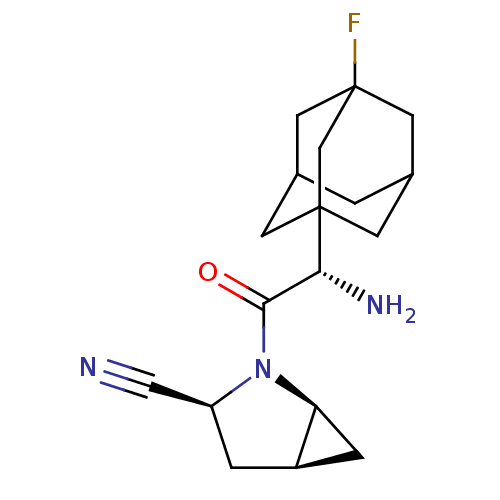
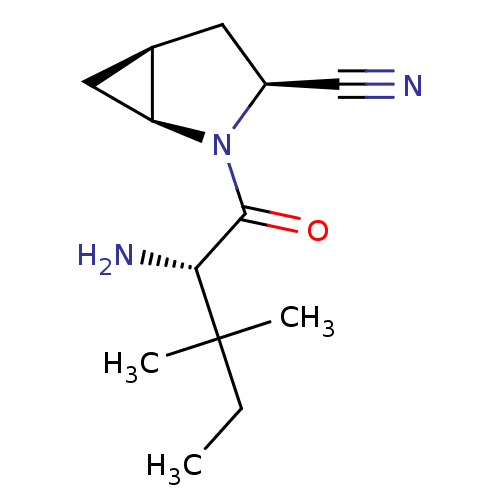
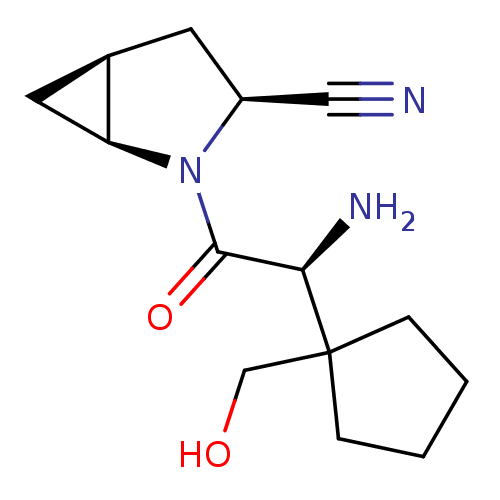
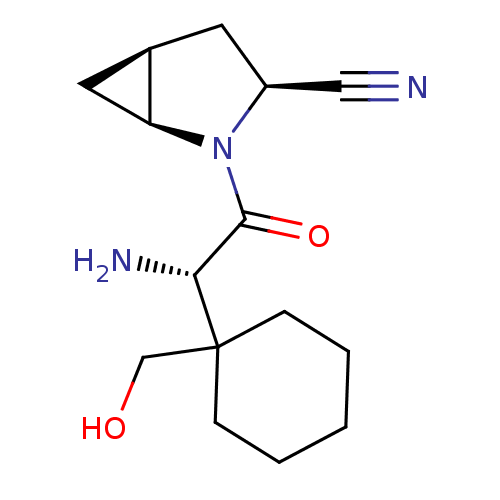
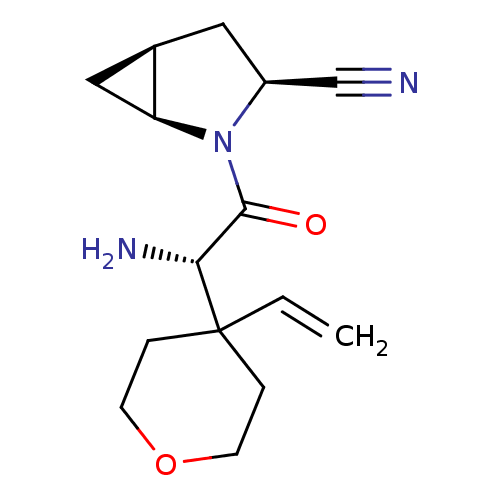
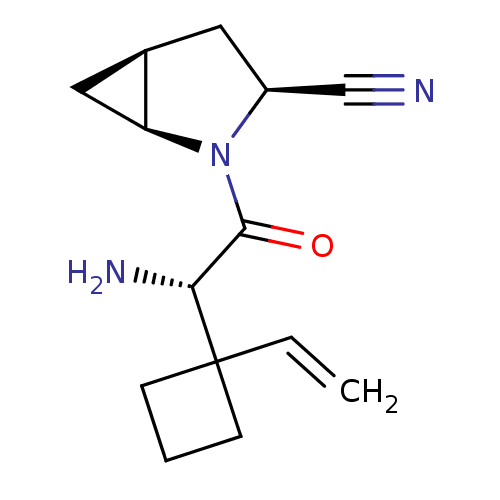
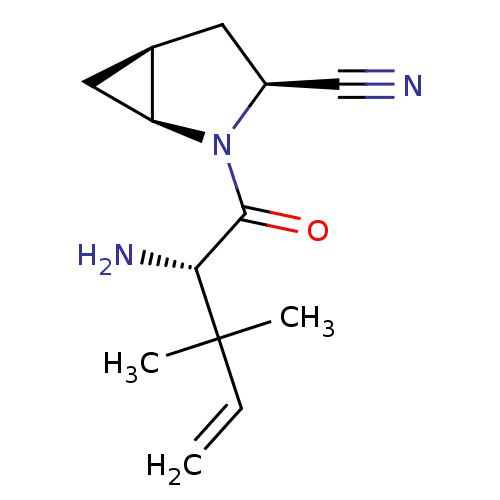
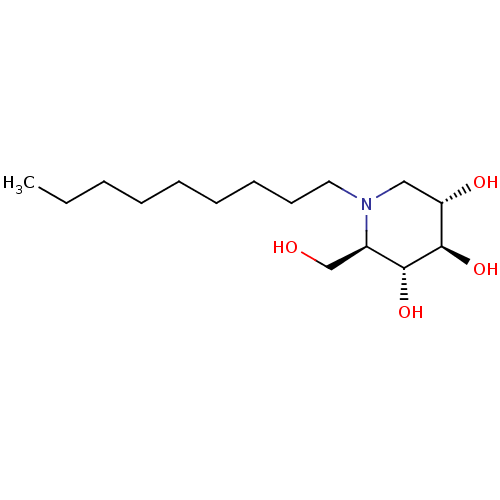
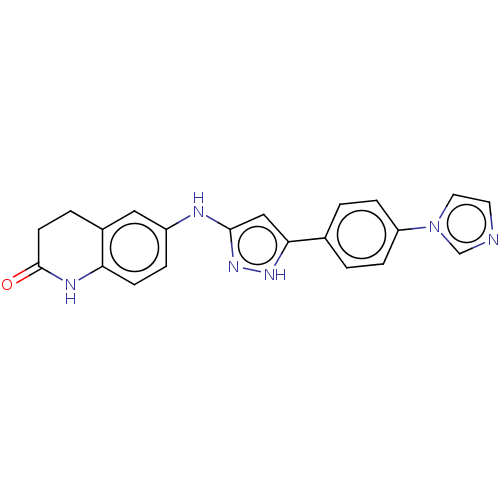
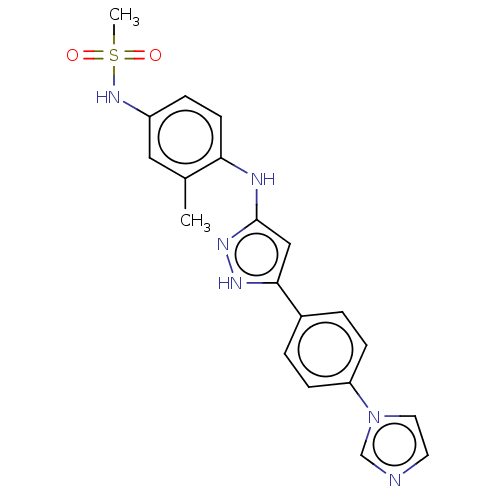
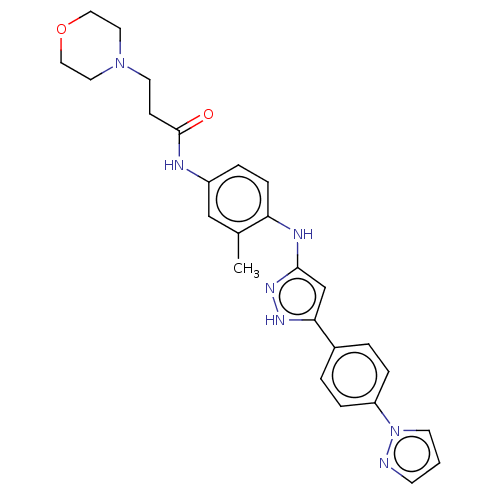
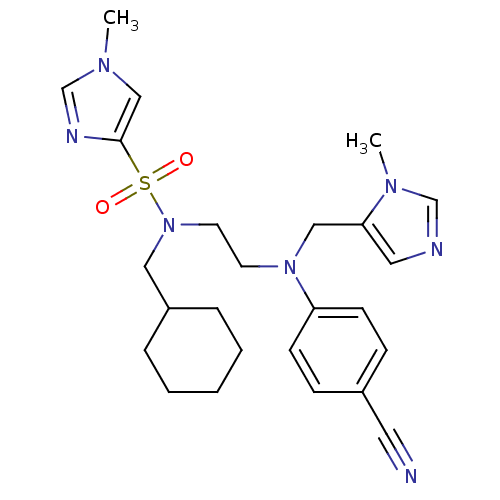
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 0.600nM ΔG°: -52.1kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
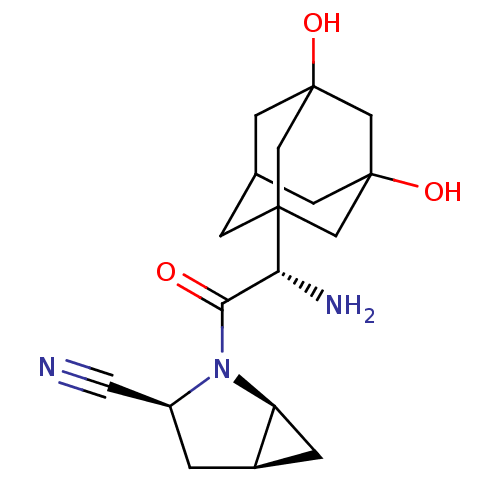
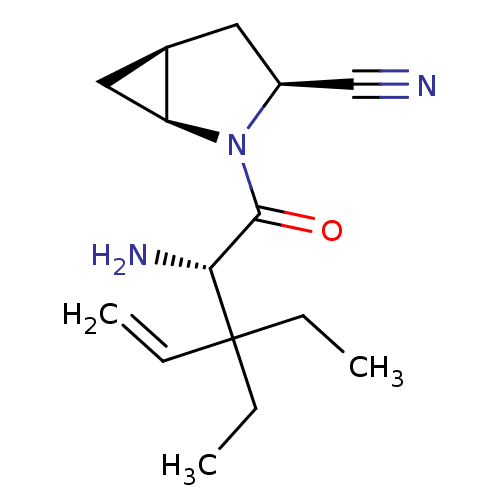
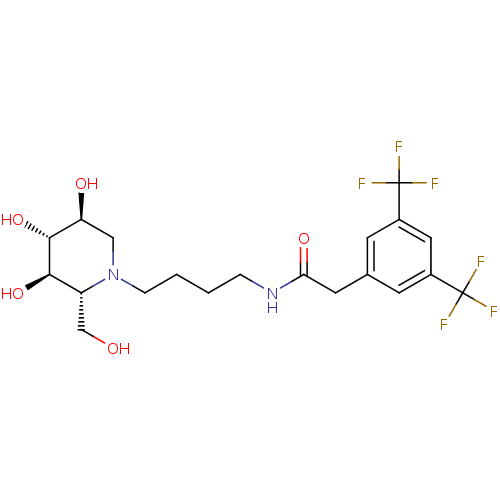
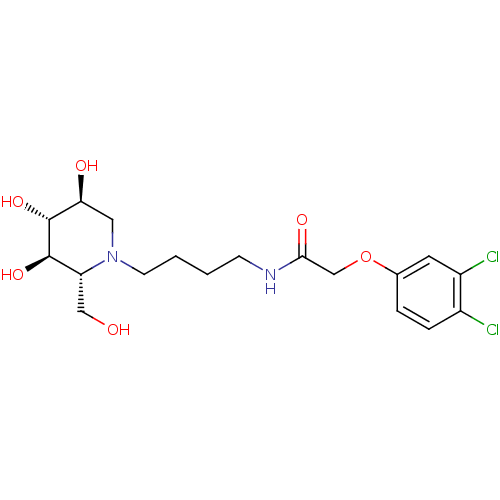
