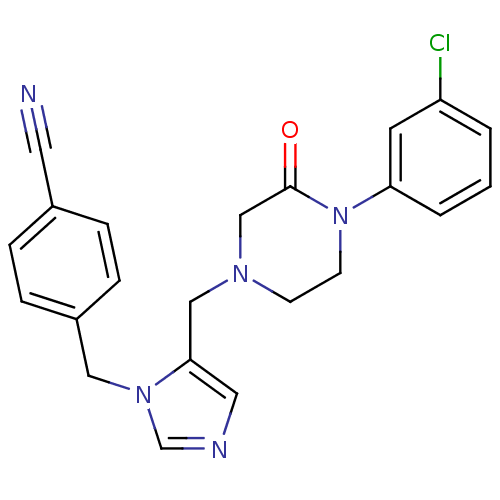
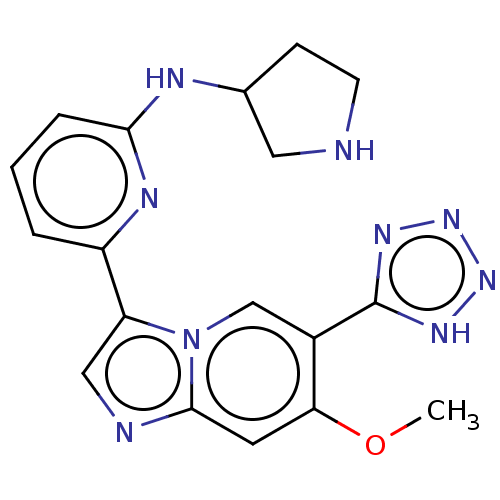
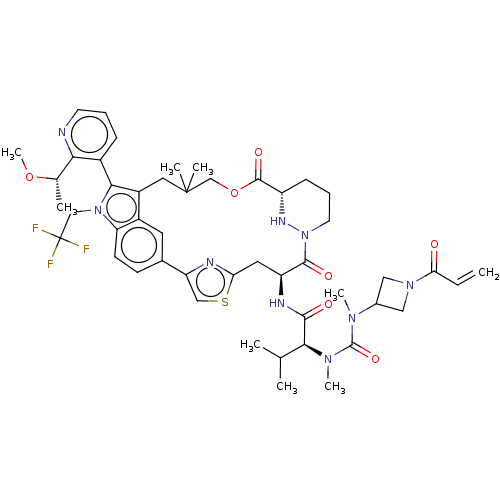
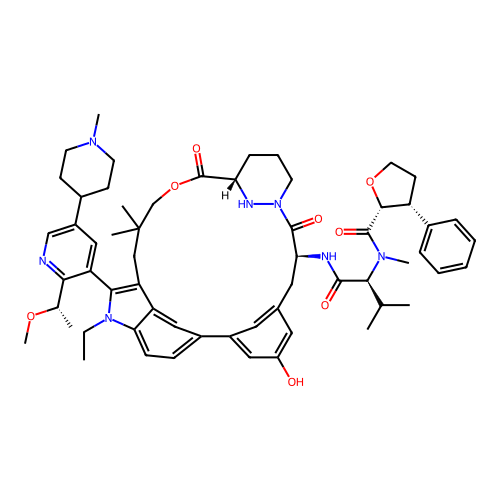
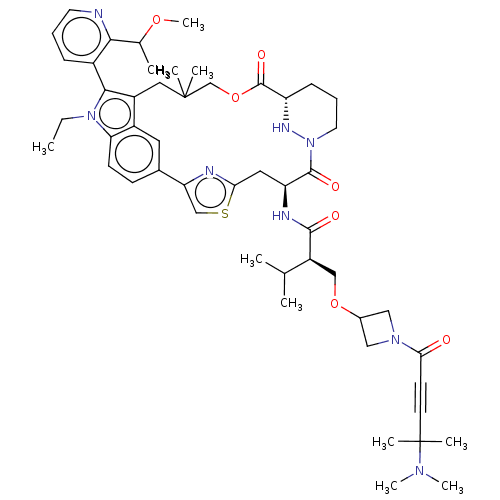
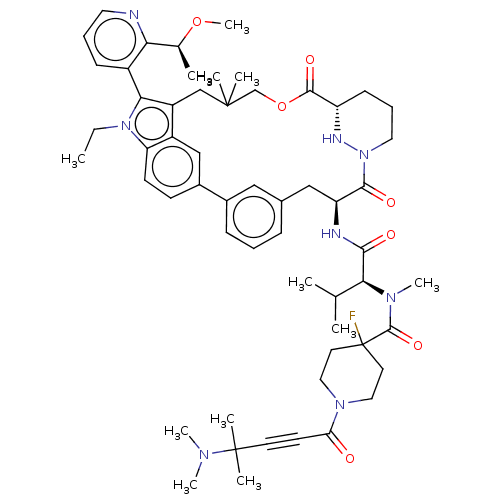
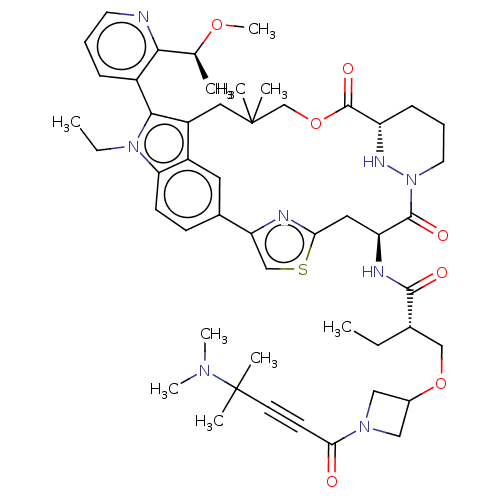
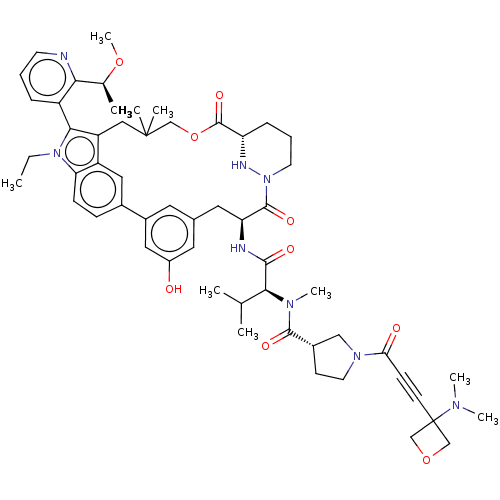
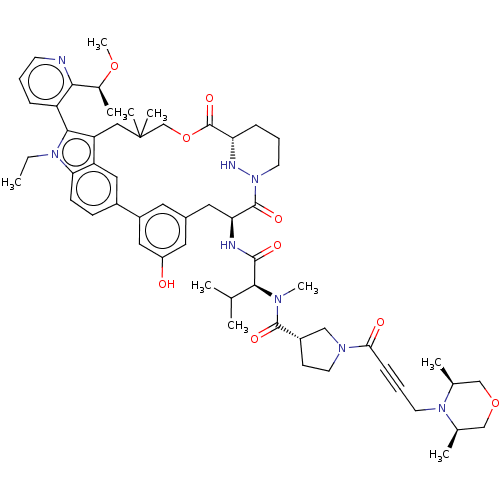
