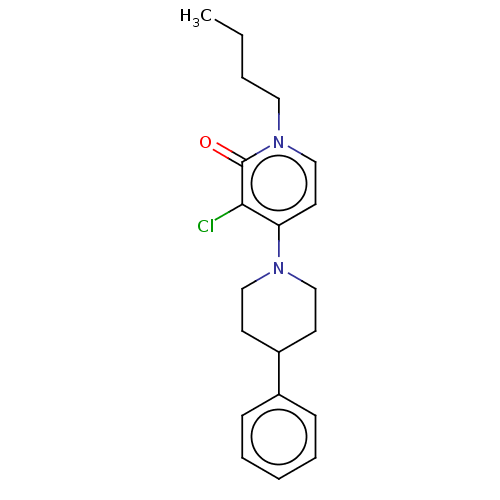
BDBM50051408 CHEMBL3337527::US11071729, Compound 2
SMILES CCCCn1ccc(N2CCC(CC2)c2ccccc2)c(Cl)c1=O
InChI Key InChIKey=HYOGJHCDLQSAHX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50051408
Found 6 hits for monomerid = 50051408
Affinity DataKi: 180nMAssay Description:Displacement of [3H]8-Trifluoromethyl-3-cyclopropylmethyl-7-[(4-phenyl-1-piperidinyl)methyl]-1,2,4-triazolo[4,3-a]pyridine from mGlu2 receptor (unkno...More data for this Ligand-Target Pair
Affinity DataEC50: 147nMAssay Description:Positive allosteric modulation of human mGlu2 receptor expressed in CHO cells assessed as potentiation of glutamate-induced effect incubated for 30 m...More data for this Ligand-Target Pair
Affinity DataEC50: 182nMAssay Description:Measurement of mGluR2 positive allosteric modulatory activity of test compounds in membranes containing human mGluR2 was performed using frozen membr...More data for this Ligand-Target Pair
Affinity DataEC50: 65nMAssay Description:Positive allosteric modulation of wild-type human mGlu2 receptor expressed in CHO-K1 cells assessed as potentiation of glutamate-induced effect incub...More data for this Ligand-Target Pair
Affinity DataEC50: 147nMAssay Description:Positive allosteric modulation of human mGlu2 receptor expressed in CHO cell membranes assessed as potentiation of glutamate-induced effect incubated...More data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Displacement of [3H]19 from human mGlu2 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)