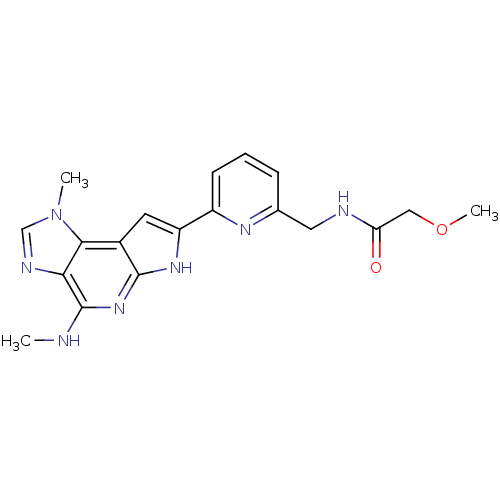
BDBM50358591 CHEMBL1923983
SMILES CNc1nc2[nH]c(cc2c2n(C)cnc12)-c1cccc(CNC(=O)COC)n1
InChI Key InChIKey=ULTCRVJUAZCGPP-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50358591
Found 13 hits for monomerid = 50358591
TargetVascular endothelial growth factor receptor 3(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
TargetEphrin type-A receptor 1(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
TargetInhibitor of nuclear factor kappa-B kinase subunit alpha(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 4.96E+3nMAssay Description:Inhibition of IKK1More data for this Ligand-Target Pair
TargetInhibitor of nuclear factor kappa-B kinase subunit beta(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of human IKK2-mediated GST-IkappaBalpha protein fusion phosphorylationMore data for this Ligand-Target Pair
TargetCytochrome P450 1A2(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of CYP1A2More data for this Ligand-Target Pair
TargetCytochrome P450 2D6(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 1.49E+3nMAssay Description:Inhibition of KDRMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
TargetCytochrome P450 2C9(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of CYP2C9More data for this Ligand-Target Pair
TargetProtein-tyrosine kinase 6(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
TargetProto-oncogene tyrosine-protein kinase receptor Ret(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: 1.11E+3nMAssay Description:Inhibition of EphB1More data for this Ligand-Target Pair
TargetCytochrome P450 2C19(Homo sapiens (Human))
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Bristol-Myers Squibb Research And Development
Curated by ChEMBL
Affinity DataIC50: >4.00E+4nMAssay Description:Inhibition of CYP2C19More data for this Ligand-Target Pair