Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Suppressor of tumorigenicity 14 protein
Ligand
BDBM50421510
Substrate
n/a
Meas. Tech.
ChEBML_105805
Ki
0.920000±n/a nM
Citation
 Long, YQ; Lee, SL; Lin, CY; Enyedy, IJ; Wang, S; Li, P; Dickson, RB; Roller, PP Synthesis and evaluation of the sunflower derived trypsin inhibitor as a potent inhibitor of the type II transmembrane serine protease, matriptase. Bioorg Med Chem Lett 11:2515-9 (2001) [PubMed] Article
Long, YQ; Lee, SL; Lin, CY; Enyedy, IJ; Wang, S; Li, P; Dickson, RB; Roller, PP Synthesis and evaluation of the sunflower derived trypsin inhibitor as a potent inhibitor of the type II transmembrane serine protease, matriptase. Bioorg Med Chem Lett 11:2515-9 (2001) [PubMed] Article More Info.:
Target
Name:
Suppressor of tumorigenicity 14 protein
Synonyms:
Epithin | Hepatocyte growth factor activator/Serine protease hepsin/Suppressor of tumorigenicity 14 protein | MT-SP1 | Membrane-type serine protease 1 | PRSS14 | Prostamin | SNC19 | ST14 | ST14_HUMAN | Serine protease TADG-15 | Suppressor of tumorigenicity 14 protein | Suppressor of tumorigenicity protein 14 | TADG15
Type:
Single-pass type II membrane protein
Mol. Mass.:
94769.23
Organism:
Homo sapiens (Human)
Description:
Q9Y5Y6
Residue:
855
Sequence:
MGSDRARKGGGGPKDFGAGLKYNSRHEKVNGLEEGVEFLPVNNVKKVEKHGPGRWVVLAAVLIGLLLVLLGIGFLVWHLQYRDVRVQKVFNGYMRITNENFVDAYENSNSTEFVSLASKVKDALKLLYSGVPFLGPYHKESAVTAFSEGSVIAYYWSEFSIPQHLVEEAERVMAEERVVMLPPRARSLKSFVVTSVVAFPTDSKTVQRTQDNSCSFGLHARGVELMRFTTPGFPDSPYPAHARCQWALRGDADSVLSLTFRSFDLASCDERGSDLVTVYNTLSPMEPHALVQLCGTYPPSYNLTFHSSQNVLLITLITNTERRHPGFEATFFQLPRMSSCGGRLRKAQGTFNSPYYPGHYPPNIDCTWNIEVPNNQHVKVRFKFFYLLEPGVPAGTCPKDYVEINGEKYCGERSQFVVTSNSNKITVRFHSDQSYTDTGFLAEYLSYDSSDPCPGQFTCRTGRCIRKELRCDGWADCTDHSDELNCSCDAGHQFTCKNKFCKPLFWVCDSVNDCGDNSDEQGCSCPAQTFRCSNGKCLSKSQQCNGKDDCGDGSDEASCPKVNVVTCTKHTYRCLNGLCLSKGNPECDGKEDCSDGSDEKDCDCGLRSFTRQARVVGGTDADEGEWPWQVSLHALGQGHICGASLISPNWLVSAAHCYIDDRGFRYSDPTQWTAFLGLHDQSQRSAPGVQERRLKRIISHPFFNDFTFDYDIALLELEKPAEYSSMVRPICLPDASHVFPAGKAIWVTGWGHTQYGGTGALILQKGEIRVINQTTCENLLPQQITPRMMCVGFLSGGVDSCQGDSGGPLSSVEADGRIFQAGVVSWGDGCAQRNKPGVYTRLPLFRDWIKENTGV
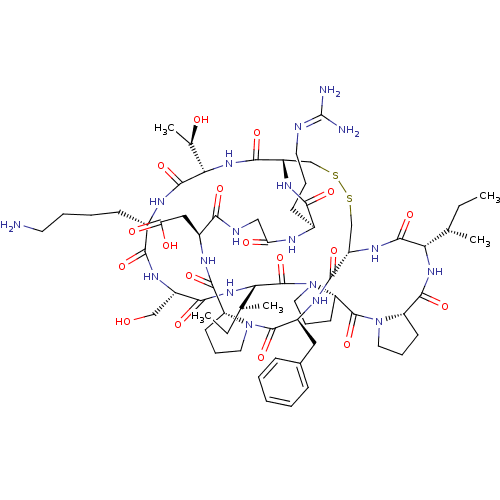
Inhibitor
Name:
BDBM50421510
Synonyms:
CHEMBL239127
Type:
Small organic molecule
Emp. Form.:
C67H104N18O18S2
Mol. Mass.:
1513.782
SMILES:
[#6]-[#6]-[#6@H](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6]-1=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-2)-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#6]-[#6]