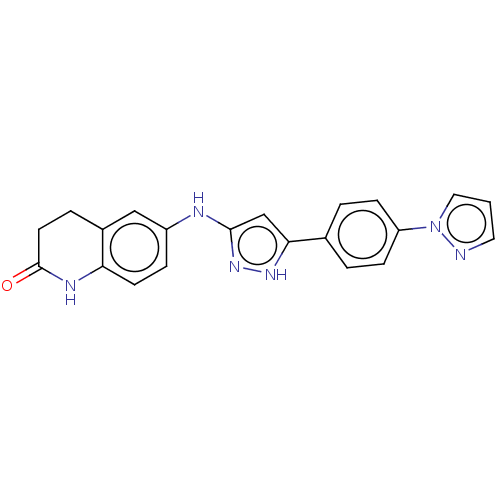
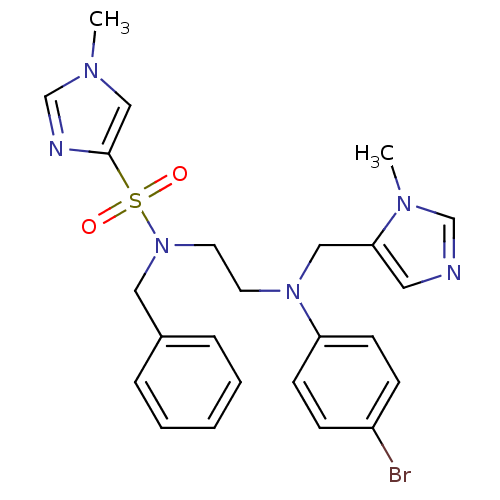
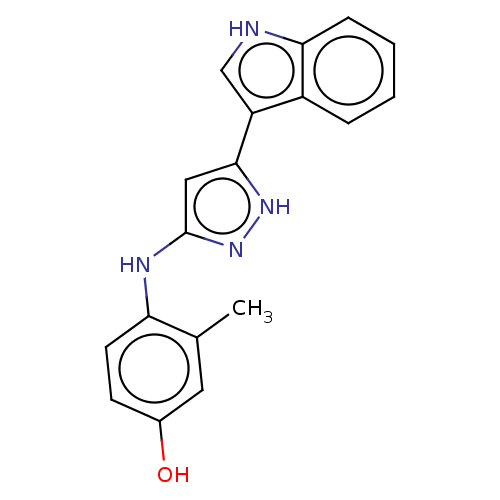
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 0.900nM ΔG°: -51.1kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
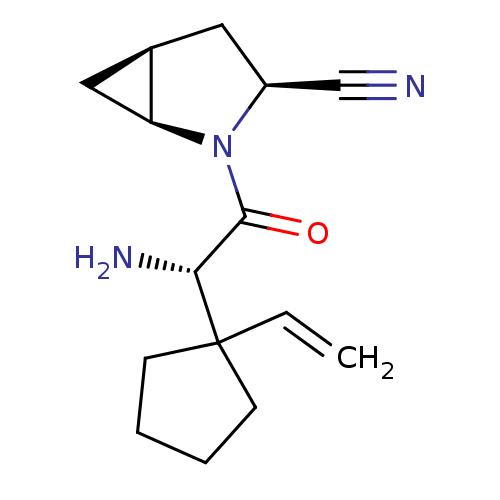
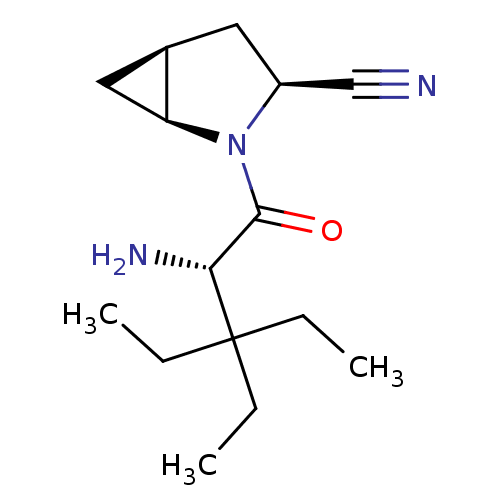
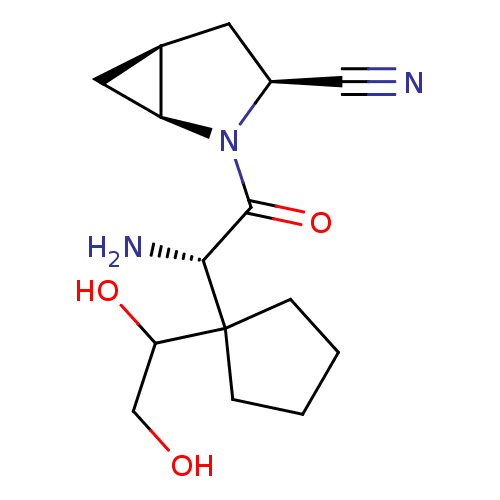
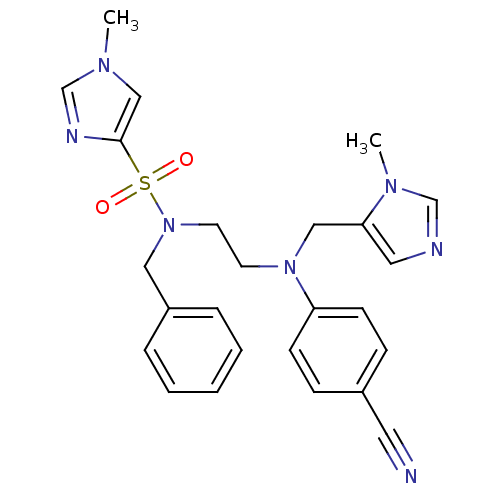
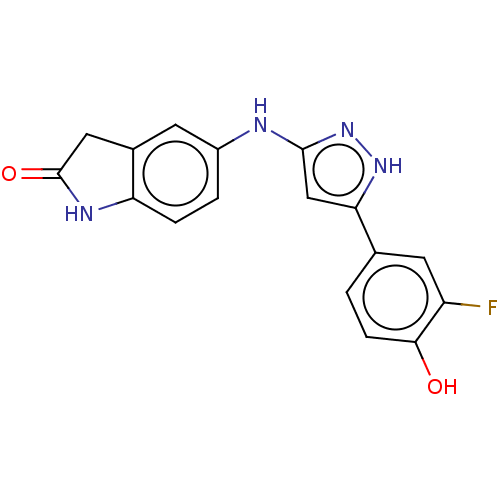
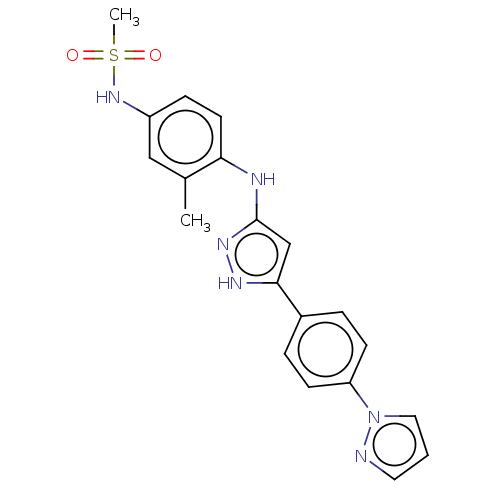
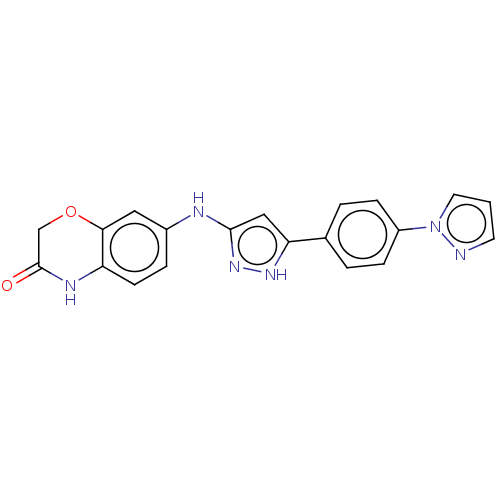
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 1.40nM ΔG°: -50.0kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
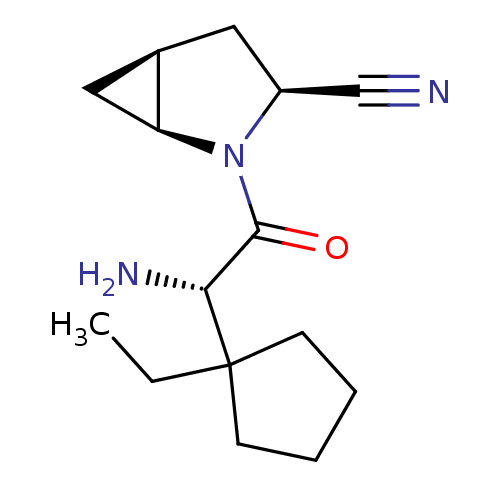
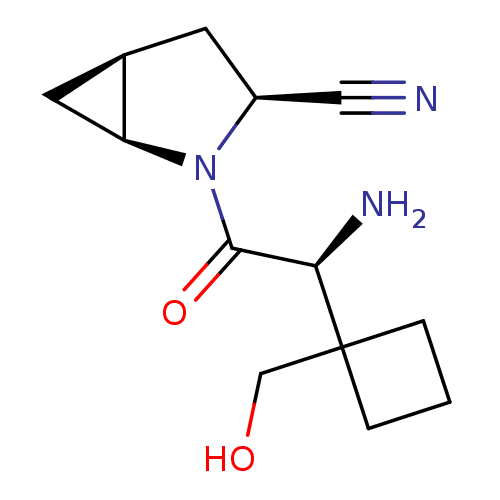
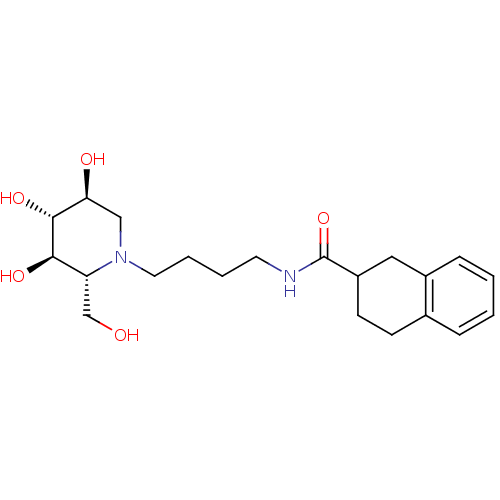
