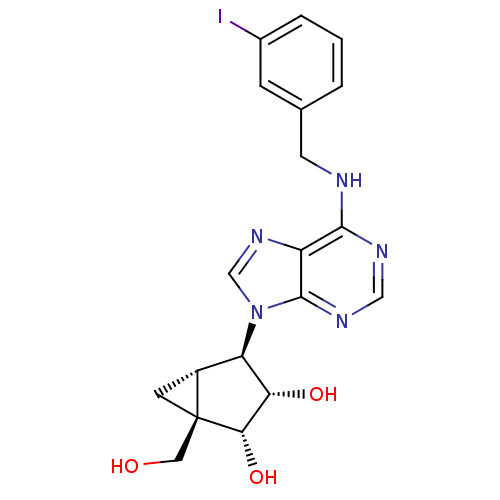
BDBM50088425 (1R,2R,3S,4R,5S)-1-Hydroxymethyl-4-[6-(3-iodo-benzylamino)-purin-9-yl]-bicyclo[3.1.0]hexane-2,3-diol::1-Hydroxymethyl-4-[6-(3-iodo-benzylamino)-purin-9-yl]-bicyclo[3.1.0]hexane-2,3-diol::1-hydroxymethyl-4-[6-(3-iodobenzylamino)-9H-9-purinyl]-(1R,2R,3S,4R,5S)-bicyclo[3.1.0]hexane-2,3-diol::CHEMBL417292
SMILES OC[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(I)c3)ncnc12
InChI Key InChIKey=VIXCVSNTFYSXDO-DPNDMPAOSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50088425
Found 7 hits for monomerid = 50088425
Affinity DataKi: 4.10nMAssay Description:Displacement of [125I]- AB-MECA from human adenosine A3 receptor expressed in HEK cellsMore data for this Ligand-Target Pair
Affinity DataKi: 69nMAssay Description:Displacement of [3H]R-PIA from rat brain membrane Adenosine A1 receptorMore data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Rattus norvegicus (rat))
National Institute Of Diabetes
Curated by ChEMBL
National Institute Of Diabetes
Curated by ChEMBL
Affinity DataKi: 601nMAssay Description:Displacement of specific [3H]-CGS- 21680 binding to adenosine A2A receptor in rat striatal membranesMore data for this Ligand-Target Pair
Affinity DataKi: 1.21E+4nMAssay Description:Displacement of [3H]- ZM-241385 from human adenosine A2B receptor expressed in HEK cellsMore data for this Ligand-Target Pair
Affinity DataEC50: 218nMAssay Description:Stimulation of [35S]GTP-gamma-S, binding to human adenosine A1 receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 940nMAssay Description:Stimulation of [35S]GTP-gamma-S, binding to adenosine A1 receptor of rat cerebral cortical membraneMore data for this Ligand-Target Pair
Affinity DataEC50: 0.700nMAssay Description:Stimulation of [35S]GTP-gamma-S, against human adenosine A3 receptorMore data for this Ligand-Target Pair