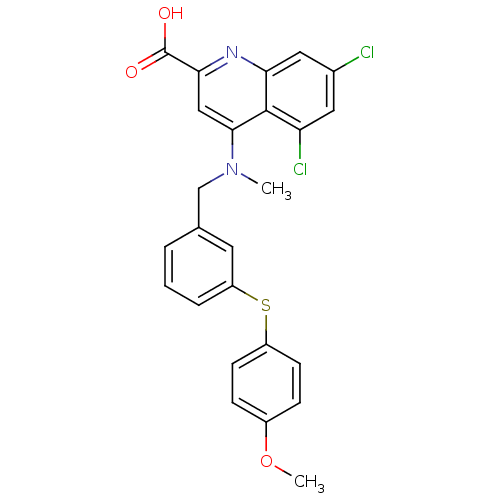
BDBM50343530 5,7-dichloro-4-((3-(4-methoxyphenylthio)benzyl)(methyl)amino)quinoline-2-carboxylic acid::CHEMBL1774309
SMILES COc1ccc(Sc2cccc(CN(C)c3cc(nc4cc(Cl)cc(Cl)c34)C(O)=O)c2)cc1
InChI Key InChIKey=FUUIYLWURGZBJB-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50343530
Found 13 hits for monomerid = 50343530
TargetTyrosine-protein phosphatase non-receptor type 22(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataKi: 5.53E+3nMAssay Description:Competitive inhibition of N-terminal His-tagged Lyp (unknown origin) catalytic domain (1 to 294 residues) expressed in Escherichia coli BL21 (DE3) as...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 5(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: 6.06E+4nMAssay Description:Inhibition of STEP (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 9(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: 6.61E+4nMAssay Description:Inhibition of MEG2 (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: 3.88E+4nMAssay Description:Inhibition of PTP1B (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
TargetDual specificity protein phosphatase 3(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: 5.07E+4nMAssay Description:Inhibition of VHR (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
TargetProtein phosphatase Slingshot homolog 2(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of SSH2 (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of human microsomal PGES1 in cell-free system assessed as inhibition of conversion of PGH2 to PGE2 by HPLC assayMore data for this Ligand-Target Pair
Affinity DataIC50: 7.48E+4nMAssay Description:Inhibition of PPM1G (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.80E+3nMAssay Description:Inhibition of KDM5A (unknown origin) using substrate peptide/ascorbate/2-OG/ Fe(2) as substrate preincubated for 15 mins followed by addition of subs...More data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
University Of Innsbruck
Curated by ChEMBL
University Of Innsbruck
Curated by ChEMBL
Affinity DataIC50: 5.20E+3nMAssay Description:Inhibition of human recombinant 5-LO in cell-free system assessed as inhibition of LTB4 production after 10 mins by RP-HPLC assayMore data for this Ligand-Target Pair
TargetPolyunsaturated fatty acid 5-lipoxygenase(Homo sapiens (Human))
University Of Innsbruck
Curated by ChEMBL
University Of Innsbruck
Curated by ChEMBL
Affinity DataIC50: 850nMAssay Description:Inhibition of 5-LO in human polymorphonuclear leukocytes assessed as inhibition of LTB4 production after 10 mins by RP-HPLC assayMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 18(Homo sapiens (Human))
Shandong University
Curated by ChEMBL
Shandong University
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of PTPN18 (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of PPM1A (unknown origin) assessed as reduction in pNPP hydrolysisMore data for this Ligand-Target Pair