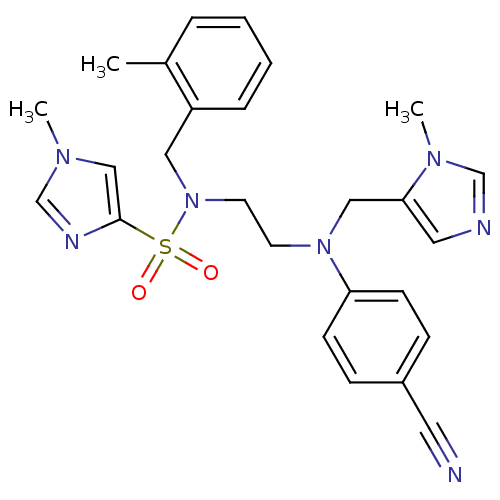
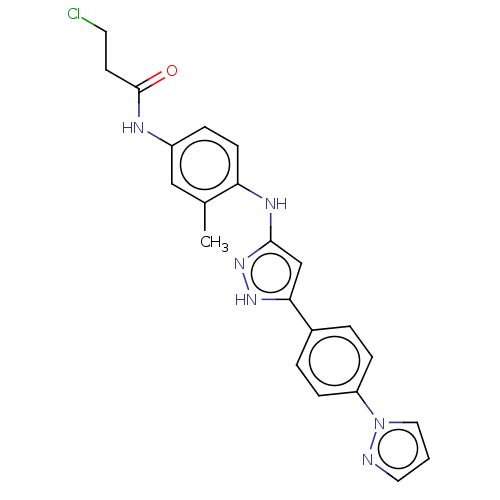
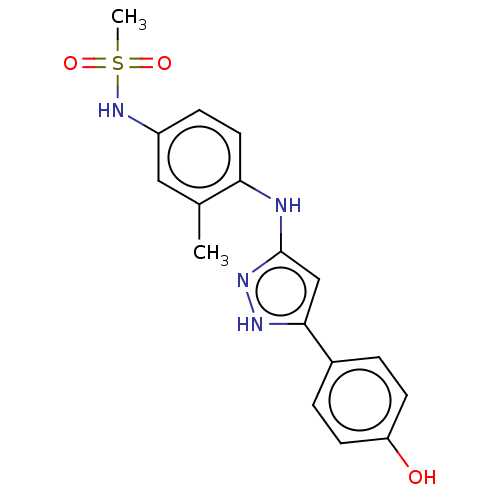
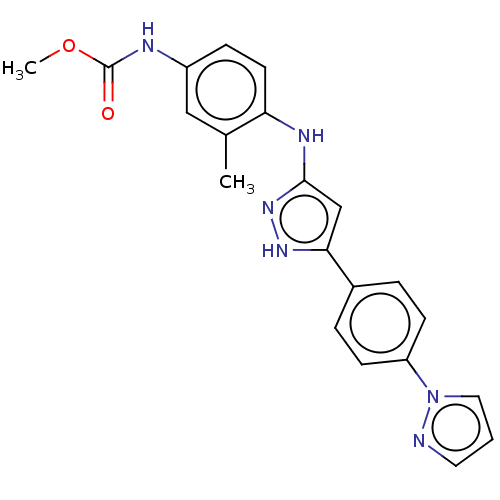
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 1.80nM ΔG°: -49.4kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 2.10nM ΔG°: -49.0kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 3.90nM ΔG°: -47.5kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 5.5nM ΔG°: -46.7kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 7.10nM ΔG°: -46.0kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 7.40nM ΔG°: -45.9kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 8nM ΔG°: -45.7kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 10nM ΔG°: -45.2kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 