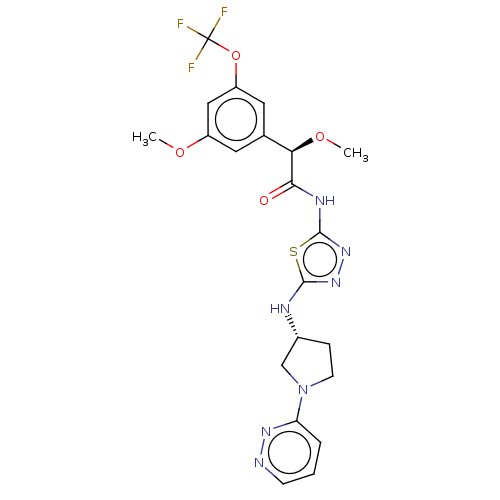
BDBM416667 (2R)-2-Methoxy-2-[3-methoxy-5-(trifluoromethoxy)phenyl]-N-[5-[[(3R)-1-pyridazin-3-ylpyrrolidin-3-yl]amino]-1,3,4-thiadiazol-2-yl]acetamide and (2R)-2-methoxy-2-[3-methoxy-5-(trifluoromethoxy)phenyl]-N-[5-[[(3R)-1-pyridazin-3-ylpyrrolidin-3-yl]amino]-1,3,4-thiadiazol-2-yl]acetamide::US10323028, Example 11(b)::US10981904, Example 11(a)
SMILES CO[C@@H](C(=O)Nc1nnc(N[C@@H]2CCN(C2)c2cccnn2)s1)c1cc(OC)cc(OC(F)(F)F)c1
InChI Key InChIKey=ZFCXIZUETUNJKI-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 416667
Found 3 hits for monomerid = 416667
Affinity DataIC50: 6nMAssay Description:Inhibition of human KGA (63 to 669 residues) preincubated for 15 mins using 50 mM glutamine as substrate by resorufin dye based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.11nMAssay Description:A Glutamate Oxidase/AmplexRed coupled assay was used to measure the ability of compounds to bind to and inhibit the activity of GLS1 in vitro. 6His t...More data for this Ligand-Target Pair
Affinity DataIC50: 6.35nMAssay Description:A Glutamate Oxidase/AmplexRed coupled assay was used to measure the ability of compounds to bind to and inhibit the activity of GLS1 in vitro. 6His t...More data for this Ligand-Target Pair