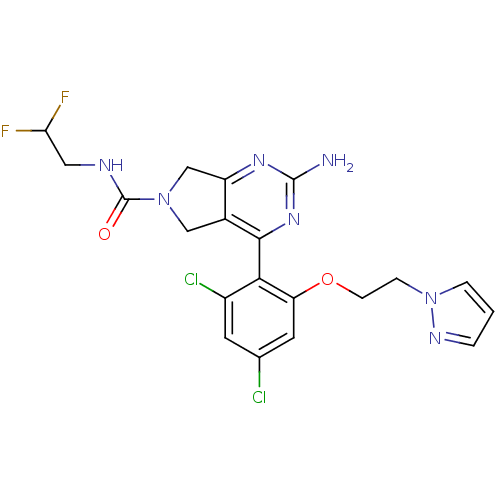
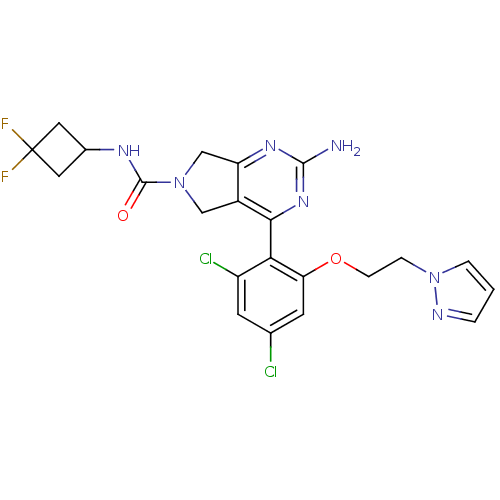
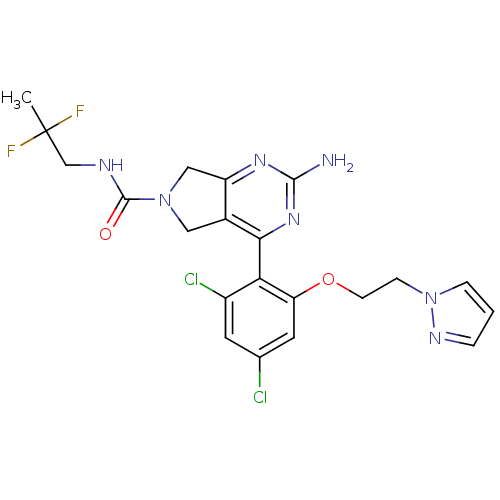
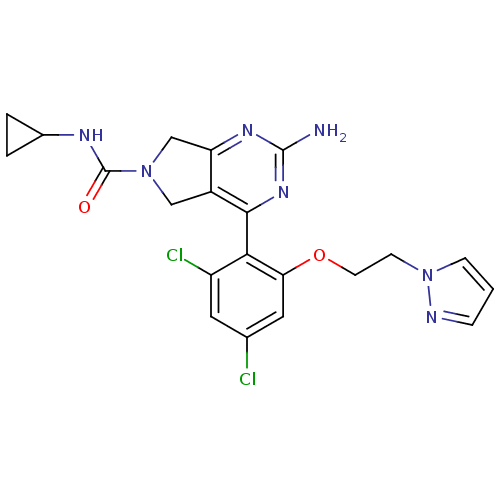
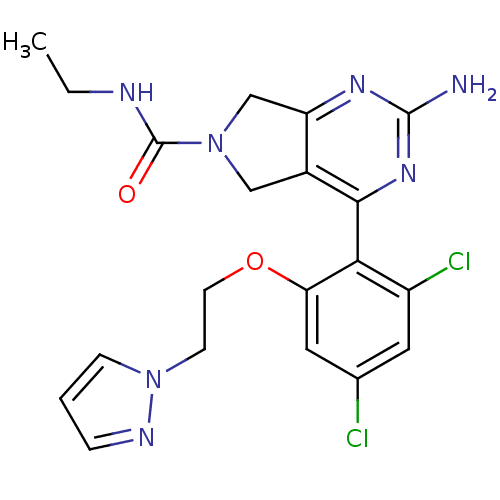
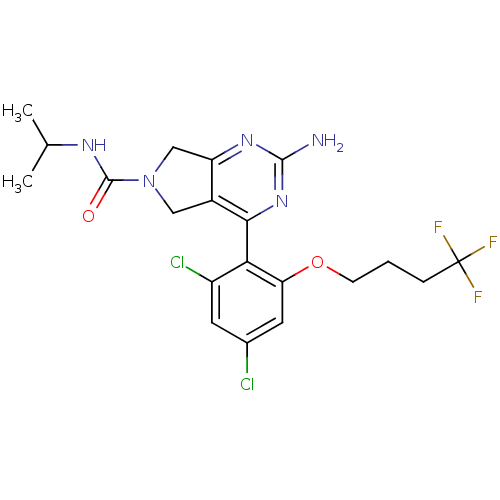
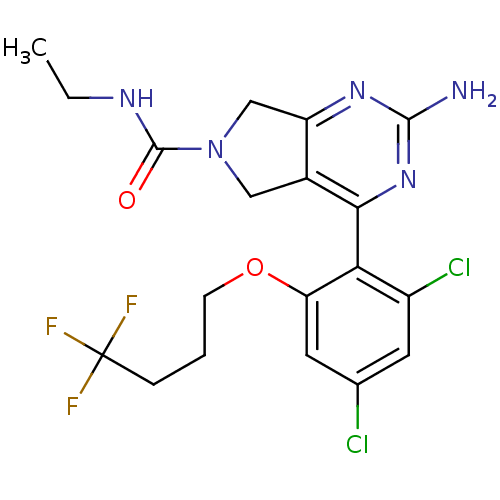
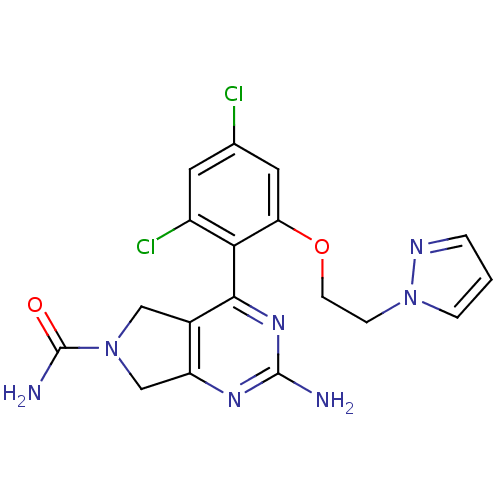
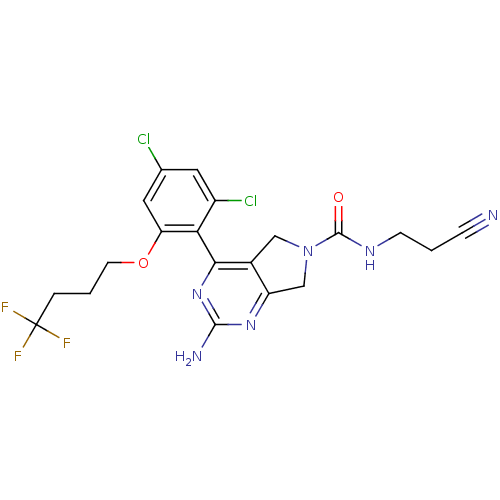
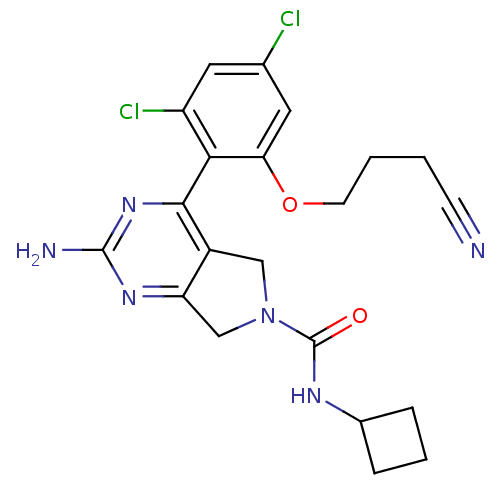
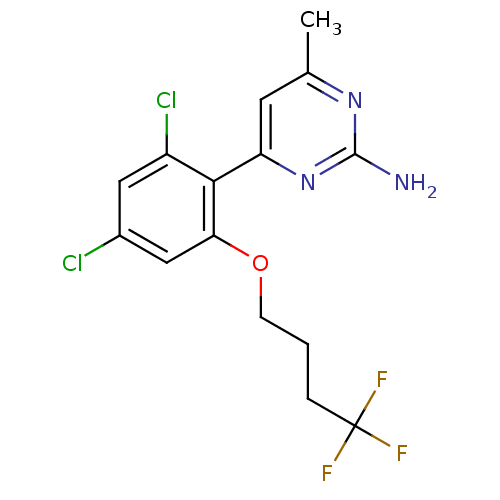
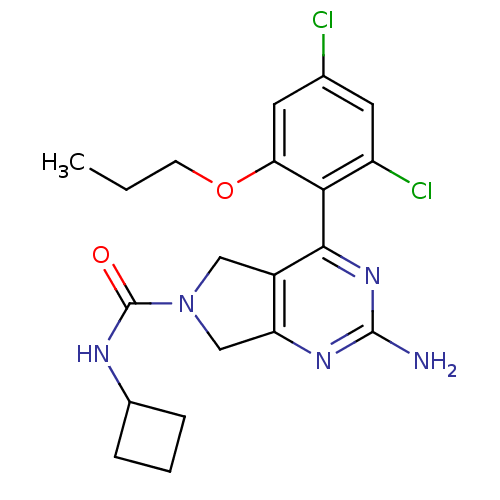
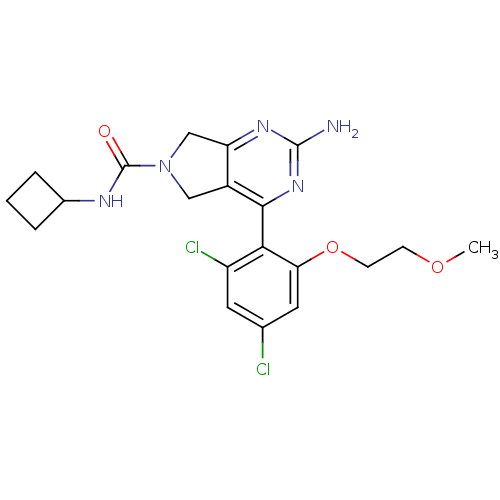
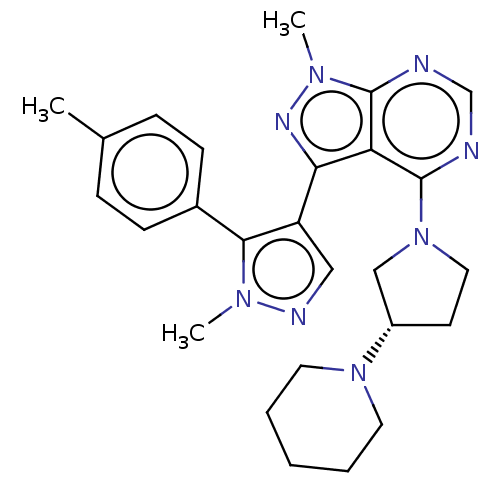