10nM ΔG°: -45.2kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 12nM ΔG°: -44.8kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 21nM ΔG°: -43.4kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 25nM ΔG°: -43.0kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 31nM ΔG°: -42.4kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 42nM ΔG°: -41.7kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 57nM ΔG°: -40.9kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
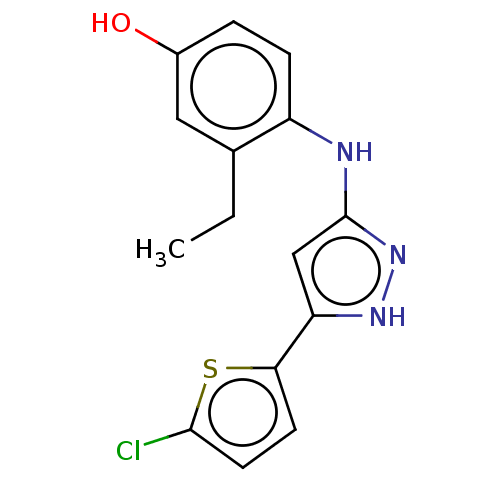
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 71nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetDipeptidyl peptidase 4(Homo sapiens (Human))
Bristol-Myers Squibb Pharmaceutical Research Institute
Bristol-Myers Squibb Pharmaceutical Research Institute