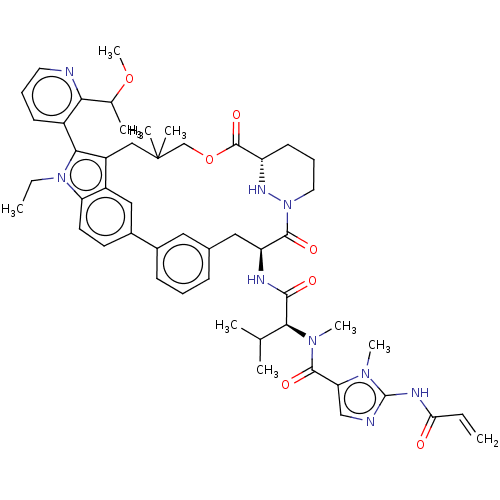
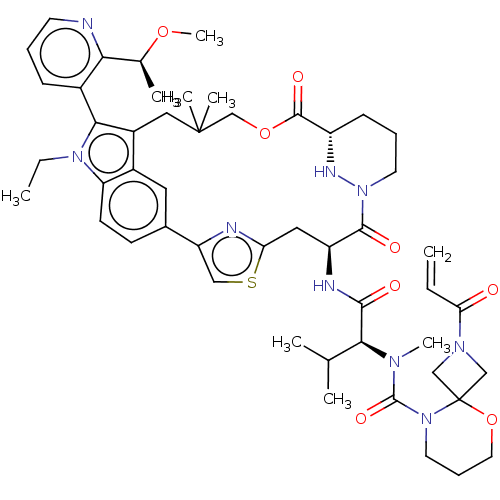
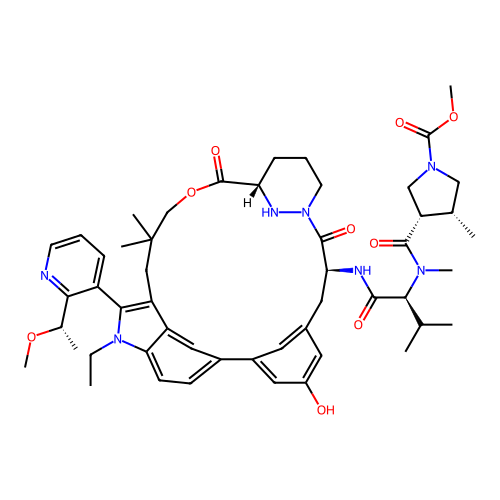
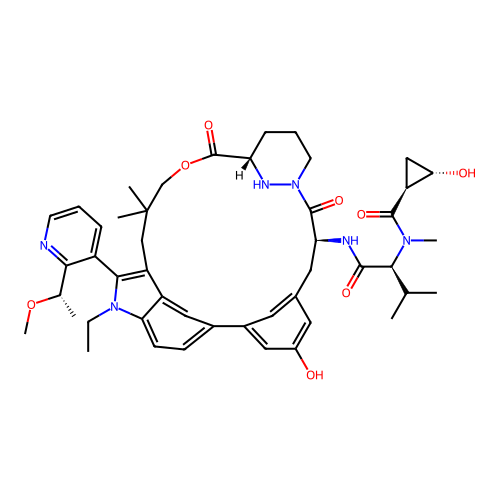
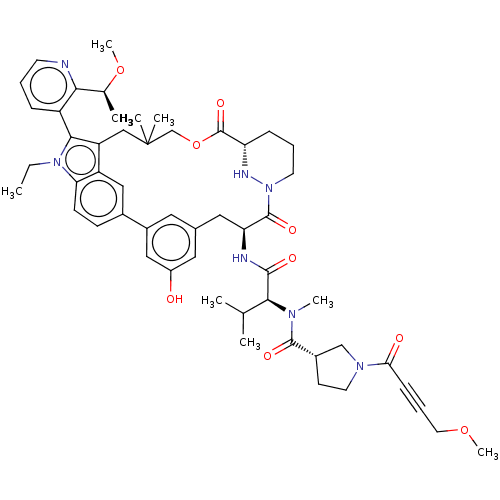
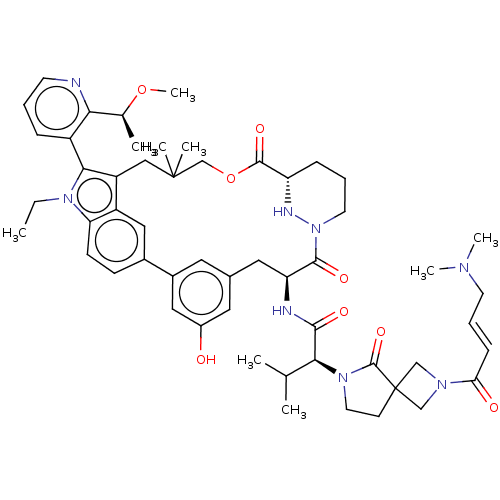
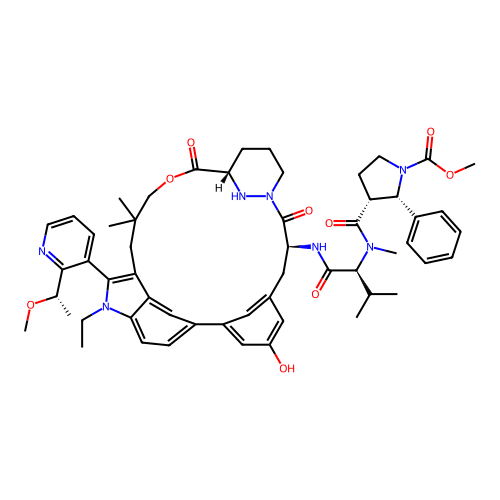
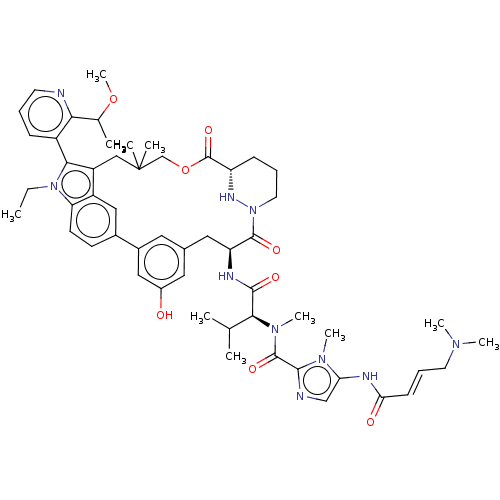
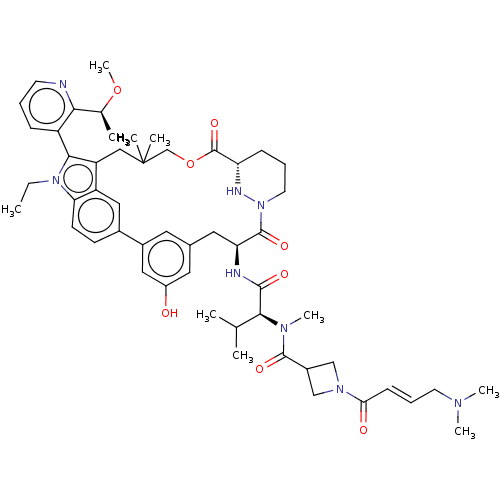
