Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50258200
Substrate
n/a
Meas. Tech.
ChEMBL_497350 (CHEMBL999389)
IC50
13200±n/a nM
Citation
 Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem 43:1621-31 (2008) [PubMed] Article
Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem 43:1621-31 (2008) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
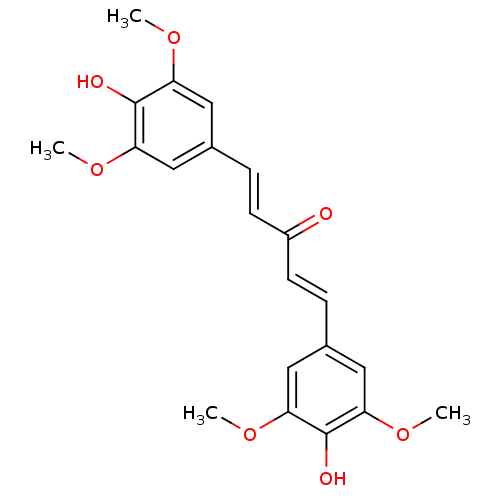
Inhibitor
Name:
BDBM50258200
Synonyms:
(1E,4E)-1,5-bis(4-hydroxy-3,5-dimethoxyphenyl)penta-1,4-dien-3-one | 1,5-bis(4-hydroxy-3,5-dimethoxyphenyl)penta-1,4-dien-3-one | CHEMBL493001 | Go-Y026
Type:
Small organic molecule
Emp. Form.:
C21H22O7
Mol. Mass.:
386.3952
SMILES:
COc1cc(\C=C\C(=O)\C=C\c2cc(OC)c(O)c(OC)c2)cc(OC)c1O