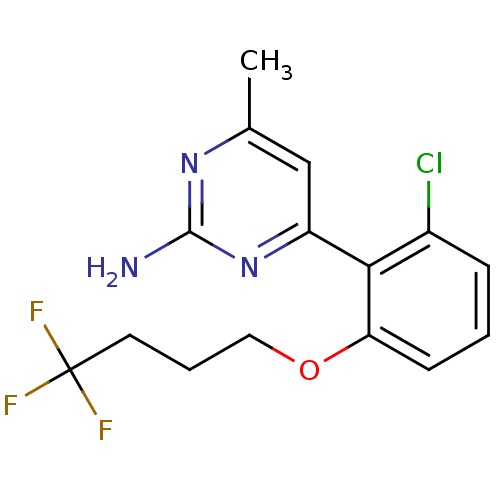
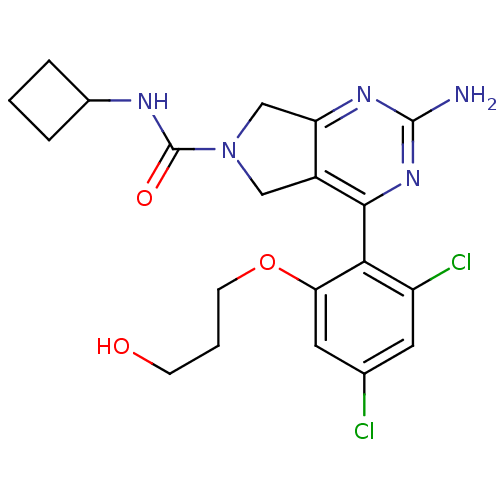
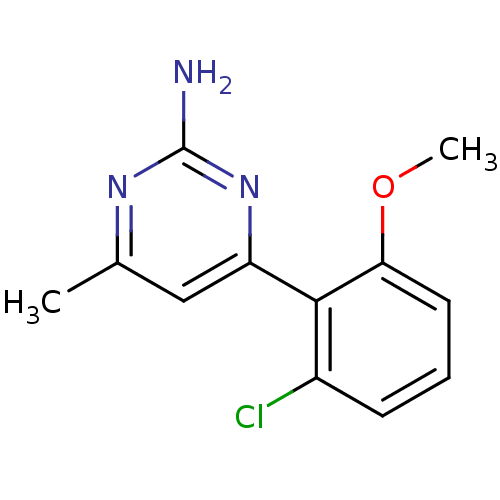
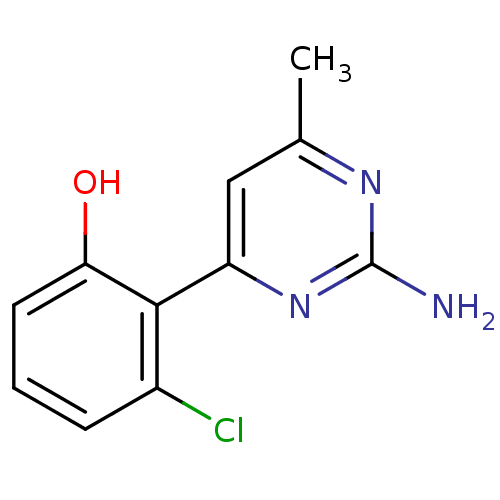
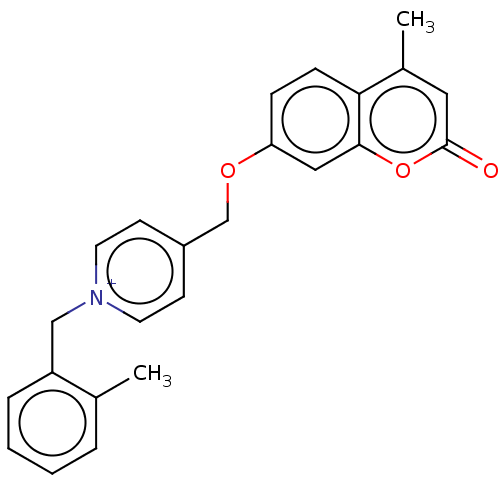
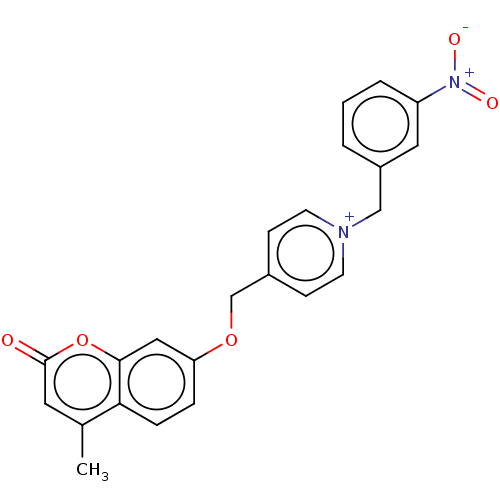
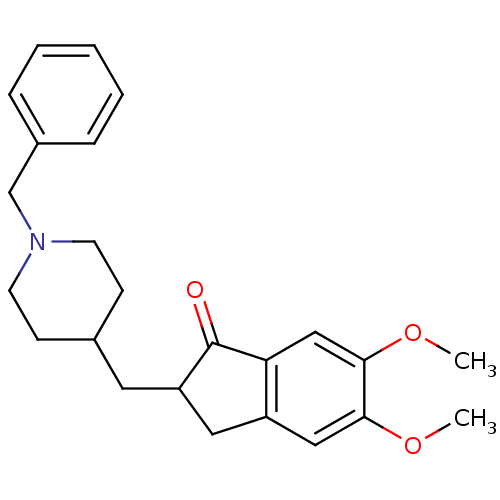
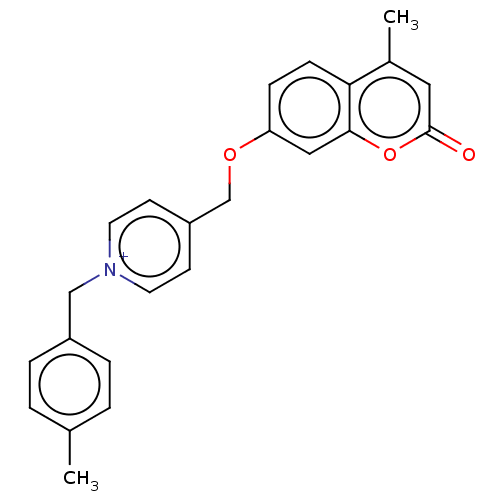
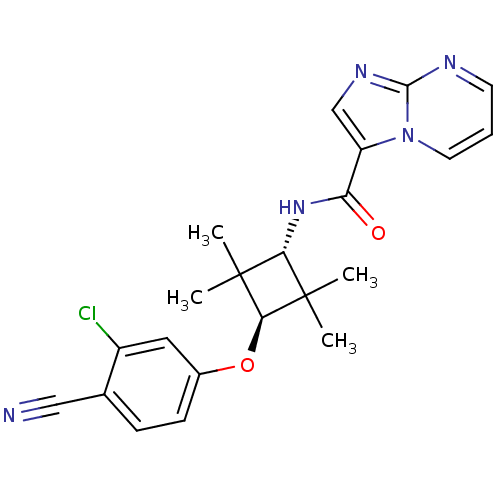
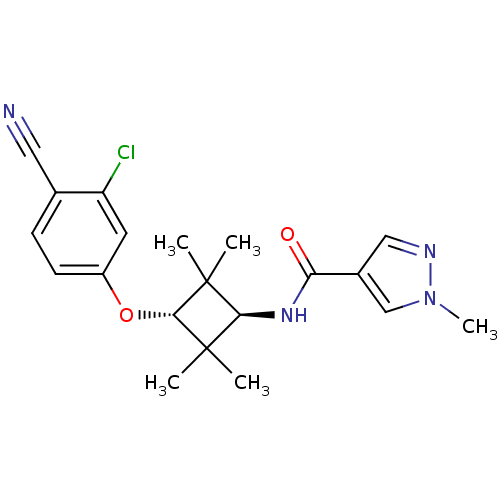
Affinity DataKi: 6nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
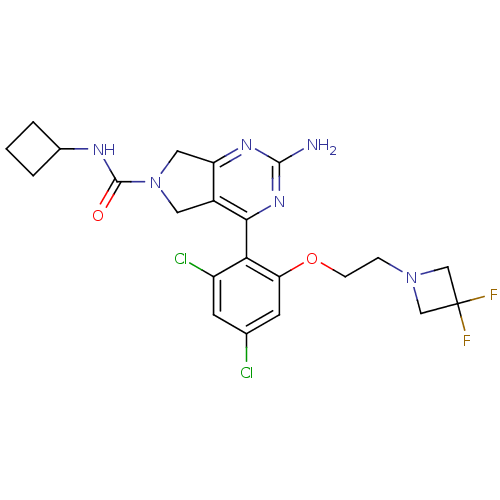
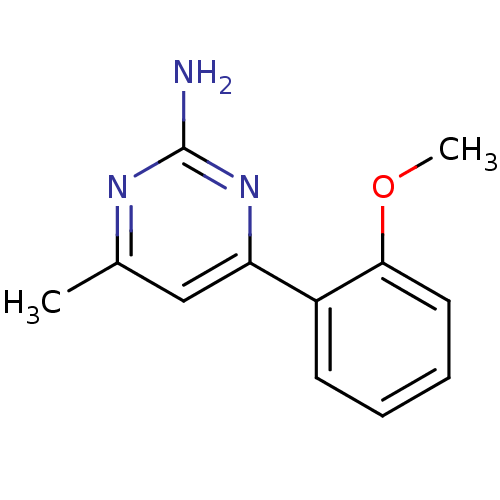
