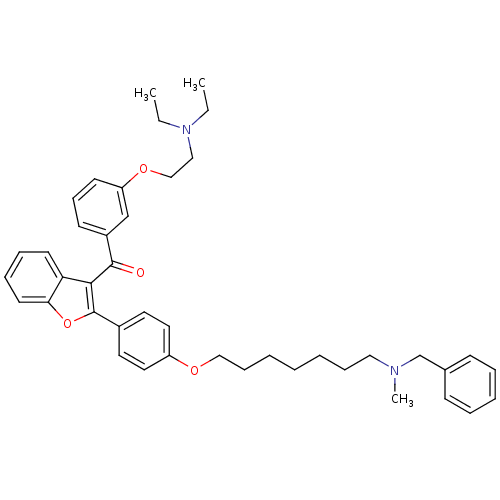
BDBM50394568 CHEMBL2160223
SMILES CCN(CC)CCOc1cccc(c1)C(=O)c1c(oc2ccccc12)-c1ccc(OCCCCCCCN(C)Cc2ccccc2)cc1
InChI Key InChIKey=VKYASPBNOSOSCP-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50394568
Found 6 hits for monomerid = 50394568
Affinity DataKi: 690nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK293 cells after 90 minsMore data for this Ligand-Target Pair
Affinity DataKi: 2.57E+3nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells after 90 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.71E+4nMAssay Description:Inhibition of human recombinant AChE using acetylthiocholine iodide as substrate preincubated for 20 mins prior to substrate addition by Ellman's met...More data for this Ligand-Target Pair
Affinity DataEC50: 2.71E+3nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB2 receptor expressed in HEK293 cells after 90 minsMore data for this Ligand-Target Pair
Affinity DataEC50: 6.38E+3nMAssay Description:Displacement of [3H]CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells after 90 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.81E+3nMAssay Description:Inhibition of human serum BuChE using butyrylthiocholine iodide as substrate preincubated for 20 mins prior to substrate addition by Ellman's methodMore data for this Ligand-Target Pair