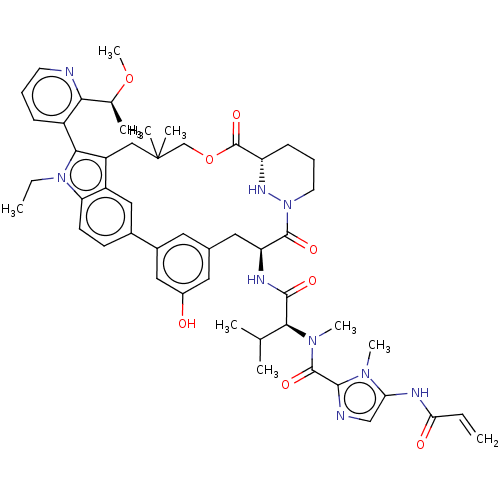
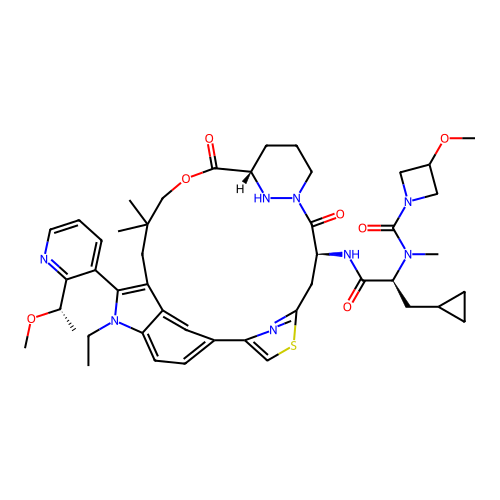
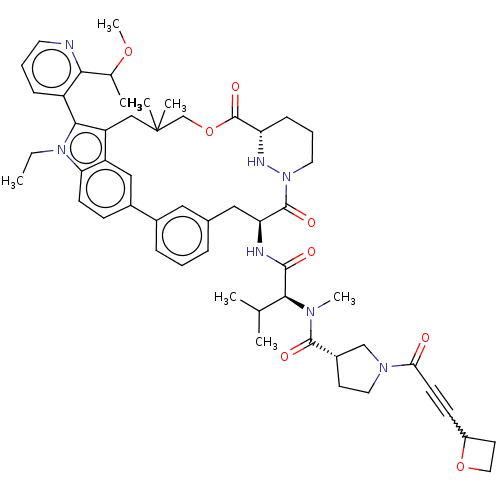
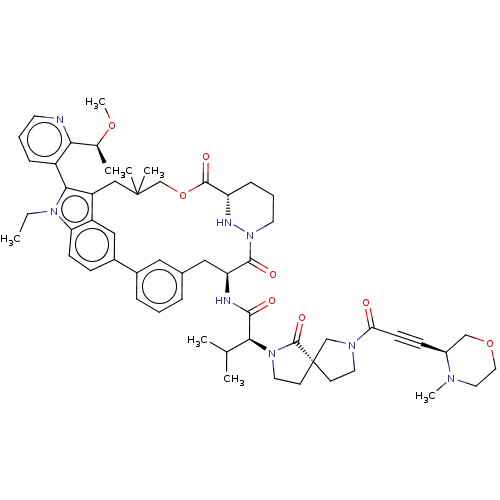
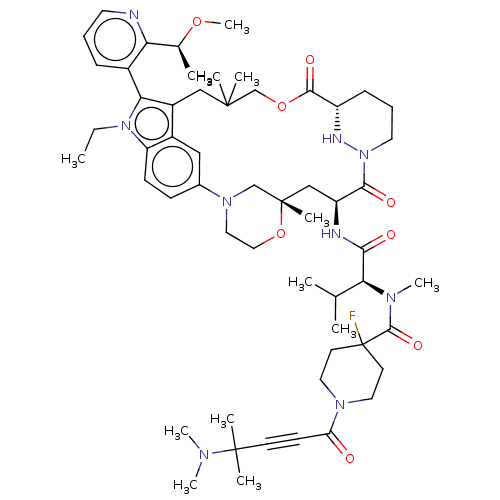
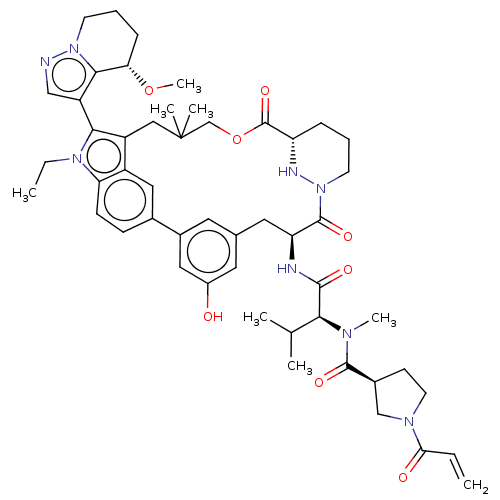
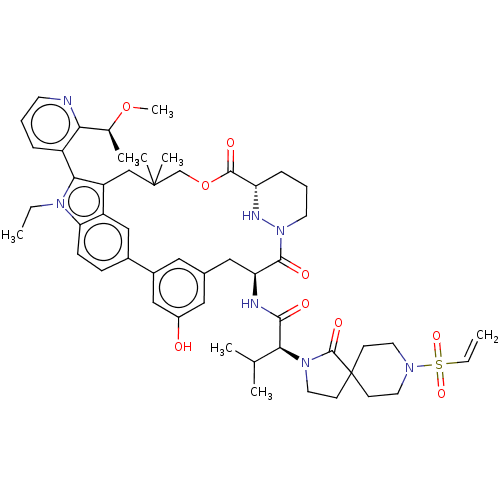
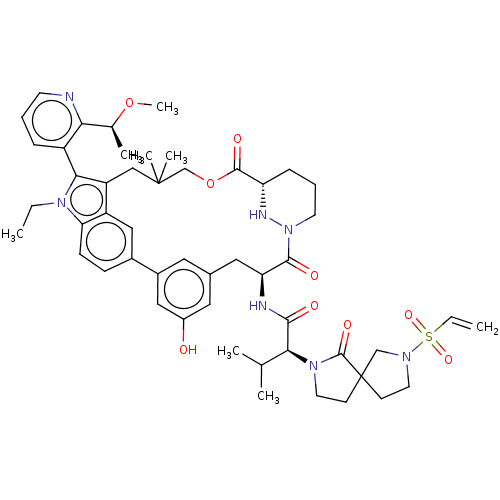
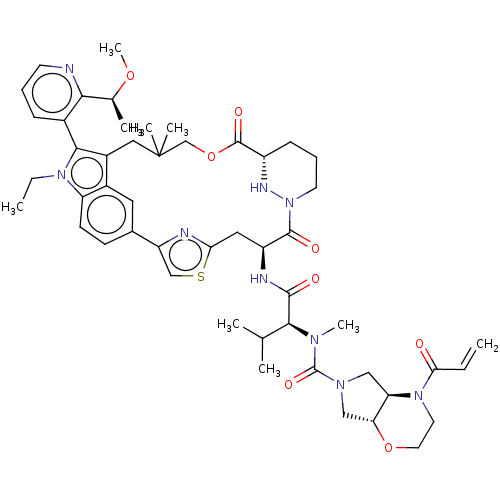
TargetGTPase NRas(Human)
