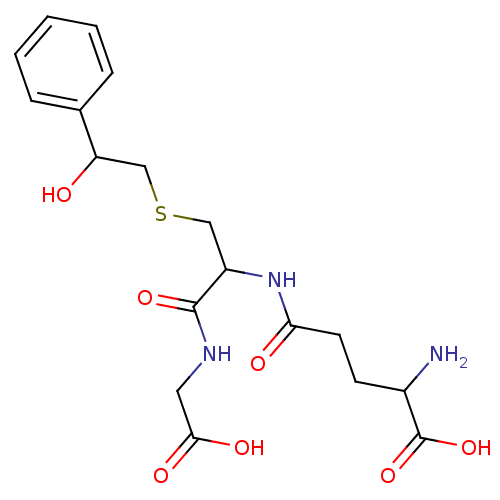
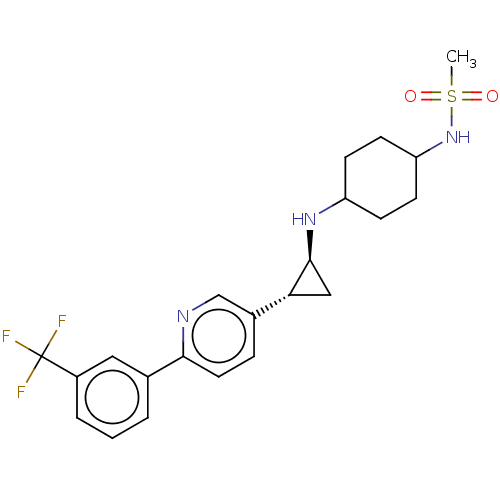
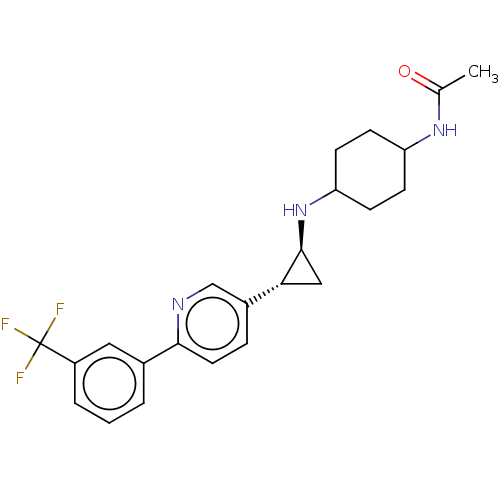
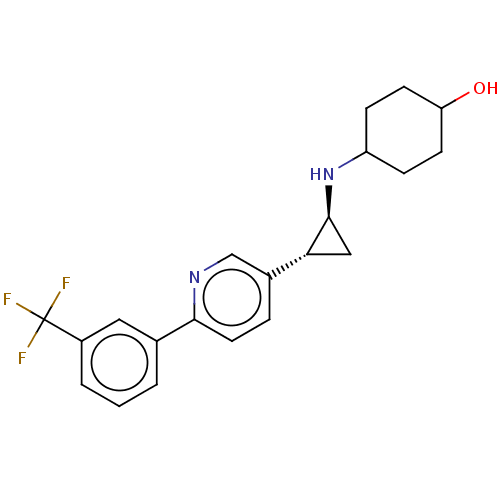
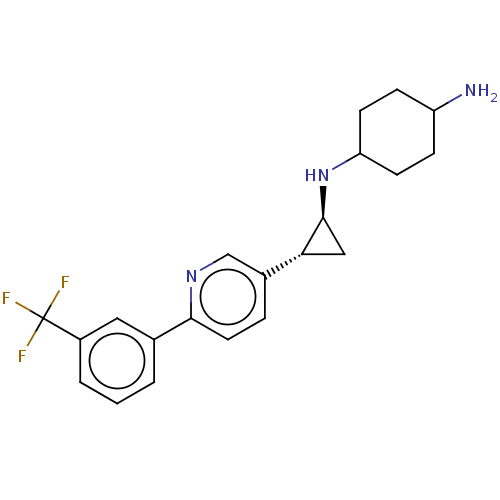
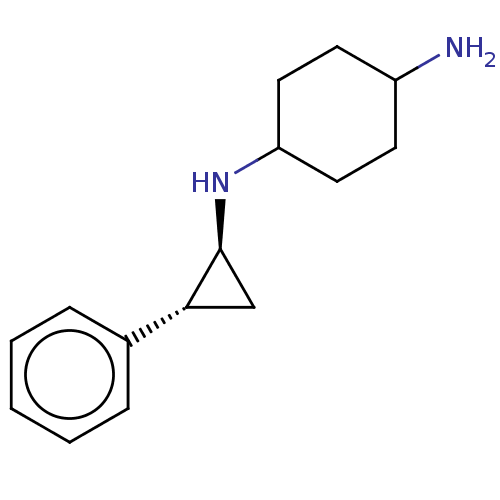
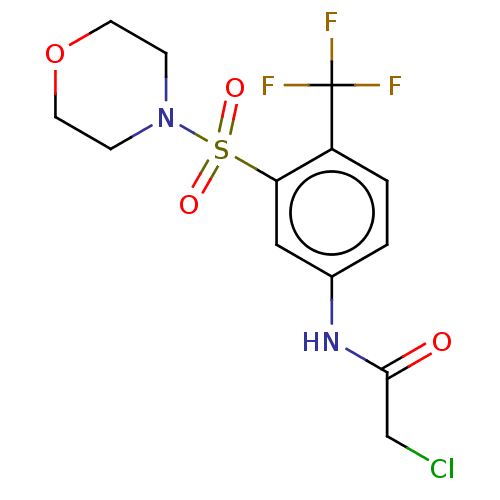
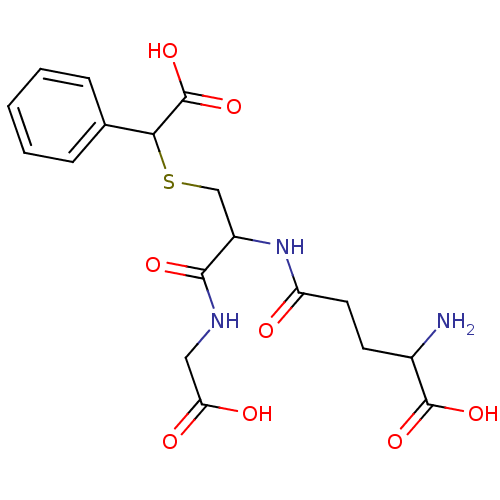
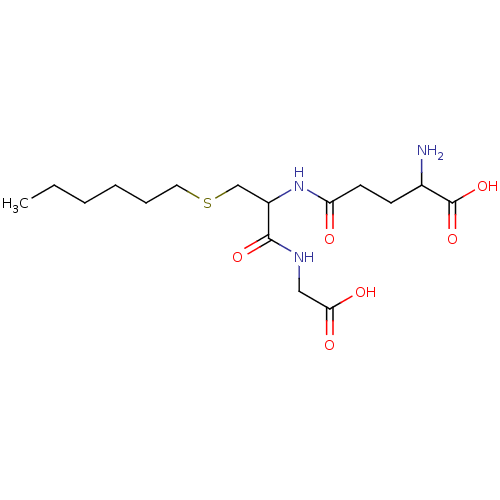
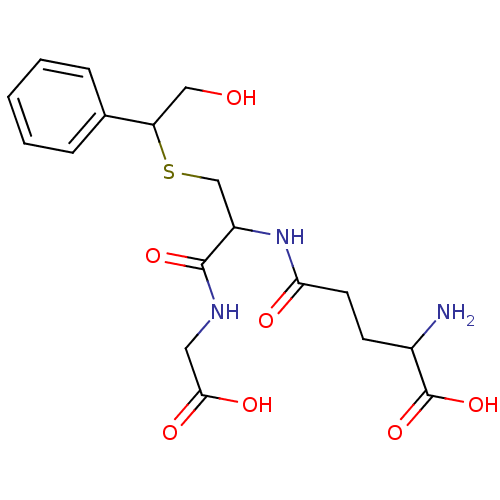
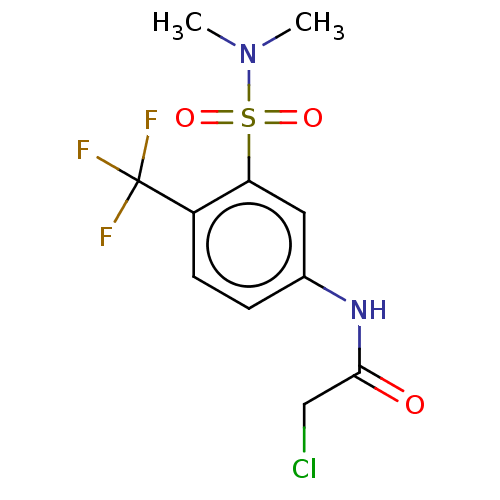
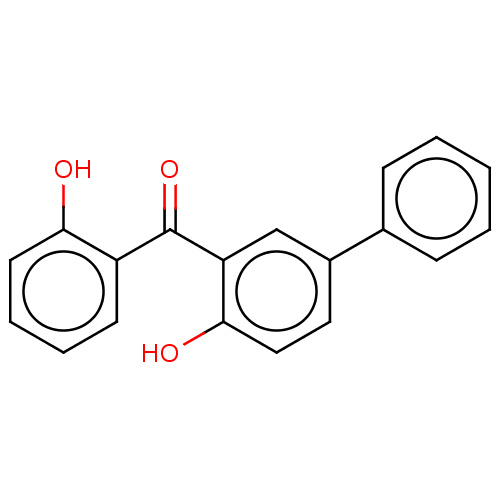
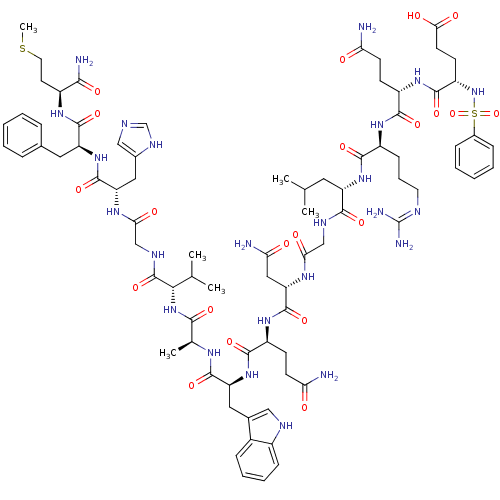
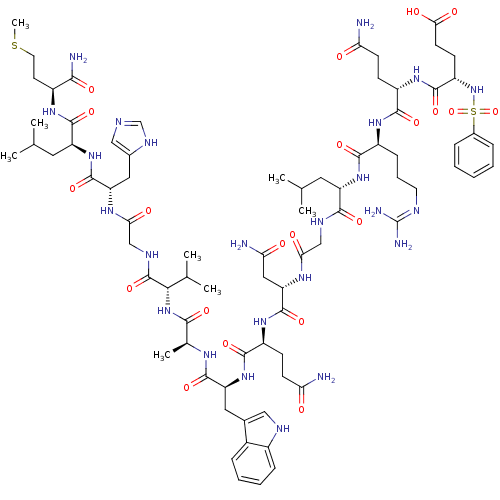
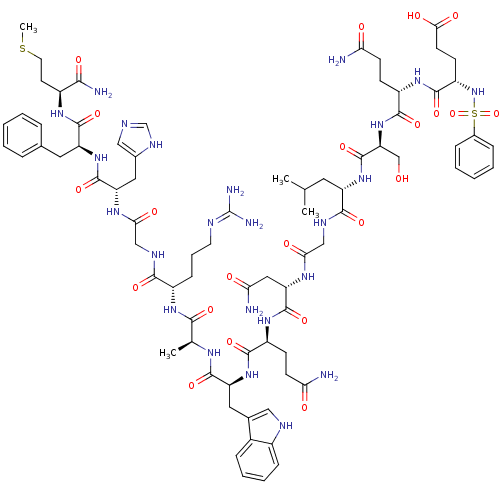
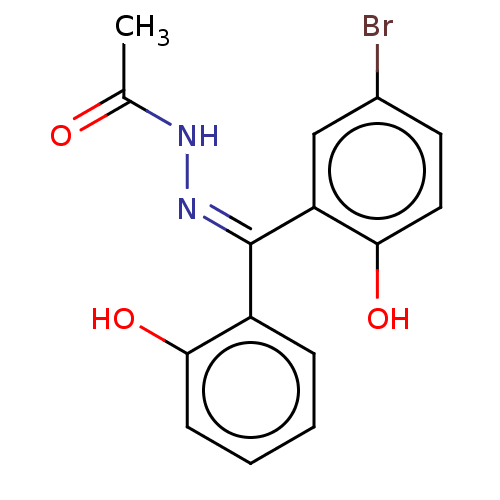
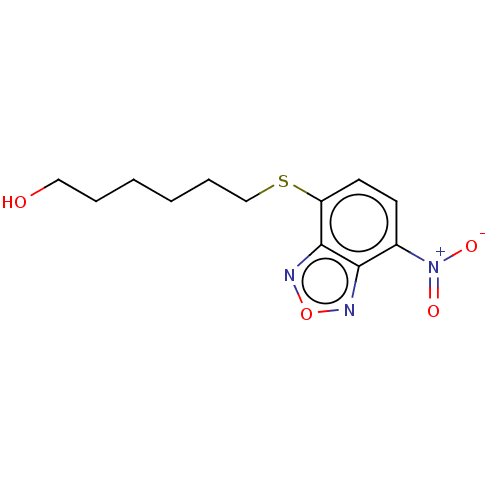
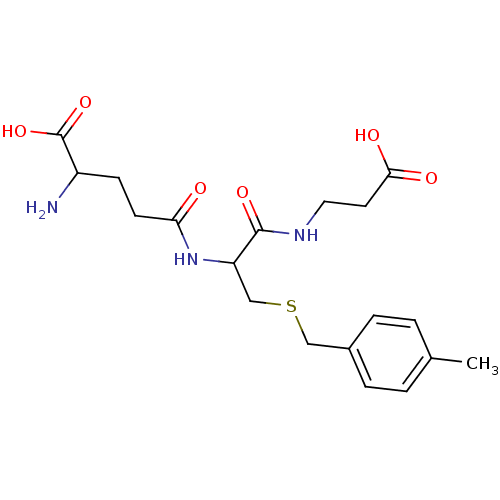
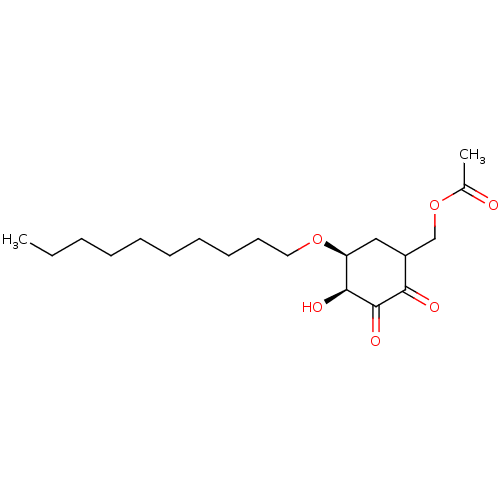
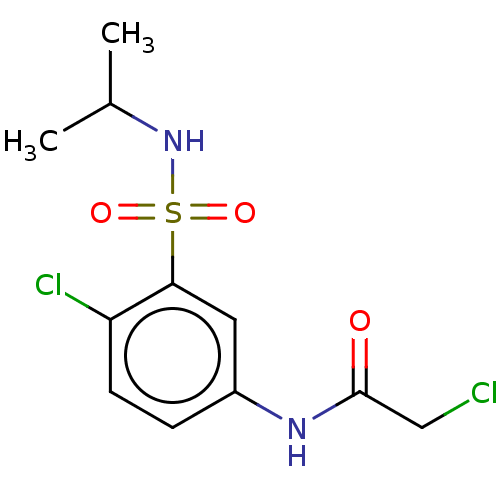
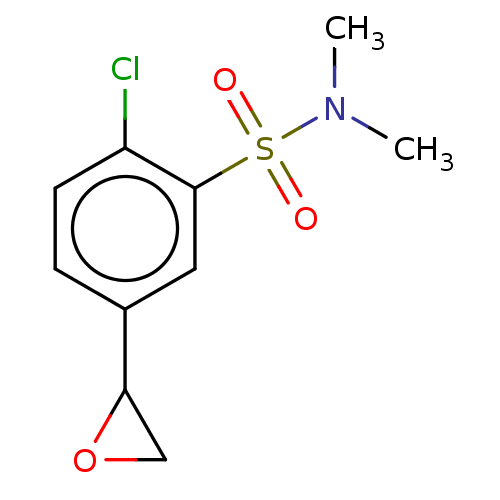
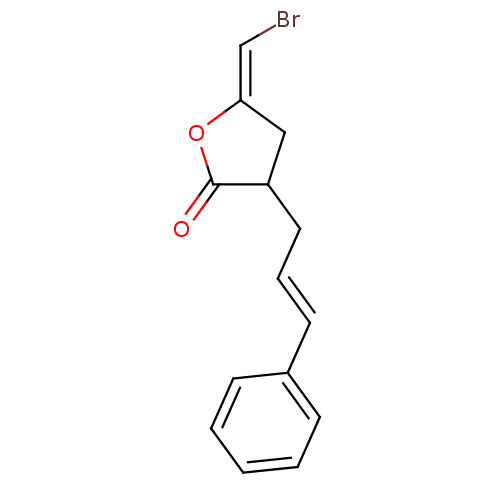
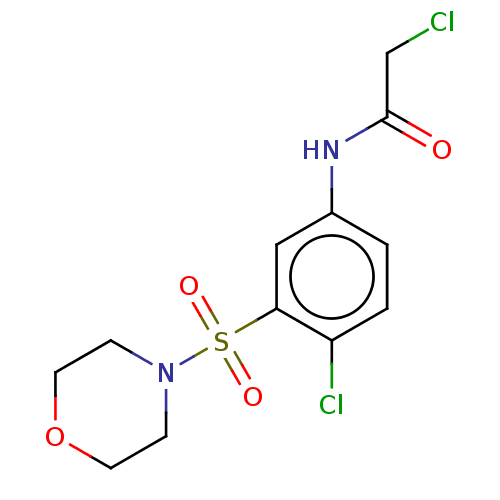
TargetGlutathione S-transferase theta-1(Rattus norvegicus)
Research Triangle Institute
Curated by ChEMBL
