Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Tyrosine-protein kinase BTK [386-659]
Ligand
BDBM472480
Substrate
n/a
Meas. Tech.
Kinase Lanthascreen Binding Assay
IC50
7.94±n/a nM
Citation
 Arora, N; Bacani, GM; Barbay, JK; Bembenek, SD; Cai, M; Chen, W; Deckhut, CP; Edwards, JP; Ghosh, B; Hao, B; Kreutter, KD; Li, G; Tichenor, MS; Venable, JD; Wei, J; Wiener, JJ; Wu, Y; Zhu, Y; Zhang, F; Zhang, Z; Xiao, K Inhibitors of Bruton's tyrosine kinase and methods of their use US Patent US10822348 Publication Date 11/3/2020
Arora, N; Bacani, GM; Barbay, JK; Bembenek, SD; Cai, M; Chen, W; Deckhut, CP; Edwards, JP; Ghosh, B; Hao, B; Kreutter, KD; Li, G; Tichenor, MS; Venable, JD; Wei, J; Wiener, JJ; Wu, Y; Zhu, Y; Zhang, F; Zhang, Z; Xiao, K Inhibitors of Bruton's tyrosine kinase and methods of their use US Patent US10822348 Publication Date 11/3/2020 More Info.:
Target
Name:
Tyrosine-protein kinase BTK [386-659]
Synonyms:
AGMX1 | ATK | BPK | BTK | BTK_HUMAN | Tyrosine-protein kinase BTK (386-659)
Type:
Enzyme Catalytic Domain
Mol. Mass.:
31815.18
Organism:
Homo sapiens (Human)
Description:
aa 386-659
Residue:
274
Sequence:
STAGLGYGSWEIDPKDLTFLKELGTGQFGVVKYGKWRGQYDVAIKMIKEGSMSEDEFIEEAKVMMNLSHEKLVQLYGVCTKQRPIFIITEYMANGCLLNYLREMRHRFQTQQLLEMCKDVCEAMEYLESKQFLHRDLAARNCLVNDQGVVKVSDFGLSRYVLDDEYTSSVGSKFPVRWSPPEVLMYSKFSSKSDIWAFGVLMWEIYSLGKMPYERFTNSETAEHIAQGLRLYRPHLASEKVYTIMYSCWHEKADERPTFKILLSNILDVMDEES
Inhibitor
Name:
BDBM472480
Synonyms:
(R)-N-(1-Acryloylpiperidin-3-yl)-5-(*S)-(6-isopropoxy-4- | US10822348, Example 789
Type:
Small organic molecule
Emp. Form.:
C26H30N6O3S
Mol. Mass.:
506.62
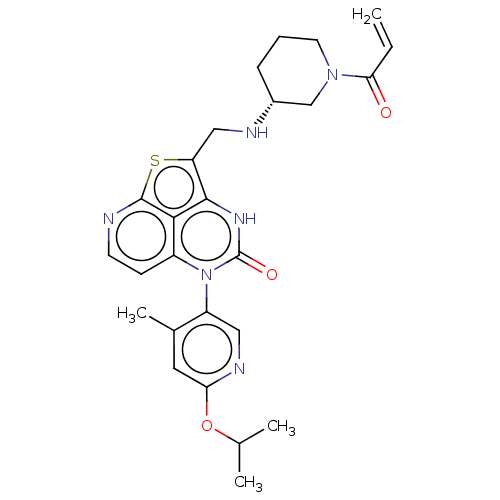
SMILES:
CC(C)Oc1cc(C)c(cn1)-n1c2ccnc3sc(CN[C@@H]4CCCN(C4)C(=O)C=C)c([nH]c1=O)c23 |r,wD:21.21,(-10.57,5.78,;-9.24,5.01,;-9.24,3.47,;-7.9,5.78,;-6.57,5.02,;-5.23,5.79,;-3.9,5.02,;-2.57,5.78,;-3.91,3.48,;-5.23,2.7,;-6.57,3.48,;-2.58,2.7,;-2.58,1.16,;-3.91,.39,;-3.91,-1.15,;-2.58,-1.92,;-1.24,-1.15,;.24,-1.55,;1.33,-.46,;2.87,-.46,;3.64,-1.79,;5.18,-1.79,;5.95,-.46,;7.49,-.46,;8.26,-1.79,;7.49,-3.12,;5.95,-3.12,;8.26,-4.46,;7.49,-5.79,;9.8,-4.46,;10.57,-5.79,;.09,1.16,;.09,2.7,;-1.24,3.47,;-1.24,5.01,;-1.24,.39,)|