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Affinity DataKi: 0.300nMAssay Description:Binding affinity to NRAS (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
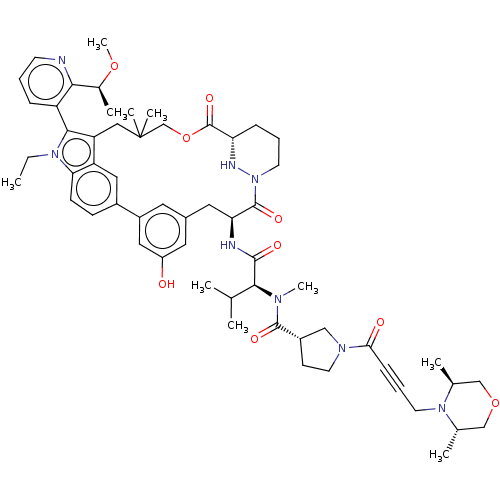
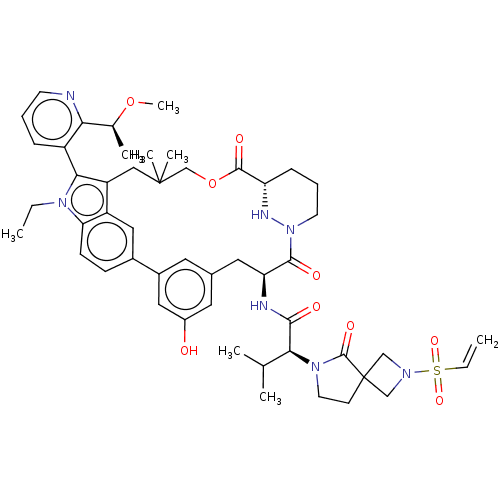
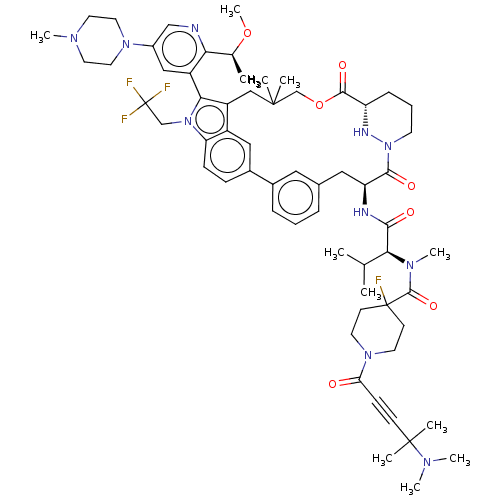
TargetGTPase NRas(Human)