Research Triangle Institute
Curated by ChEMBL
Affinity DataKi: 0.680nMAssay Description:The compound was tested for binding activity against muscarinic acetylcholine receptor M3, using [3H]-QNB as the radioligand.More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 50nM ΔG°: -43.3kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
Affinity DataKi: 120nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione S-transferase PMore data for this Ligand-Target Pair
Affinity DataKi: 170nMAssay Description:Time dependent inhibition of GSTP1-1 (unknown origin) assessed as inhibition constant using reduced GSH and CDNB as substrate by spectrophotometric m...More data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 360nMAssay Description:Competitive inhibition of human GSTA1 activity by double reciprocal Lineweaver-Burk graphMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 380nMAssay Description:Competitive inhibition of human GSTA1 activity by double reciprocal Lineweaver-Burk graphMore data for this Ligand-Target Pair
Affinity DataKi: 420nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione S-transferase PMore data for this Ligand-Target Pair
Affinity DataKi: 440nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
Affinity DataKi: 490nMAssay Description:Inhibition constant against glutathione S-transferase pi using GSH (0.1-3mM), 1 mM 1-chloro-2,4-dinitrobenzeneMore data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 550nM ΔG°: -37.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
Affinity DataKi: 610nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
Affinity DataKi: 660nMAssay Description:Inhibition constant against glutathione S-transferase pi using GSH (0.1-3mM), 1 mM 1-chloro-2,4-dinitrobenzeneMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 840nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione-S-transferase A1 enzymeMore data for this Ligand-Target Pair
Affinity DataKi: 850nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione S-transferase PMore data for this Ligand-Target Pair
