67,;10.7,-6.91,;12.01,-7.68,;12.01,-9.23,;13.33,-10,;13.32,-11.53,;14.65,-12.3,;13.32,-13.05,;15.99,-11.54,;17.33,-12.3,;18.66,-11.54,;19.99,-12.3,;19.99,-13.85,;18.66,-14.62,;17.32,-13.85,;15.99,-14.63,;14.45,-14.64,;15.23,-15.98,;15.99,-10.01,;17.33,-9.23,;17.32,-7.68,;15.99,-6.92,;15.98,-5.38,;14.66,-7.69,;14.66,-9.23,;18.65,-6.91,;19.99,-7.68,;19.99,-9.22,;21.32,-6.91,;21.32,-5.36,;19.97,-4.6,;18.64,-5.38,;17.3,-4.61,;8.02,-6.89,;8.04,-5.35,;6.68,-7.65,;5.35,-6.87,)|