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:AlphaScreen SureFire STAT5 (pTyr694;Tyr699) Assay kit (Perkin Elmer). Assay was performed according to manufacturer protocol.More data for this Ligand-Target Pair
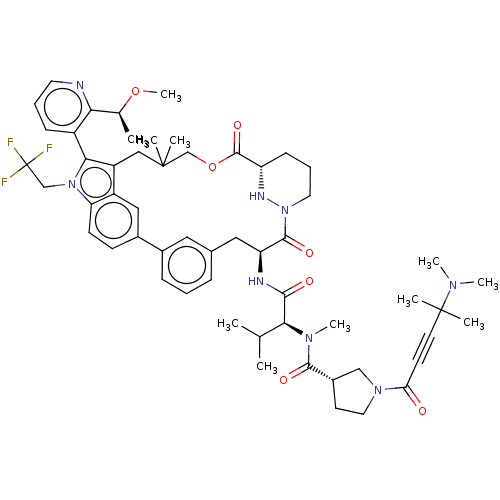
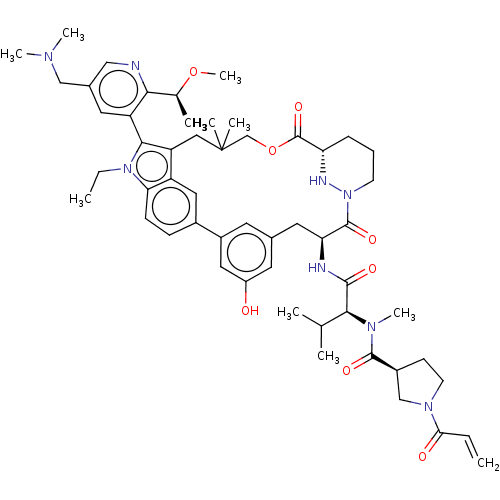
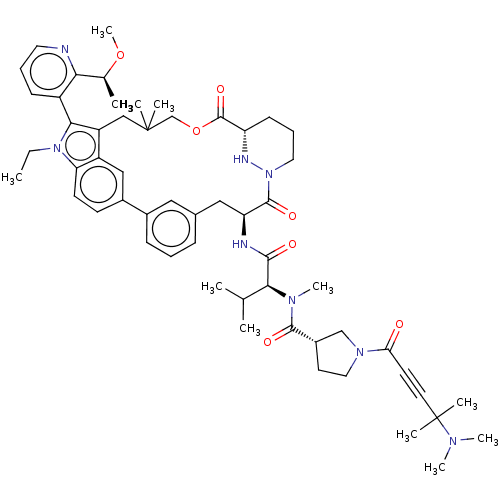
TargetGTPase NRas(Human)