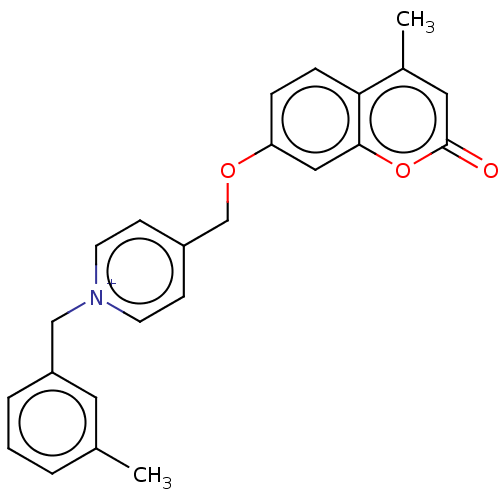
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
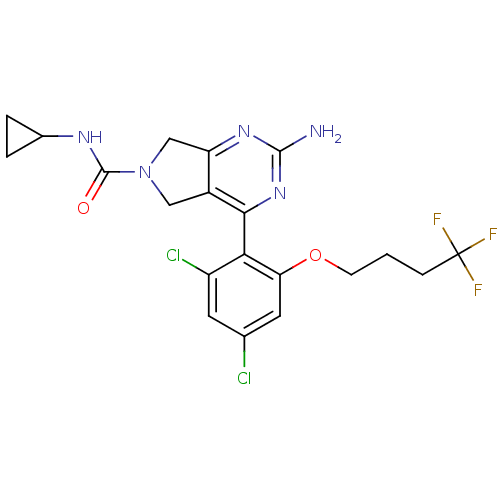
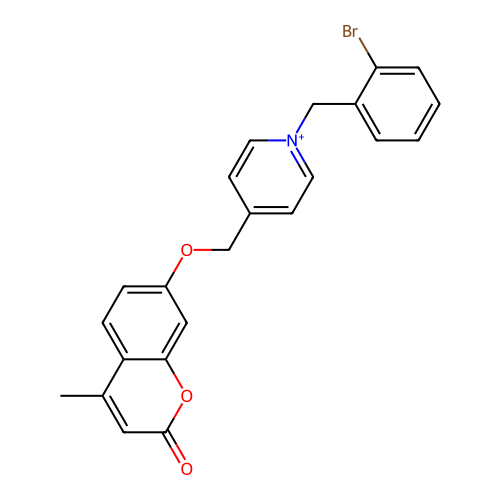
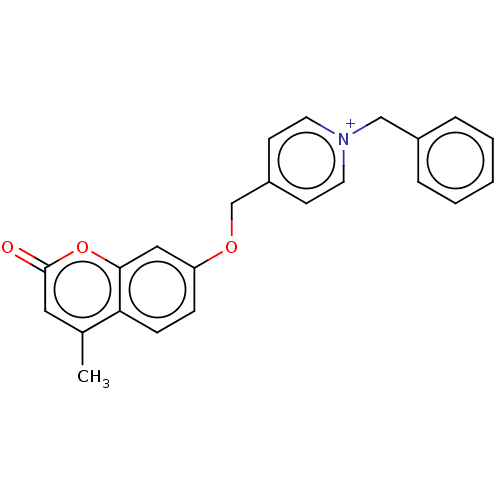
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
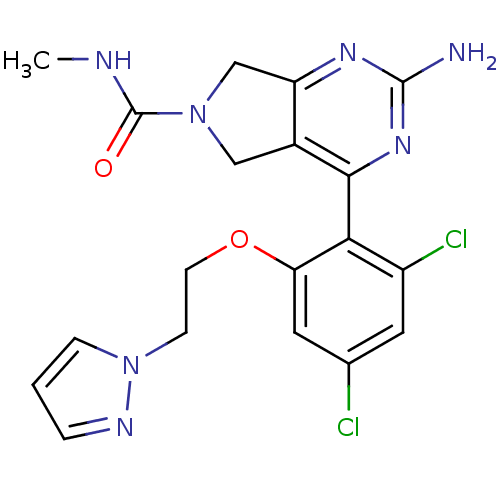
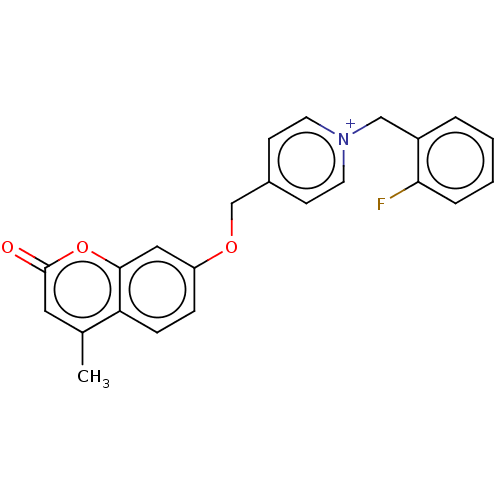
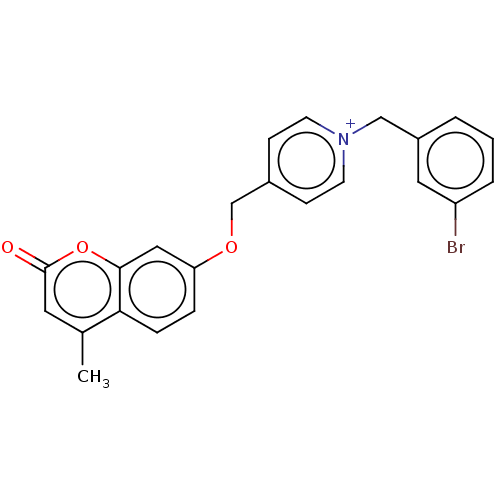
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 21nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 27nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 35nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 110nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Inhibition of midazolam 1'-hydroxylase activity of human recombinant CYP3A4 harboring human P450 oxidoreductase and b5 assessed as decrease in enzyme...More data for this Ligand-Target Pair
Affinity DataKi: 130nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:Inhibition of testosterone 6beta-hydroxylase activity of human recombinant CYP3A4 in presence of human P450 oxidoreductase and b5 assessed as decreas...More data for this Ligand-Target Pair
Affinity DataKi: 190nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 250nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 700nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:Inhibition of testosterone 6beta-hydroxylase activity of human recombinant CYP3A5 in presence of human P450 oxidoreductase and b5 assessed as decreas...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Inhibition of midazolam 1'-hydroxylase activity of human recombinant CYP3A5 harboring human P450 oxidoreductase and b5 assessed as decrease in enzyme...More data for this Ligand-Target Pair
Affinity DataKi: 8.30E+3nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Pfizer
Curated by ChEMBL
Pfizer
Curated by ChEMBL
Affinity DataKi: 1.16E+4nMAssay Description:Displacement of [3H]dofetilide from human ERG channel expressed in HEK293 cellsMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 0.480nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 2.70nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 4.20nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 4.40nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 4.40nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 4.60nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 4.90nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 7.70nMAssay Description:Inhibition of equine serum BuChE using S-butyrylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:Antagonist activity at human AR overexpressed in human LNCAP cells by luciferase reporter gene assayMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Antagonist activity at human AR overexpressed in human LNCAP cells by luciferase reporter gene assayMore data for this Ligand-Target Pair
