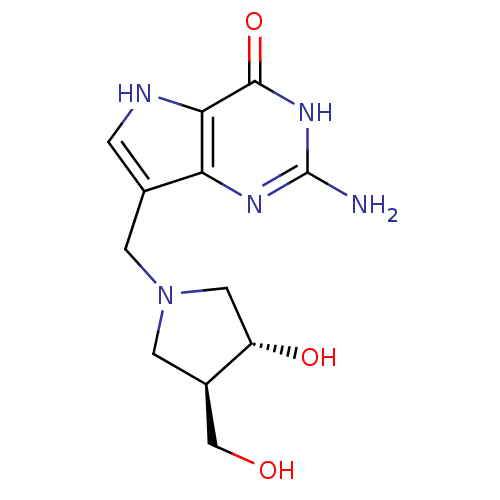
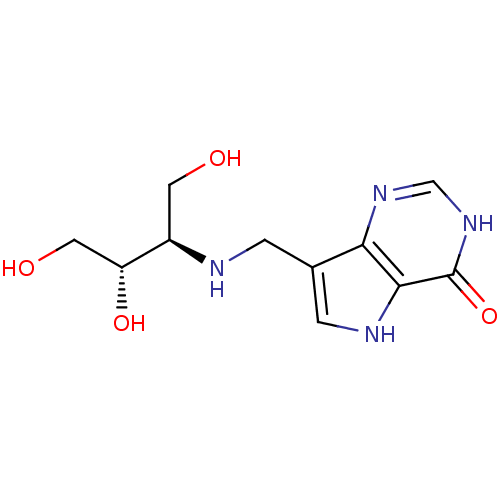
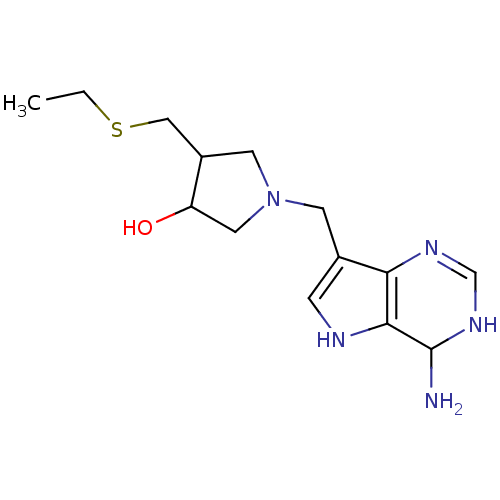
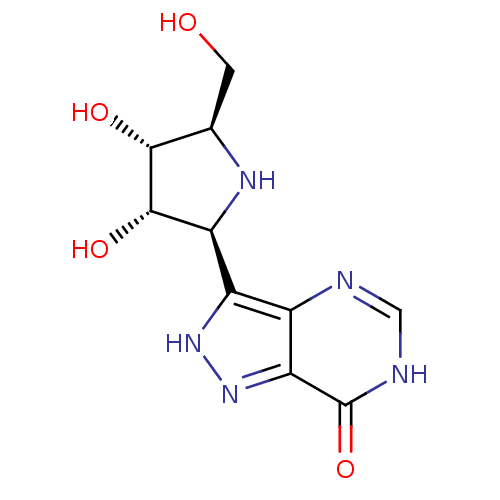
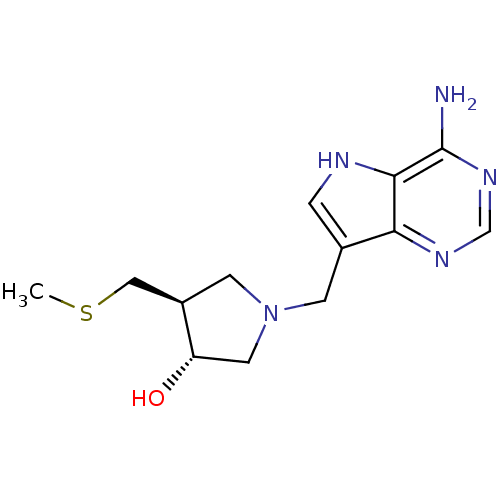
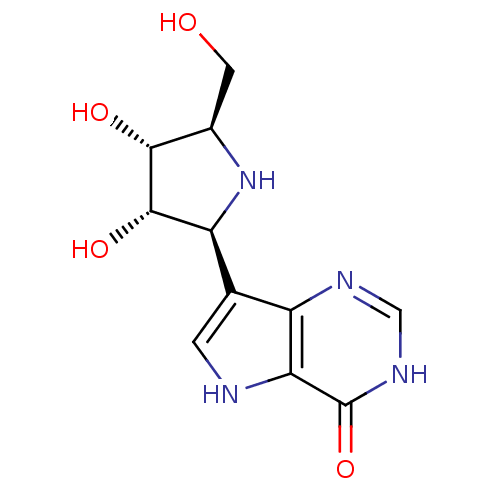
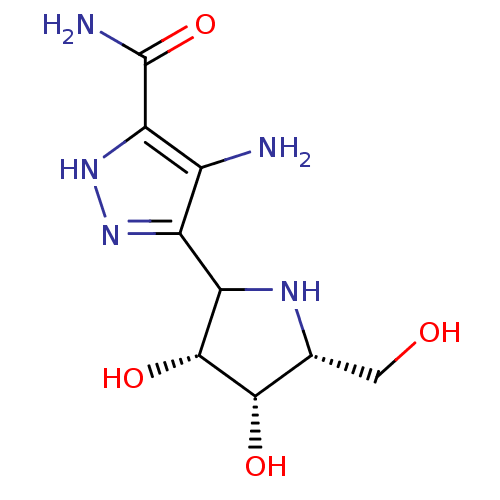
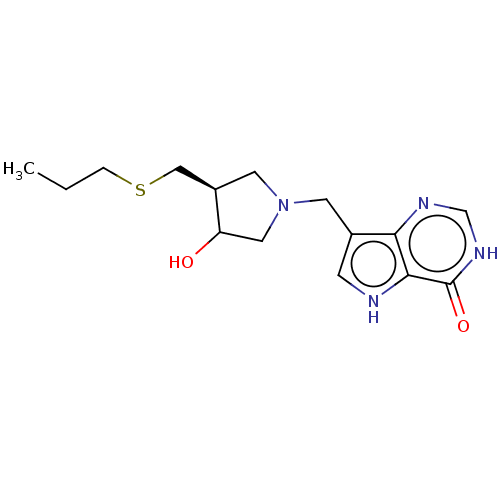
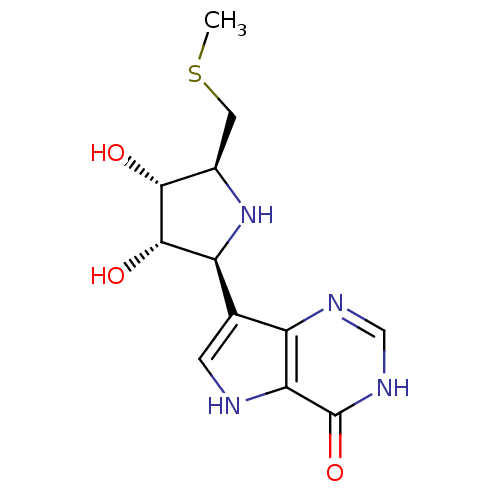
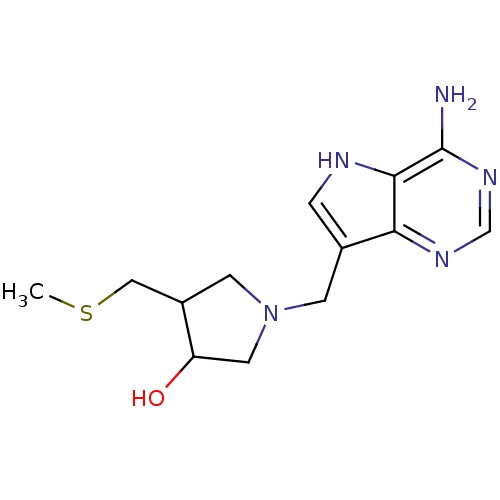
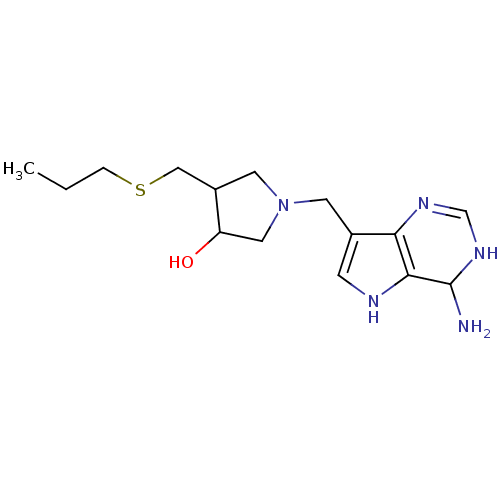
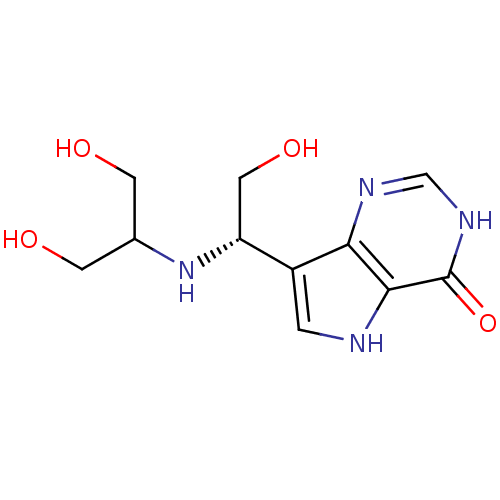
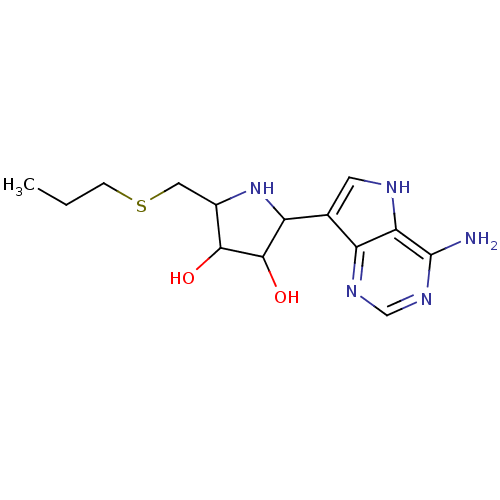
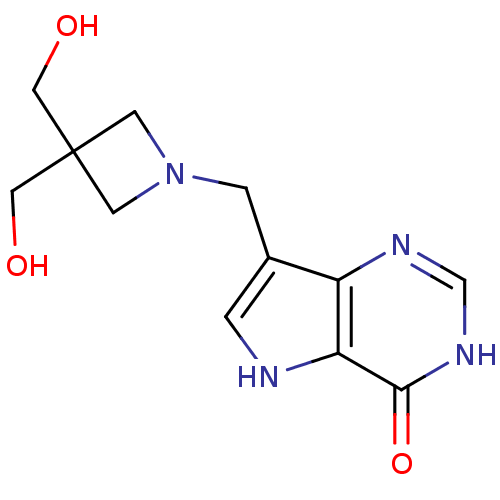
Affinity DataKi: 0.00200nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00300nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00500nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00680nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 0.00700nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00900nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.00900nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 0.0230nMAssay Description:Inhibitory activity of compound against bovine purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
Affinity DataKi: 0.0290nMAssay Description:Inhibitory activity of compound against human purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:Inhibitory activity of compound against bovine purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.0340nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0350nM ΔG°: -59.7kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0350nM ΔG°: -59.7kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Equilibrium dissociation constant determined against human purine nucleoside phosphorylase (PNP) after slow-onset inhibitionMore data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Inhibitory activity of compound against bovine purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Target5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase(Escherichia coli (strain K12))
Industrial Research
Industrial Research
Affinity DataKi: 0.0480nM ΔG°: -58.3kJ/molepH: 7.0 T: 2°CAssay Description:Enzyme activity was monitored by absorbance change in the xanthine oxidase coupled assay, which measures the formation of 2,8-dihydroxyadenine at 293...More data for this Ligand-Target Pair
Affinity DataKi: 0.0560nMAssay Description:Equilibrium dissociation constant determined against human purine nucleoside phosphorylase (PNP) after slow-onset inhibitionMore data for this Ligand-Target Pair
Affinity DataKi: 0.0560nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 0.0560nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0560nMAssay Description:Inhbitory activity of compound against human purine nucleoside phosphorylase (PNP)More data for this Ligand-Target Pair
Affinity DataKi: 0.0600nMAssay Description:Inhibitory activity of compound against bovine purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0720nM ΔG°: -57.9kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
Affinity DataKi: 0.0720nMAssay Description:Inhibitory activity of compound against human purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0760nM ΔG°: -57.8kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0760nM ΔG°: -57.8kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.0900nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
Affinity DataKi: 0.0960nMAssay Description:Inhibitory activity of compound against human purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0960nM ΔG°: -57.2kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
TargetS-methyl-5'-thioinosine phosphorylase(Pseudomonas aeruginosa)
Albert Einstein College Of Medicine
US Patent
Albert Einstein College Of Medicine
US Patent
Affinity DataKi: 0.0960nM ΔG°: -57.2kJ/molepH: 7.4 T: 2°CAssay Description:Assays for slow-onset inhibitors were carried out by adding 1 nM PaMTIP into reaction mixtures at 25 °C. containing 100 mM Hepes, pH 7.4, 100 mM ...More data for this Ligand-Target Pair
Affinity DataKi: 0.104nMAssay Description:Inhibitory activity of compound against human purine nucleoside phosphorylase(PNP)More data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.110nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.120nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.160nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.160nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
Affinity DataKi: 0.163nMAssay Description:Binding affinity towards Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.166nMAssay Description:Equilibrium dissociation constant towards human 5'-methylthioadenosine phosphorylaseMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.170nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair
