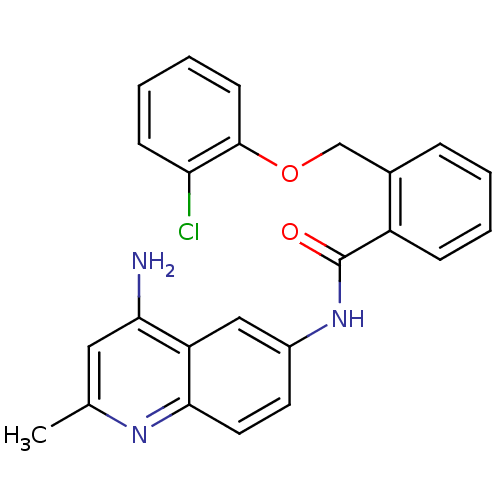
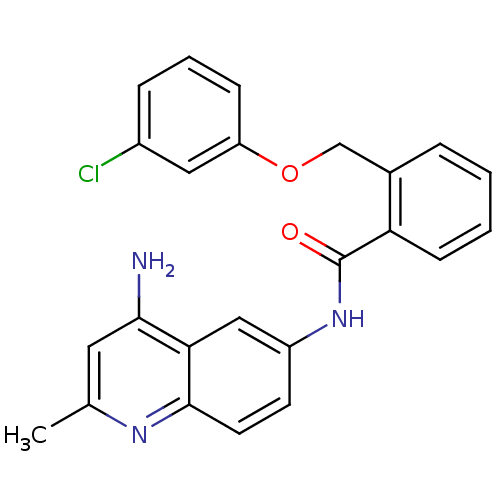
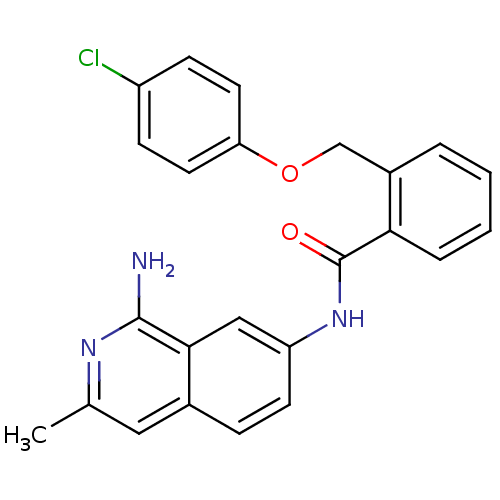
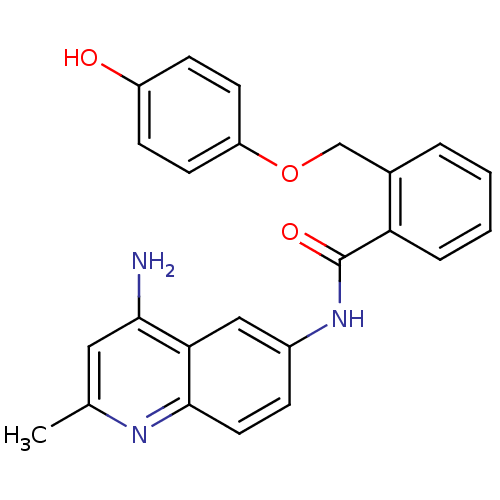
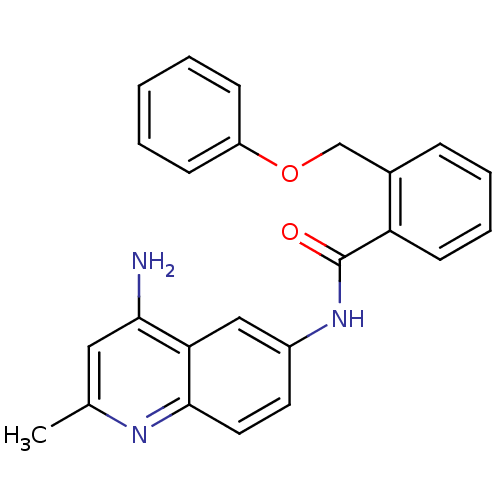
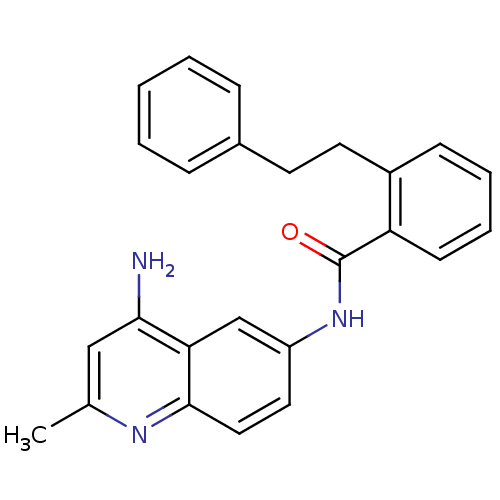
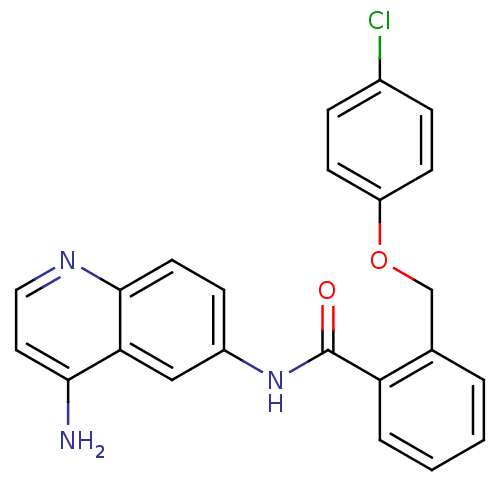
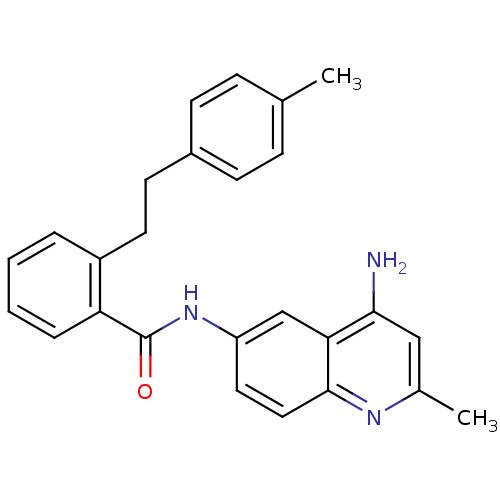
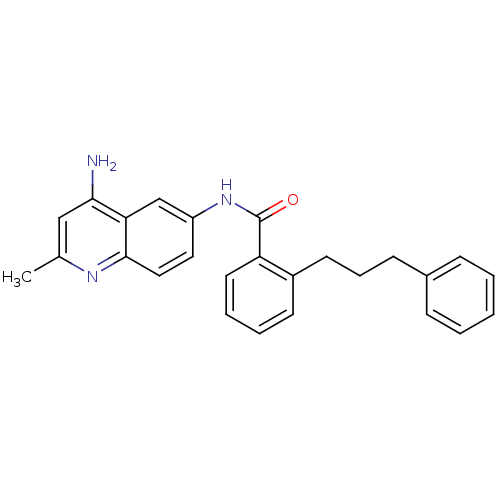
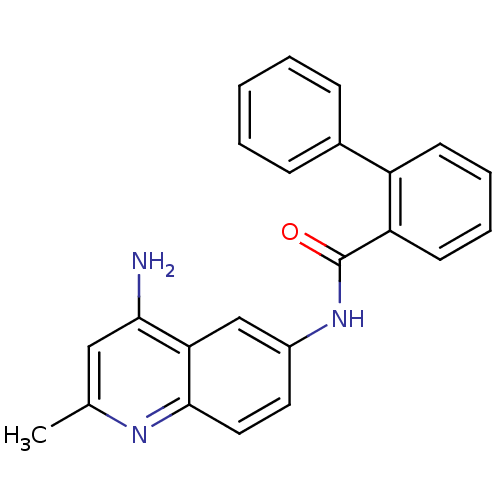
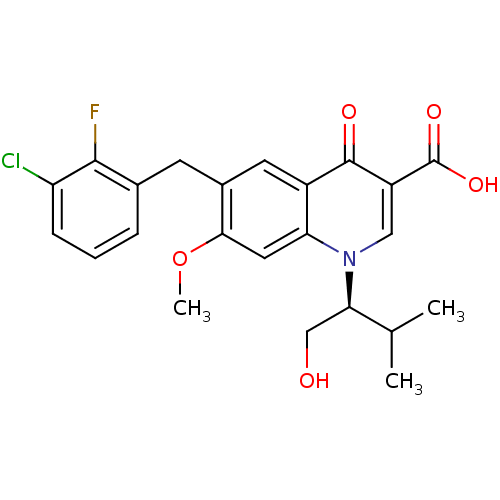
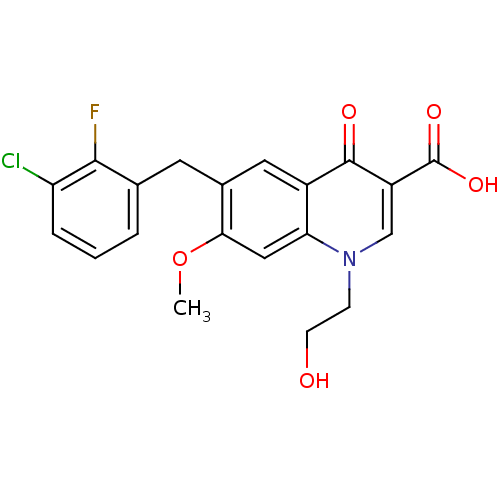
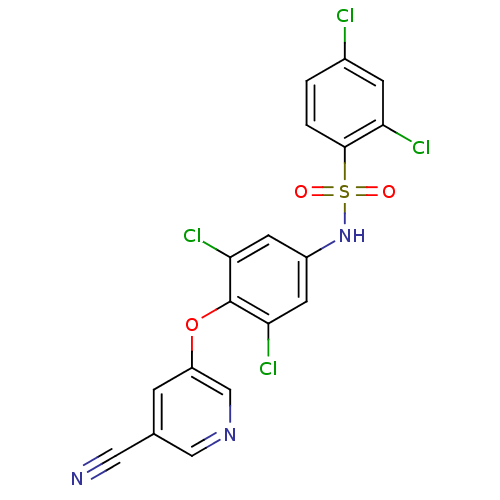
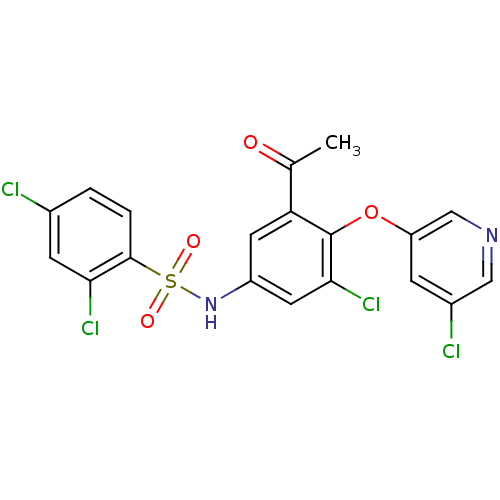
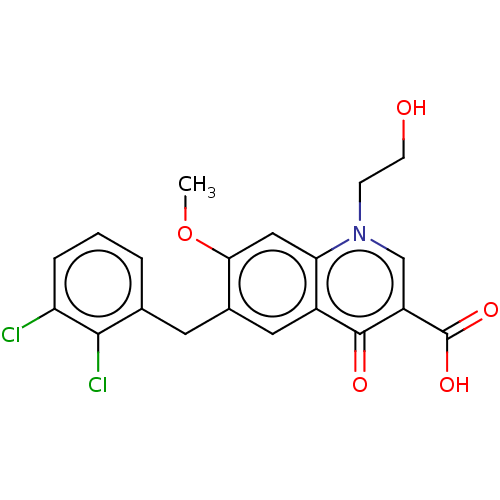
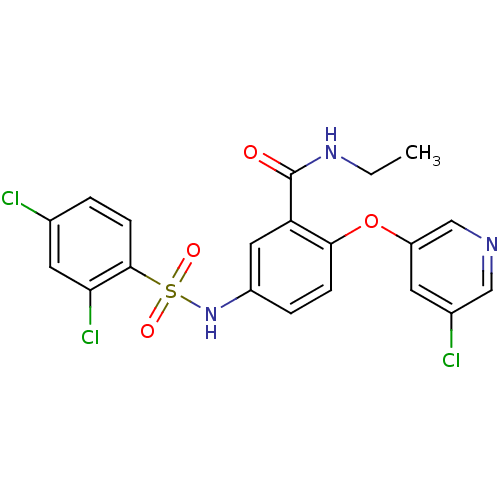
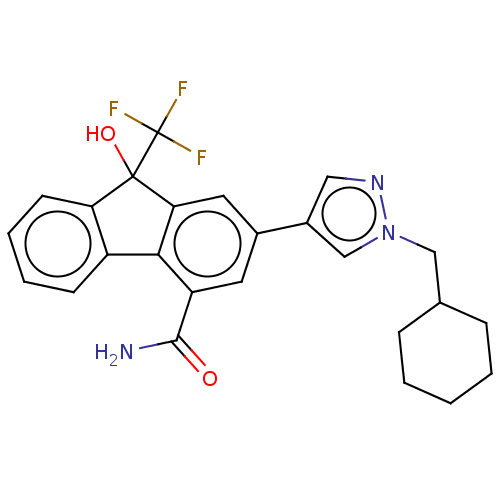
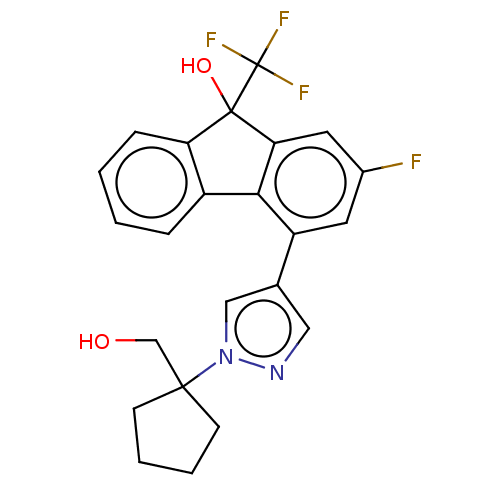
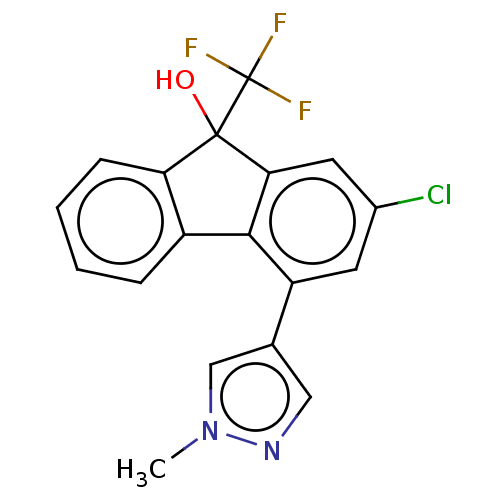
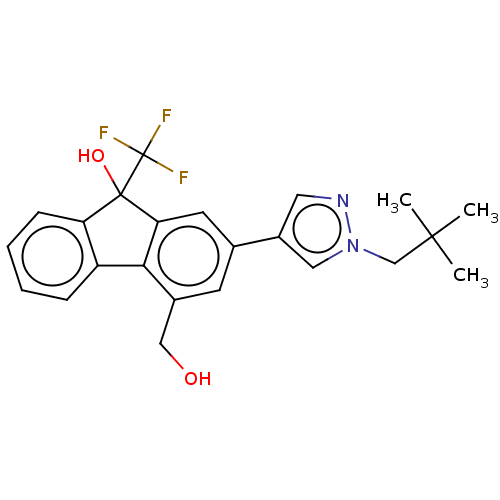
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
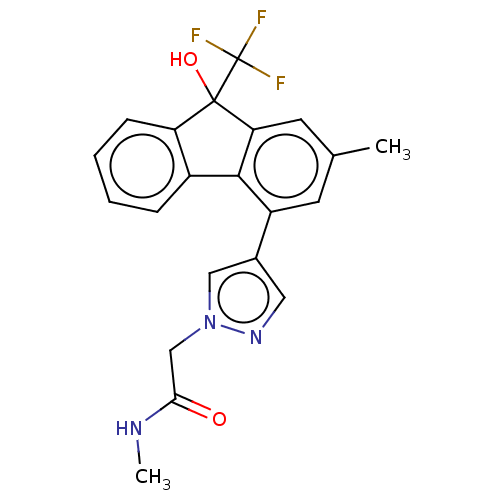
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
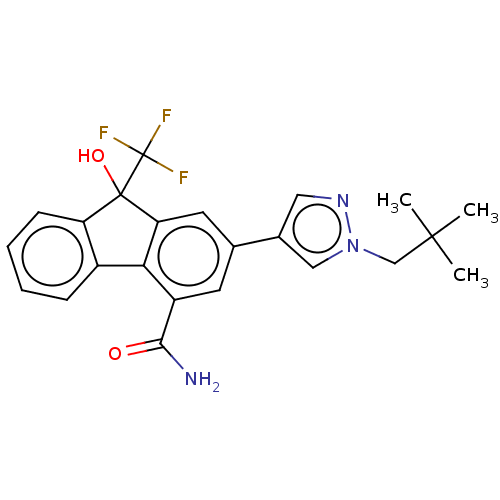
