Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50344100
Substrate
n/a
Meas. Tech.
ChEMBL_748610 (CHEMBL1780463)
IC50
>30000±n/a nM
Citation
 Chen, JJ; Nguyen, T; D'Amico, DC; Qian, W; Human, J; Aya, T; Biswas, K; Fotsch, C; Han, N; Liu, Q; Nishimura, N; Peterkin, TA; Yang, K; Zhu, J; Riahi, BB; Hungate, RW; Andersen, NG; Colyer, JT; Faul, MM; Kamassah, A; Wang, J; Jona, J; Kumar, G; Johnson, E; Askew, BC 3-Oxo-2-piperazinyl acetamides as potent bradykinin B1 receptor antagonists for the treatment of pain and inflammation. Bioorg Med Chem Lett 21:3384-9 (2011) [PubMed] Article
Chen, JJ; Nguyen, T; D'Amico, DC; Qian, W; Human, J; Aya, T; Biswas, K; Fotsch, C; Han, N; Liu, Q; Nishimura, N; Peterkin, TA; Yang, K; Zhu, J; Riahi, BB; Hungate, RW; Andersen, NG; Colyer, JT; Faul, MM; Kamassah, A; Wang, J; Jona, J; Kumar, G; Johnson, E; Askew, BC 3-Oxo-2-piperazinyl acetamides as potent bradykinin B1 receptor antagonists for the treatment of pain and inflammation. Bioorg Med Chem Lett 21:3384-9 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
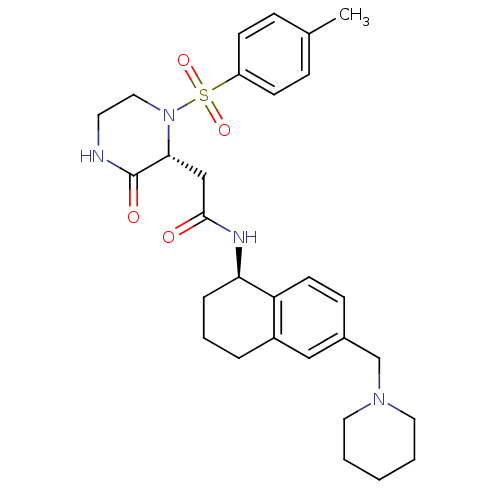
Inhibitor
Name:
BDBM50344100
Synonyms:
2-((2R)-1-((4-methylphenyl)sulfonyl)-3-oxo-2-piperazinyl)-N-((1R)-6-(1-piperidinylmethyl)-1,2,3,4-tetrahydro-1-naphthalenyl)acetamide | CHEMBL1777958
Type:
Small organic molecule
Emp. Form.:
C29H38N4O4S
Mol. Mass.:
538.701
SMILES:
Cc1ccc(cc1)S(=O)(=O)N1CCNC(=O)[C@H]1CC(=O)N[C@@H]1CCCc2cc(CN3CCCCC3)ccc12 |r|