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Table 8-10: Compounds were tested in 10-dose IC50 mode with 3-fold serial dilution starting at 10 uM, and are relative to DMSO, the negative control....More data for this Ligand-Target Pair
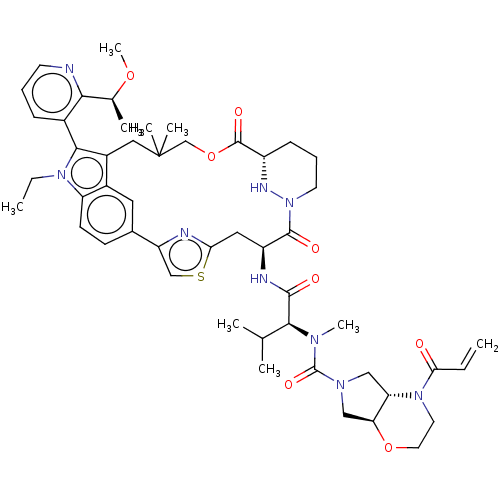
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
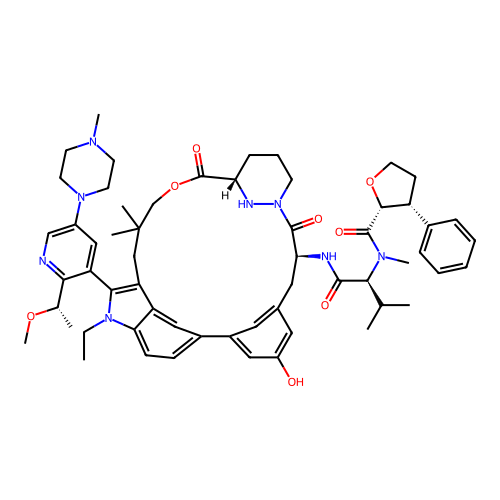
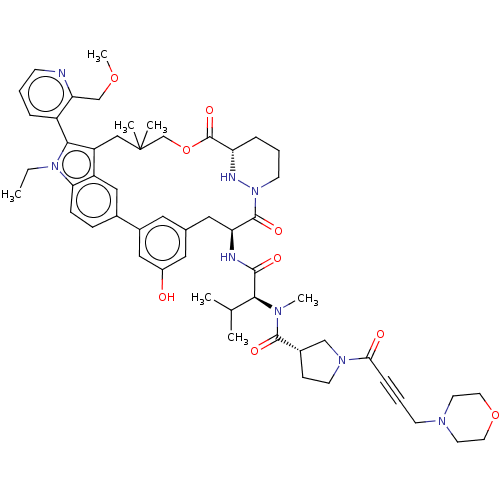
TargetGTPase NRas(Human)
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Massachusetts College of Pharmacy and Health Sciences University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:A variety of Ras proteins may be inhibited by compounds of the present invention (e.g., K-Ras, N-Ras, H-Ras, and mutants thereof at positions 12, 13 ...More data for this Ligand-Target Pair