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
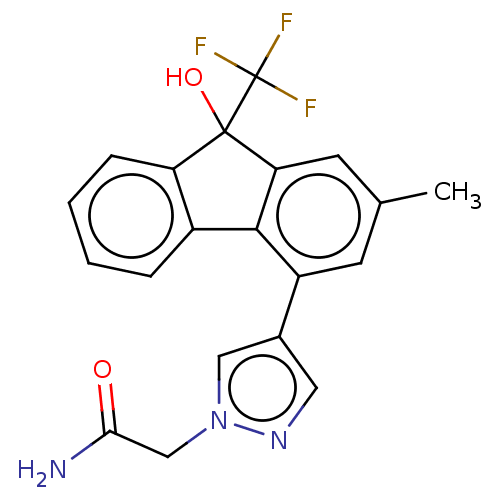
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 2.20nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 2.60nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 6.5nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 8.20nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
Affinity DataKi: 10nMAssay Description:Displacement of [3H]-rosiglitazone from GST-tagged PPARgammaLBD (unknown origin) after 1 hr by scintillation proximity assayMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 12nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 13nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 20nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 37nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 51nMAssay Description:Compound was evaluated for its ability to displace [3H]-nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 80nMAssay Description:Compound was evaluated for its ability to displace [3H]-nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 82nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 86nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 89nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetMu-type opioid receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 103nMAssay Description:Inhibition of [3H]diprenorphine (0.33 nM) binding from human Opioid receptor mu 1 expressed in CHO-K1 cells.More data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 121nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetNociceptin receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 369nMAssay Description:Compound was evaluated for its ability to displace [3H]nociceptin ( 0.5 nM ) binding from Opioid receptor like 1 expressed in HeLa cellsMore data for this Ligand-Target Pair
TargetKappa-type opioid receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 1.06E+3nMAssay Description:Inhibition of [3H]naltrindole (0.55 nM) binding from human Opioid receptor kappa 1More data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Homo sapiens (Human))
