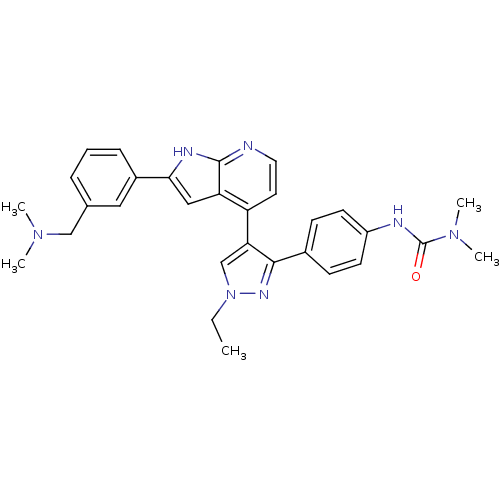
BDBM50315769 3-(4-(4-(2-(3-((dimethylamino)methyl)phenyl)-1H-pyrrolo[2,3-b]pyridin-4-yl)-1-ethyl-1H-pyrazol-3-yl)phenyl)-1,1-dimethylurea::4-[3-(4-N,N-Dimethylcarbamylaminophenyl)-1-ethyl-1H-pyrazol-4-yl]-2-[3-(dimethylaminomethyl)phenyl]-1H-pyrrolo[2,3-b]-pyridine::CHEMBL1090479::GSK-1070916
SMILES CCn1cc(c(n1)-c1ccc(NC(=O)N(C)C)cc1)-c1ccnc2[nH]c(cc12)-c1cccc(CN(C)C)c1
InChI Key InChIKey=QTBWCSQGBMPECM-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 18 hits for monomerid = 50315769
Found 18 hits for monomerid = 50315769
Affinity DataKi: 0.380nMAssay Description:Competitive inhibition of human Aurora B ATP binding site by rapid dilution methodMore data for this Ligand-Target Pair
Affinity DataKi: 0.380nMAssay Description:Competitive inhibition of Aurora B ATP binding siteMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Competitive inhibition of human Aurora C ATP binding siteMore data for this Ligand-Target Pair
Affinity DataKi: 490nMAssay Description:Competitive inhibition of human Aurora A ATP binding siteMore data for this Ligand-Target Pair
Affinity DataIC50: >3.30E+4nMAssay Description:Inhibition of CYP1A2 in human liver microsomes using phenacetin as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.80E+4nMAssay Description:Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 8.10E+4nMAssay Description:Inhibition of CYP2C19 in human liver microsomes using S-mephenytoin as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP2D6 in human liver microsomes using bufuralol as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using nifedipine as substrateMore data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 1(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
TargetVascular endothelial growth factor receptor 3(Homo sapiens (Human))
Glaxosmithkline
Curated by ChEMBL
Glaxosmithkline
Curated by ChEMBL
Affinity DataIC50: 1.26E+3nMAssay Description:Inhibition of human Aurora AMore data for this Ligand-Target Pair
Affinity DataIC50: 6.5nMAssay Description:Competitive inhibition of Aurora C ATP binding siteMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human Aurora B by rapid dilution methodMore data for this Ligand-Target Pair