Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cysteinyl leukotriene receptor 1
Ligand
BDBM50070921
Substrate
n/a
Meas. Tech.
ChEBML_99992
Ki
24000±n/a nM
Citation
 Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett 5:1365-1370 (1995) Article
Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett 5:1365-1370 (1995) Article More Info.:
Target
Name:
Cysteinyl leukotriene receptor 1
Synonyms:
CLTR1_CAVPO | CYSLTR1
Type:
Enzyme Catalytic Domain
Mol. Mass.:
39007.58
Organism:
GUINEA PIG
Description:
Leukotriene D4 0 GUINEA PIG::Q2NNR5
Residue:
340
Sequence:
MDETGNPTIPPASNNTCYDSIDDFRNQVYSTLYSMISVVGFFGNGFVLYVLVKTYHEKSAFQVYMINLAVADLLCVCTLPLRVAYYVHKGIWLFGDFLCRLSTYALYVNLYCSIFFMTAMSFFRCVAIVFPVQNISLVTQKKARLVCIAIWMFVILTSSPFLMANTYKDEKNNTKCFEPPQDNQAKNYVLILHYVSLFIGFIIPFITIIVCYTMIIFTLLKSSMKKNLSSRKRAIGMIIVVTAAFLVSFMPYHIQRTIHLHFLHNKTKPCDSILRMQKSVVITLSLAASNCCFDPLLYFFSGGNFRRRLSTIRKYSLSSMTYIPKKKTSLPQKGKDICKE
Inhibitor
Name:
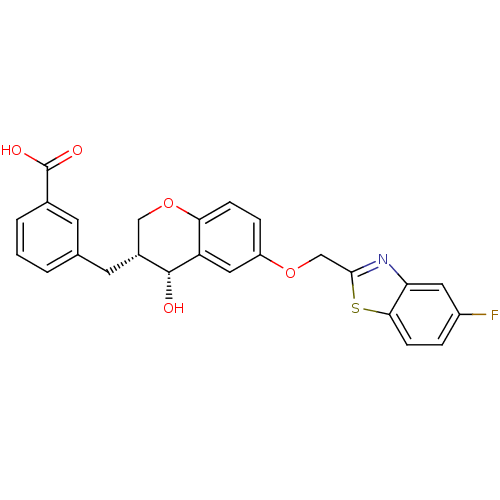
BDBM50070921
Synonyms:
3-[(3R,4R)-6-(5-Fluoro-benzothiazol-2-ylmethoxy)-4-hydroxy-chroman-3-ylmethyl]-benzoic acid | 3-[6-(5-Fluoro-benzothiazol-2-ylmethoxy)-4-hydroxy-chroman-3-ylmethyl]-benzoic acid | CHEMBL442150
Type:
Small organic molecule
Emp. Form.:
C25H20FNO5S
Mol. Mass.:
465.493
SMILES:
O[C@@H]1[C@H](Cc2cccc(c2)C(O)=O)COc2ccc(OCc3nc4cc(F)ccc4s3)cc12