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataKi: 8.65E+3nMAssay Description:Inhibition of [3H]naltrindole (0.55 nM) binding from human Opioid receptor delta 1 expressed in CHO-K1 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
TargetIntegrase(Human immunodeficiency virus 1)
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 7.20nMAssay Description:Inhibition of recombinant HIV1 integrase strand transfer activityMore data for this Ligand-Target Pair
TargetIntegrase(Human immunodeficiency virus 1)
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 8.20nMAssay Description:Inhibition of recombinant HIV1 integrase strand transfer activityMore data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Inhibition of HIV1 integrase strand transfer activityMore data for this Ligand-Target Pair
TargetIntegrase(Human immunodeficiency virus 1)
Central Pharmaceutical Research Institute
Curated by ChEMBL
Central Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9.10nMAssay Description:Inhibition of recombinant HIV1 integrase strand transfer activityMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Displacement of [3H]-rosiglitazone from GST-tagged PPARgammaLBD (unknown origin) after 1 hr by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Inhibition of HIV1 integrase strand transfer activityMore data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 14nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Displacement of [3H]-rosiglitazone from GST-tagged PPARgammaLBD (unknown origin) after 1 hr by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Displacement of [3H]-rosiglitazone from GST-tagged PPARgammaLBD (unknown origin) after 1 hr by scintillation proximity assayMore data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 15nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 16nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 16nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 16nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 16nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 16nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:Displacement of [3H]-rosiglitazone from GST-tagged PPARgammaLBD (unknown origin) after 1 hr by scintillation proximity assayMore data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 17nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair
Target[Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 2, mitochondrial(Homo sapiens (Human))
Japan Tobacco
US Patent
Japan Tobacco
US Patent
Affinity DataIC50: 17nMpH: 8.0 T: 2°CAssay Description:The inhibitory action of PDHK activity was indirectly evaluated by performing a kinase reaction in the presence of a test compound and measuring the ...More data for this Ligand-Target Pair



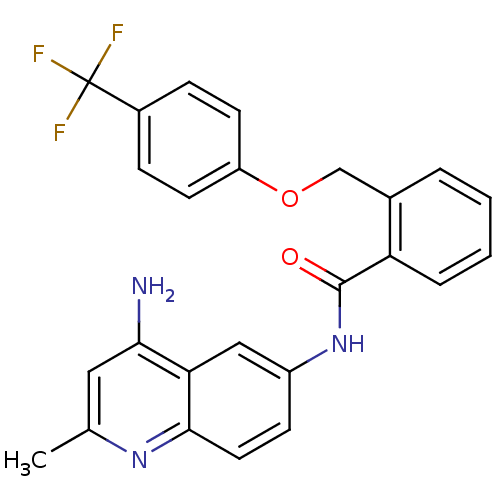




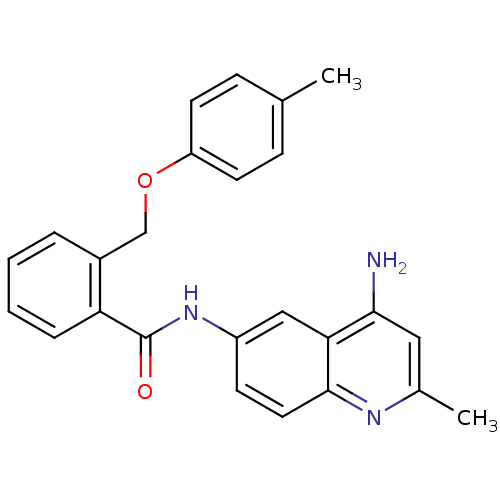


 3D Structure (crystal)
3D Structure (crystal)