Affinity DataKi: 1.11E+3nMAssay Description:Inhibition constant against glutathione S-transferase pi using GSH (0.1-3mM), 1 mM 1-chloro-2,4-dinitrobenzeneMore data for this Ligand-Target Pair
Affinity DataKi: 1.20E+3nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 1.47E+3nMAssay Description:Competitive inhibition of human GSTA1 activity by double reciprocal Lineweaver-Burk graphMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 1.70E+3nMAssay Description:Inhibition of human GSTA1-1 expressed in Escherichia coli BL21 (DE3) by 1-chloro-2,4-dinitrobenzene competitive assayMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 1.70E+3nMAssay Description:Inhibition of human GSTA1-1 expressed in Escherichia coli BL21 (DE3) by 1-chloro-2,4-dinitrobenzene competitive assayMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 1.70E+3nMAssay Description:Inhibition of human GSTA1-1 expressed in Escherichia coli BL21 (DE3) by 1-chloro-2,4-dinitrobenzene competitive assayMore data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 1.75E+3nMAssay Description:Competitive inhibition of human GSTA1 activity by double reciprocal Lineweaver-Burk graphMore data for this Ligand-Target Pair
Affinity DataKi: 1.85E+3nMAssay Description:Time dependent inhibition of GSTP1-1 (unknown origin) assessed as inhibition constant using reduced GSH and CDNB as substrate by spectrophotometric m...More data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione S-transferase Mu 1More data for this Ligand-Target Pair
Affinity DataKi: 2.10E+3nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione S-transferase Mu 1More data for this Ligand-Target Pair
Affinity DataKi: 2.50E+3nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
Affinity DataKi: 3.20E+3nMAssay Description:Competitive inhibition of human GSTM1 using GSH as substrate by Lineweaver-Burk plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 4.70E+3nMAssay Description:Noncompetitive inhibition of human GSTM1 using CDNB as substrate by Lineweaver-Burk plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 4.80E+3nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876](Homo sapiens (Human))
