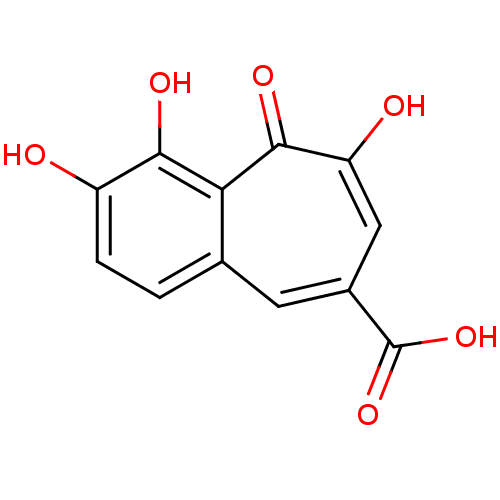
BDBM33413 benzo[7]annulene-8-carboxylic acid, 5
SMILES OC(=O)c1cc(O)c(=O)c2c(O)c(O)ccc2c1
InChI Key InChIKey=XCSLJQFPTAZKDV-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 33413
Found 8 hits for monomerid = 33413
Affinity DataEC50: 54nMAssay Description:Agonist activity at GPR35 receptor in human HT-29 cells after 10 mins by dynamic mass redistribution assayMore data for this Ligand-Target Pair
Affinity DataIC50: 97nMAssay Description:Inhibition of recombinant human SHP2 expressed in Escherichia coli using 2P-IRS1 peptide as substrate after 1 hr by microplate reader analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:Desensitization of GPR35 receptor in human HT-29 cells assessed as inhibition of zaprinast-induced dynamic mass redistribution after 10 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of SHP2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of wild type recombinant Atg4B (unknown origin) expressed in Escherichia coli BL21 DE3 using N-terminal His6-tagged LC3B-PLA2 as substrate...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of recombinant ATG4B (unknown origin) expressed in Escherichia coli BL21 using LC3B-PLA2 as substrate incubated for 30 to 60 mins by High-...More data for this Ligand-Target Pair
Affinity DataEC50: 1.24E+4nMAssay Description:Agonist activity at GPR35 receptor in human U2OS cells coexpressing Gal4-VP16-TEV assessed as beta arrestin translocation after 5 hrs by beta lactama...More data for this Ligand-Target Pair
Affinity DataIC50: 2.32E+4nMpH: 8.0 T: 2°CAssay Description:Enzyme activity was measuring substrate hydrolysis of RNase H. The intact substrate has a low background fluorescent signal and provides up to 50-fol...More data for this Ligand-Target Pair