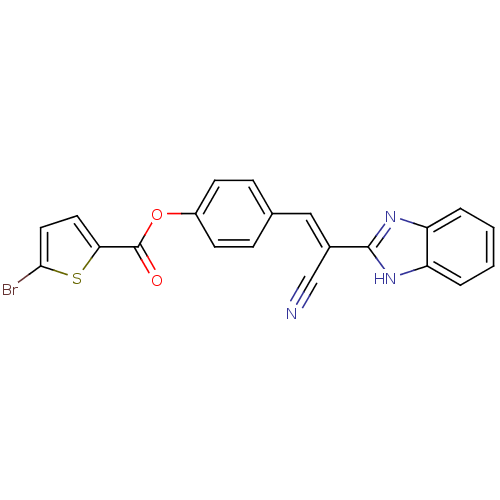
BDBM48750 5-bromo-2-thiophenecarboxylic acid [4-[(E)-2-(1H-benzimidazol-2-yl)-2-cyanoethenyl]phenyl] ester::5-bromothiophene-2-carboxylic acid [4-[(E)-2-(1H-benzimidazol-2-yl)-2-cyano-vinyl]phenyl] ester::MLS000374996::SMR000254350::[4-[(E)-2-(1H-benzimidazol-2-yl)-2-cyano-ethenyl]phenyl] 5-bromanylthiophene-2-carboxylate::[4-[(E)-2-(1H-benzimidazol-2-yl)-2-cyanoethenyl]phenyl] 5-bromothiophene-2-carboxylate::cid_6141417
SMILES Brc1ccc(s1)C(=O)Oc1ccc(\C=C(/C#N)c2nc3ccccc3[nH]2)cc1
InChI Key InChIKey=NBBLYDLYKQYOPP-SDNWHVSQSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 48750
Found 2 hits for monomerid = 48750
TargetCarboxy-terminal domain RNA polymerase II polypeptide A small phosphatase 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.78E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetEukaryotic translation initiation factor 4 gamma 1(Homo sapiens (Human))
Emory University
Curated by PubChem BioAssay
Emory University
Curated by PubChem BioAssay
Affinity DataIC50: 6.40E+4nMAssay Description:Dose Response Confirmation for Small Molecule Inhibitors of Eukaryotic Translation Initiation NIH Molecular Libraries Screening Centers Network [MLSC...More data for this Ligand-Target Pair