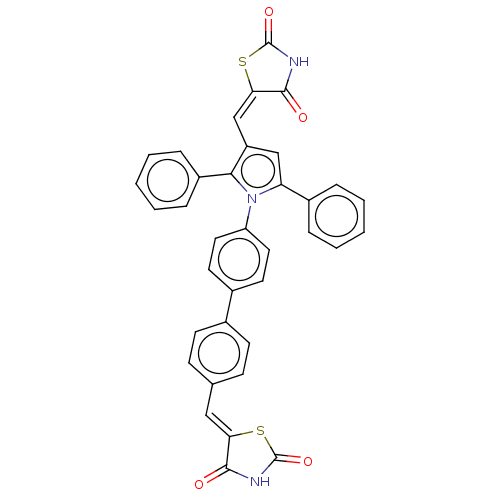
BDBM50238195 CHEMBL4090687
SMILES O=C1NC(=O)\C(S1)=C\c1cc(-c2ccccc2)n(c1-c1ccccc1)-c1ccc(cc1)-c1ccc(\C=C2/SC(=O)NC2=O)cc1
InChI Key InChIKey=REFHBOVGIOGXRN-DSAROVGPSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50238195
Found 3 hits for monomerid = 50238195
TargetGlutaminase kidney isoform, mitochondrial(Homo sapiens (Human))
National Health Research Institutes
Curated by ChEMBL
National Health Research Institutes
Curated by ChEMBL
Affinity DataIC50: 151nMAssay Description:Inhibition of binding of [3H]naloxone to Opioid receptor mu 1 in the rat brain homogenateMore data for this Ligand-Target Pair
TargetGlutaminase kidney isoform, mitochondrial(Homo sapiens (Human))
National Health Research Institutes
Curated by ChEMBL
National Health Research Institutes
Curated by ChEMBL
Affinity DataIC50: 45nMAssay Description:Inhibition of recombinant human His-tagged KGA (L123 to L669 residues) expressed in Escherichia coli BL21(DE3)pLysS using glutamine substrate preincu...More data for this Ligand-Target Pair
TargetGlutaminase liver isoform, mitochondrial(Homo sapiens)
National Health Research Institutes
Curated by ChEMBL
National Health Research Institutes
Curated by ChEMBL
Affinity DataIC50: 261nMAssay Description:Inhibition of recombinant human His-tagged GAB (P56 to V602 residues) expressed in Escherichia coli C41(DE3)pLysS using glutamine as substrate preinc...More data for this Ligand-Target Pair