Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Carbonic anhydrase 9
Ligand
BDBM11639
Substrate
n/a
Meas. Tech.
ChEMBL_878780 (CHEMBL2183720)
Ki
16±n/a nM
Citation
 Marini, AM; Maresca, A; Aggarwal, M; Orlandini, E; Nencetti, S; Da Settimo, F; Salerno, S; Simorini, F; La Motta, C; Taliani, S; Nuti, E; Scozzafava, A; McKenna, R; Rossello, A; Supuran, CT Tricyclic sulfonamides incorporating benzothiopyrano[4,3-c]pyrazole and pyridothiopyrano[4,3-c]pyrazole effectively inhibita- andß-carbonic anhydrase: X-ray crystallography and solution investigations on 15 isoforms. J Med Chem 55:9619-29 (2012) [PubMed] Article
Marini, AM; Maresca, A; Aggarwal, M; Orlandini, E; Nencetti, S; Da Settimo, F; Salerno, S; Simorini, F; La Motta, C; Taliani, S; Nuti, E; Scozzafava, A; McKenna, R; Rossello, A; Supuran, CT Tricyclic sulfonamides incorporating benzothiopyrano[4,3-c]pyrazole and pyridothiopyrano[4,3-c]pyrazole effectively inhibita- andß-carbonic anhydrase: X-ray crystallography and solution investigations on 15 isoforms. J Med Chem 55:9619-29 (2012) [PubMed] Article More Info.:
Target
Name:
Carbonic anhydrase 9
Synonyms:
CA-IX | CA9 | CAH9_HUMAN | Carbonate dehydratase IX | Carbonic anhydrase 9 (CA IX) | Carbonic anhydrase 9 (CAIX) | Carbonic anhydrase 9 precursor | Carbonic anhydrase IX (CA IX) | Carbonic anhydrase IX (CAIX) | Carbonic anhydrases IX | Carbonic anhydrases; II & IX | G250 | MN | Membrane antigen MN | RCC-associated antigen G250
Type:
Enzyme
Mol. Mass.:
49669.03
Organism:
Homo sapiens (Human)
Description:
Catalytic domain of human cloned isozyme was used in the assay
Residue:
459
Sequence:
MAPLCPSPWLPLLIPAPAPGLTVQLLLSLLLLVPVHPQRLPRMQEDSPLGGGSSGEDDPLGEEDLPSEEDSPREEDPPGEEDLPGEEDLPGEEDLPEVKPKSEEEGSLKLEDLPTVEAPGDPQEPQNNAHRDKEGDDQSHWRYGGDPPWPRVSPACAGRFQSPVDIRPQLAAFCPALRPLELLGFQLPPLPELRLRNNGHSVQLTLPPGLEMALGPGREYRALQLHLHWGAAGRPGSEHTVEGHRFPAEIHVVHLSTAFARVDEALGRPGGLAVLAAFLEEGPEENSAYEQLLSRLEEIAEEGSETQVPGLDISALLPSDFSRYFQYEGSLTTPPCAQGVIWTVFNQTVMLSAKQLHTLSDTLWGPGDSRLQLNFRATQPLNGRVIEASFPAGVDSSPRAAEPVQLNSCLAAGDILALVFGLLFAVTSVAFLVQMRRQHRRGTKGGVSYRPAEVAETGA
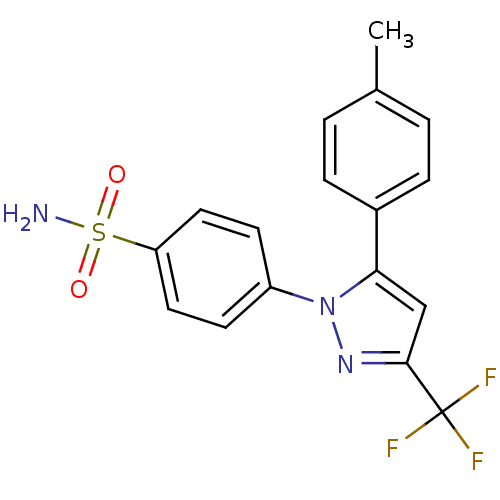
Inhibitor
Name:
BDBM11639
Synonyms:
4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene-1-sulfonamide | CHEMBL118 | CLX | Celebrex | Celecoxib | US10322118, Urea-Based Scaffold Entry 9 | US11478464, Compound Celecoxib | US11786535, Compound Celecoxib | US8741944, Comparative Compound | US9388139, Celecoxib | cid_2662
Type:
Small organic molecule
Emp. Form.:
C17H14F3N3O2S
Mol. Mass.:
381.372
SMILES:
Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F