Affinity DataKi: 0.180nMAssay Description:Equilibrium dissociation constant determined against human purine nucleoside phosphorylase (PNP) after slow-onset inhibitionMore data for this Ligand-Target Pair
Affinity DataKi: 0.180nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 0.210nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.214nMAssay Description:Equilibrium dissociation constant towards human 5'-methylthioadenosine phosphorylaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.220nMAssay Description:Inhibition of human purine nucleoside phosphorylase; Initial rate.More data for this Ligand-Target Pair
Affinity DataKi: 0.229nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.229nM ΔG°: -54.5kJ/molepH: 7.7 T: 2°CAssay Description:PNP activity was monitored by absorbance change in a coupled assay. In the assay, inosine was converted to hypoxanthine, and then hypoxanthine was co...More data for this Ligand-Target Pair
Affinity DataKi: 0.25nMAssay Description:Inhibition of human purine nucleoside phosphorylase; Initial rate.More data for this Ligand-Target Pair
TargetS-methyl-5'-thioadenosine phosphorylase(Homo sapiens (Human))
Industrial Research
Curated by ChEMBL
Industrial Research
Curated by ChEMBL
Affinity DataKi: 0.260nMAssay Description:Inhibition of human MTAP as equilibrium dissociation constantMore data for this Ligand-Target Pair


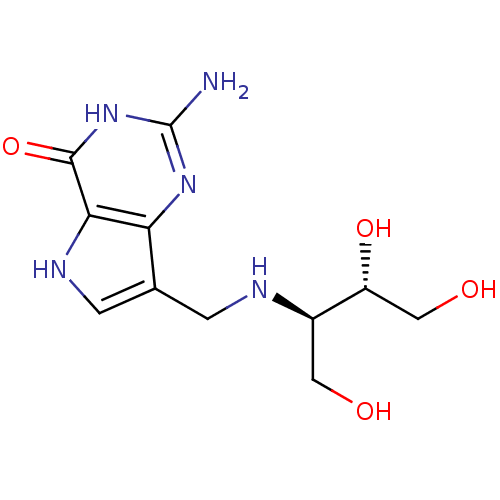
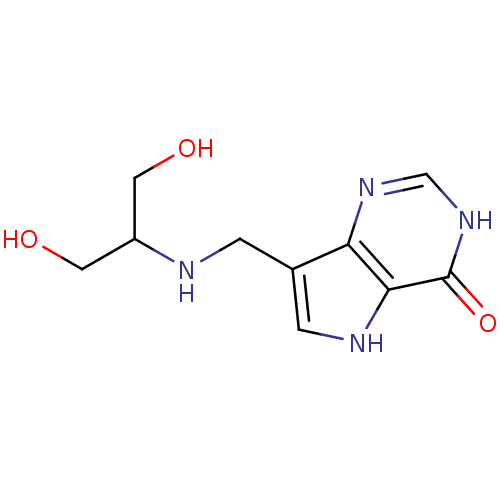
 3D Structure (crystal)
3D Structure (crystal)