Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Inositol 1,4,5-trisphosphate receptor type 1
Ligand
BDBM50184324
Substrate
n/a
Meas. Tech.
ChEMBL_337345 (CHEMBL867179)
EC50
4±n/a nM
Citation
 Terauchi, M; Abe, H; Tovey, SC; Dedos, SG; Taylor, CW; Paul, M; Trusselle, M; Potter, BV; Matsuda, A; Shuto, S A systematic study of C-glucoside trisphosphates as myo-inositol trisphosphate receptor ligands. Synthesis of beta-C-glucoside trisphosphates based on the conformational restriction strategy. J Med Chem 49:1900-9 (2006) [PubMed] Article
Terauchi, M; Abe, H; Tovey, SC; Dedos, SG; Taylor, CW; Paul, M; Trusselle, M; Potter, BV; Matsuda, A; Shuto, S A systematic study of C-glucoside trisphosphates as myo-inositol trisphosphate receptor ligands. Synthesis of beta-C-glucoside trisphosphates based on the conformational restriction strategy. J Med Chem 49:1900-9 (2006) [PubMed] Article More Info.:
Target
Name:
Inositol 1,4,5-trisphosphate receptor type 1
Synonyms:
ITPR1_RAT | Insp3r | Itpr1
Type:
PROTEIN
Mol. Mass.:
313234.63
Organism:
Rattus norvegicus
Description:
ChEMBL_818416
Residue:
2750
Sequence:
MSDKMSSFLHIGDICSLYAEGSTNGFISTLGLVDDRCVVQPEAGDLNNPPKKFRDCLFKLCPMNRYSAQKQFWKAAKPGANSTTDAVLLNKLHHAADLEKKQNETENRKLLGTVIQYGNVIQLLHLKSNKYLTVNKRLPALLEKNAMRVTLDEAGNEGSWFYIQPFYKLRSIGDSVVIGDKVVLNPVNAGQPLHASSHQLVDNPGCNEVNSVNCNTSWKIVLFMKWSDNKDDILKGGDVVRLFHAEQEKFLTCDEHRKKQHVFLRTTGRQSATSATSSKALWEVEVVQHDPCRGGAGYWNSLFRFKHLATGHYLAAEVDPDFEEECLEFQPSVDPDQDASRSRLRNAQEKMVYSLVSVPEGNDISSIFELDPTTLRGGDSLVPRNSYVRLRHLCTNTWVHSTNIPIDKEEEKPVMLKIGTSPLKEDKEAFAIVPVSPAEVRDLDFANDASKVLGSIAGKLEKGTITQNERRSVTKLLEDLVYFVTGGTNSGQDVLEVVFSKPNRERQKLMREQNILKQIFKLLQAPFTDCGDGPMLRLEELGDQRHAPFRHICRLCYRVLRHSQQDYRKNQEYIAKQFGFMQKQIGYDVLAEDTITALLHNNRKLLEKHITAAEIDTFVSLVRKNREPRFLDYLSDLCVSMNKSIPVTQELICKAVLNPTNADILIETKLVLSRFEFEGVSTGENALEAGEDEEEVWLFWRDSNKEIRSKSVRELAQDAKEGQKEDRDVLSYYRYQLNLFARMCLDRQYLAINEISGQLDVDLILRCMSDENLPYDLRASFCRLMLHMHVDRDPQEQVTPVKYARLWSEIPSEIAIDDYDSSGASKDEIKERFAQTMEFVEEYLRDVVCQRFPFSDKEKNKLTFEVVNLARNLIYFGFYNFSDLLRLTKILLAILDCVHVTTIFPISKMTKGEENKGSNVMRSIHGVGELMTQVVLRGGGFLPMTPMAAAPEGNVKQAEPEKEDIMVMDTKLKIIEILQFILNVRLDYRISCLLCIFKREFDESNSQSSETSSGNSSQEGPSNVPGALDFEHIEEQAEGIFGGSEENTPLDLDDHGGRTFLRVLLHLTMHDYPPLVSGALQLLFRHFSQRQEVLQAFKQVQLLVTSQDVDNYKQIKQDLDQLRSIVEKSELWVYKGQGPDEPMDGASGENEHKKTEEGTSKPLKHESTSSYNYRVVKEILIRLSKLCVQESASVRKSRKQQQRLLRNMGAHAVVLELLQIPYEKAEDTKMQEIMRLAHEFLQNFCAGNQQNQALLHKHINLFLNPGILEAVTMQHIFMNNFQLCSEINERVVQHFVHCIETHGRNVQYIKFLQTIVKAEGKFIKKCQDMVMAELVNSGEDVLVFYNDRASFQTLIQMMRSERDRMDENSPLFMYHIHLVELLAVCTEGKNVYTEIKCNSLLPLDDIVRVVTHEDCIPEVKIAYINFLNHCYVDTEVEMKEIYTSNHMWKLFENFLVDICRACNNTSDRKHADSVLEKYVTEIVMSIVTTFFSSPFSDQSTTLQTRQPVFVQLLQGVFRVYHCNWLMPSQKASVESCIRVLSDVAKSRAIAIPVDLDSQVNNLFLKSHNIVQKTAMNWRLSARNAARRDSVLAASRDYRNIIERLQDIVSALEDRLRPLVQAELSVLVDVLHRPELLFPENTDARRKCESGGFICKLIKHTKQLLEENEEKLCIKVLQTLREMMTKDRGYGEKQISIDELENAELPQPPEAENSTEQELEPSPPLRQLEDHKRGEALRQILVNRYYGNIRPSGRRESLTSFGNGPLSPGGPSKPGGGGGGPGSGSTSRGEMSLAEVQCHLDKEGASNLVIDLIMNASSDRVFHESILLAIALLEGGNTTIQHSFFCRLTEDKKSEKFFKVFYDRMKVAQQEIKATVTVNTSDLGNKKKDDEVDRDAPSRKKAKEPTTQITEEVRDQLLEASAATRKAFTTFRREADPDDHYQSGEGTQATTDKAKDDLEMSAVITIMQPILRFLQLLCENHNRDLQNFLRCQNNKTNYNLVCETLQFLDCICGSTTGGLGLLGLYINEKNVALINQTLESLTEYCQGPCHENQNCIATHESNGIDIITALILNDINPLGKKRMDLVLELKNNASKLLLAIMESRHDSENAERILYNMRPKELVEVIKKAYMQGEVEFEDGENGEDGAASPRNVGHNIYILAHQLARHNKELQTMLKPGGQVDGDEALEFYAKHTAQIEIVRLDRTMEQIVFPVPSICEFLTKESKLRIYYTTERDEQGSKINDFFLRSEDLFNEMNWQKKLRAQPVLYWCARNMSFWSSISFNLAVLMNLLVAFFYPFKGVRGGTLEPHWSGLLWTAMLISLAIVIALPKPHGIRALIASTILRLIFSVGLQPTLFLLGAFNVCNKIIFLMSFVGNCGTFTRGYRAMVLDVEFLYHLLYLLICAMGLFVHEFFYSLLLFDLVYREETLLNVIKSVTRNGRPIILTAALALILVYLFSIVGYLFFKDDFILEVDRLPNETAGPETGESLANDFLYSDVCRVETGENCTSPAPKEELLPVEETEQDKEHTCETLLMCIVTVLSHGLRSGGGVGDVLRKPSKEEPLFAARVIYDLLFFFMVIIIVLNLIFGVIIDTFADLRSEKQKKEEILKTTCFICGLERDKFDNKTVTFEEHIKEEHNMWHYLCFIVLVKVKDSTEYTGPESYVAEMIRERNLDWFPRMRAMSLVSSDSEGEQNELRNLQEKLESTMKLVTNLSGQLSELKDQMTEQRKQKQRIGLLGHPPHMNVNPQQPA
Inhibitor
Name:
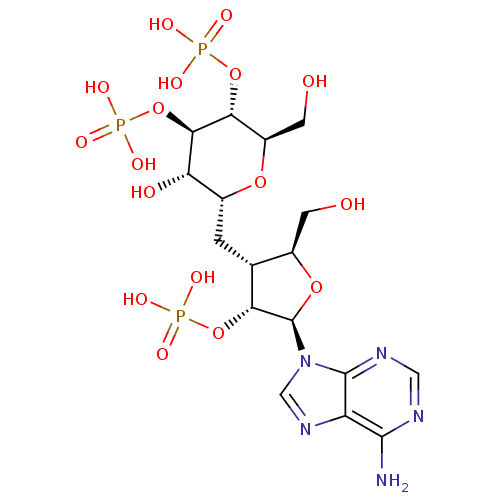
BDBM50184324
Synonyms:
CHEMBL383049 | phosphoric acid mono-{(2R,3R,4R,5S,6R)-6-[(2S,3R,4R,5R)-5-(6-amino-purin-9-yl)-2-hydroxymethyl-4-phosphonooxy-tetrahydro-furan-3-ylmethyl]-5-hydroxy-2-hydroxymethyl-4-phosphonooxy-tetrahydro-pyran-3-yl} ester
Type:
Small organic molecule
Emp. Form.:
C17H28N5O17P3
Mol. Mass.:
667.3488
SMILES:
Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](C[C@H]2O[C@H](CO)[C@@H](OP(O)(O)=O)[C@H](OP(O)(O)=O)[C@H]2O)[C@H]1OP(O)(O)=O