Affinity DataKi: 143nM ΔG°: -38.7kJ/molepH: 7.4 T: 2°CAssay Description:Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea...More data for this Ligand-Target Pair
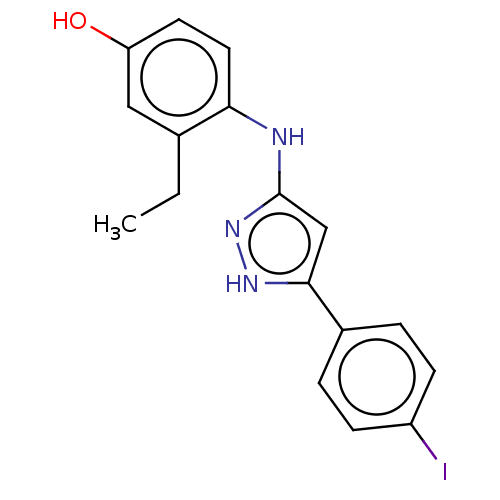
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 400nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 400nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 1.10E+3nMAssay Description:Non-competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 1.30E+3nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 3.20E+3nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetLysosomal acid glucosylceramidase(Homo sapiens (Human))
Genomics Research Center
Curated by ChEMBL
Genomics Research Center
Curated by ChEMBL
Affinity DataKi: 4.10E+3nMAssay Description:Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot methodMore data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 0.5nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 0.600nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
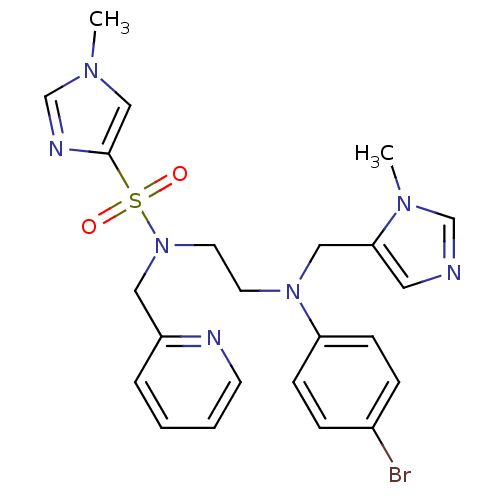
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11447469-20220920-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11447469-20220920-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11447469-20220920-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 1nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 1.90nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 2nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 2nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 2.10nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 3nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 3nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 4nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetATP-dependent RNA helicase RhlE(Plasmodium falciparum (malaria parasite P. falcipa...)
Yale University
Yale University
Affinity DataIC50: 4.5nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 6nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 6nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 7nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11447469-20220920-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 7nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetTRAF2 and NCK-interacting protein kinase(Homo sapiens (Human))
Korea Research Institute of Chemical Technology
US Patent
Korea Research Institute of Chemical Technology
US Patent
Affinity DataIC50: 7nMAssay Description:The inhibitory properties of compounds were evaluated with TNIK kinase enzyme system and luminescent ADP-GloFigure US11485711-20221101-P00001 Kinase ...More data for this Ligand-Target Pair
TargetProtein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha(Rattus norvegicus (rat))
Yale University
Yale University
Affinity DataIC50: 7.5nMpH: 7.5 T: 2°CAssay Description:Assays for PFT activity were performed with a PFT-specific scintillation assay (SPA) kit (Amersham Biosciences, Piscataway, NJ). IC50 values were cal...More data for this Ligand-Target Pair


 3D Structure (crystal)
3D Structure (crystal)