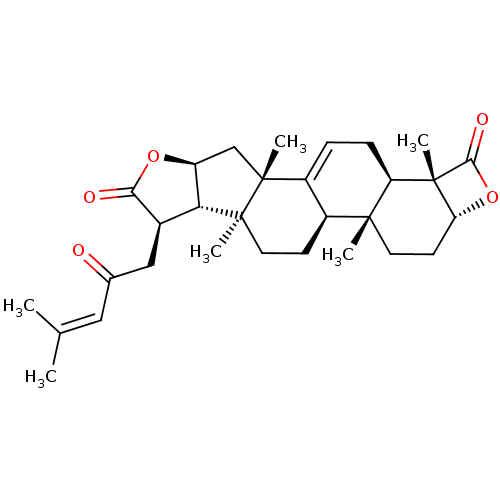
BDBM50072284 (1S,2R,4aR,6aS,6bS,7R,9aS,10aS)-2-Acetoxy-1,4a,6a,10a-tetramethyl-7-(4-methyl-2-oxo-pent-3-enyl)-8-oxo-2,3,4,4a,4b,5,6,6a,6b,7,8,9a,10,10a,12,12a-hexadecahydro-1H-9-oxa-pentaleno[2,1-a]phenanthrene-1-carboxylic acid::CHEMBL457155::GR-133686
SMILES [H][C@@]12[#6]-[#6][C@]3([#6])[C@@]4([H])[#6]-[#6][C@@]5([#6])[C@]6([H])[#6@@H](-[#6]-[#6](=O)\[#6]=[#6](\[#6])-[#6])-[#6](=O)-[#8][C@@]6([H])[#6][C@]5([#6])[#6]4=[#6]-[#6][C@@]3([H])[C@]1([#6])[#6](=O)-[#8]2
InChI Key InChIKey=UAKRLLAUOQEYFN-PQNCNOJFSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50072284
Found 5 hits for monomerid = 50072284
Affinity DataIC50: 70nMAssay Description:Compound was evaluated for the inhibition of ChymotrypsinogenMore data for this Ligand-Target Pair
TargetCoagulation factor X(Homo sapiens (Human))
Glaxo Wellcome Research And Development
Curated by ChEMBL
Glaxo Wellcome Research And Development
Curated by ChEMBL
Affinity DataIC50: >2.00E+4nMAssay Description:Inhibition of thrombinMore data for this Ligand-Target Pair
TargetNeutrophil elastase(Homo sapiens (Human))
Glaxo Wellcome Research And Development
Curated by ChEMBL
Glaxo Wellcome Research And Development
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Compound was evaluated for the inhibition of human leukocyte elastase(HLE).More data for this Ligand-Target Pair
TargetSerine protease 1(Homo sapiens (Human))
Glaxo Wellcome Research And Development
Curated by ChEMBL
Glaxo Wellcome Research And Development
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:Compound was evaluated for the inhibition of Coagulation factor XMore data for this Ligand-Target Pair