TargetAcetylcholinesterase(Electrophorus electricus (Electric eel))
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Shanghai University Of Traditional Chinese Medicine
Curated by ChEMBL
Affinity DataIC50: 25nMAssay Description:Inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 6 mins followed by substrate addition measured up to 180...More data for this Ligand-Target Pair

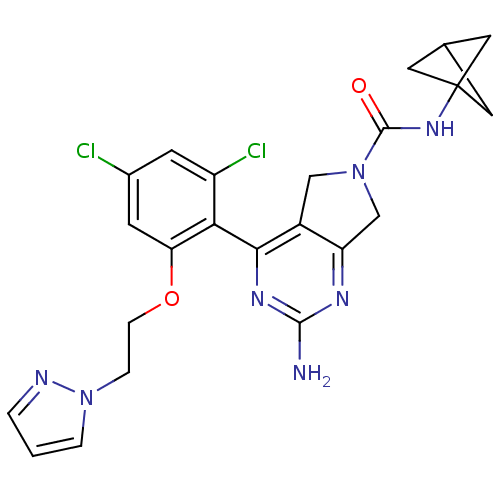

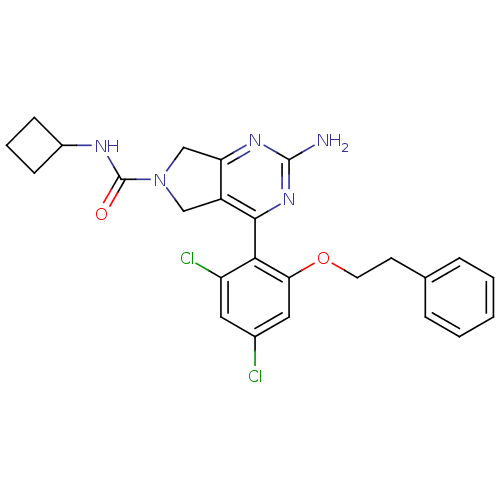
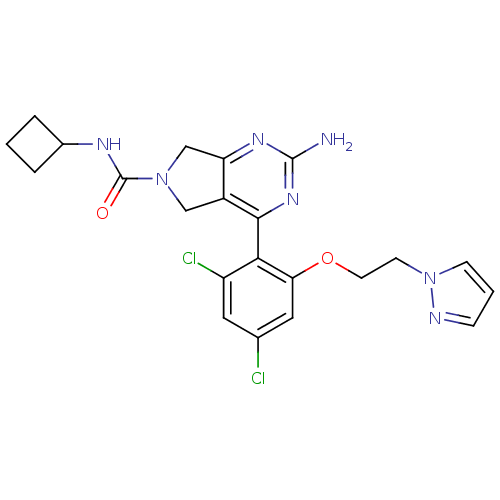
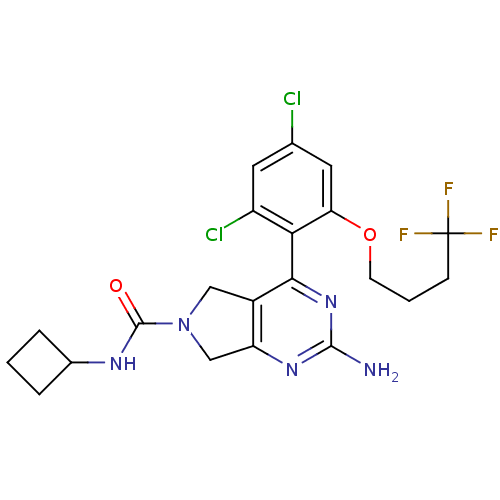
 3D Structure (crystal)
3D Structure (crystal)