Oryzon Genomics
US Patent
Oryzon Genomics
US Patent
Affinity DataKi: 5.50E+3nM ΔG°: -31.2kJ/molepH: 7.4 T: 2°CAssay Description:The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ...More data for this Ligand-Target Pair
TargetGlutathione S-transferase A1(Homo sapiens (Human))
Agricultural University Of Athens
Curated by ChEMBL
Agricultural University Of Athens
Curated by ChEMBL
Affinity DataKi: 5.80E+3nMAssay Description:Inhibitory activity of the compound was measured on recombinant human Glutathione-S-transferase A1 enzymeMore data for this Ligand-Target Pair
Affinity DataKi: 6.10E+3nMAssay Description:Inhibitory activity of the compound for human Glutathione S-transferase P was determinedMore data for this Ligand-Target Pair
Affinity DataKi: 6.70E+3nMAssay Description:Inhibition of recombinant GSTO1-1 (unknown origin) expressed in Escherichia coli assessed as inhibitor constant using S-(4-nitrophenacyl)glutathione ...More data for this Ligand-Target Pair




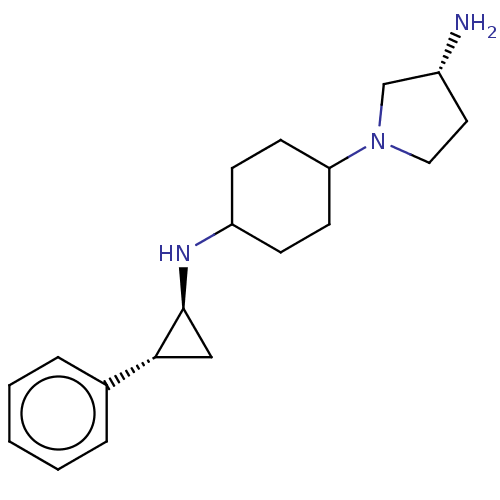
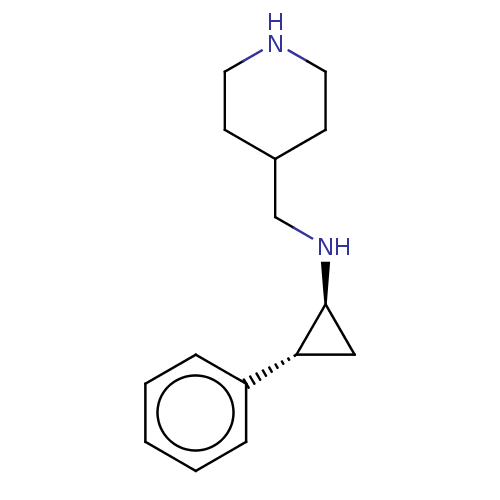

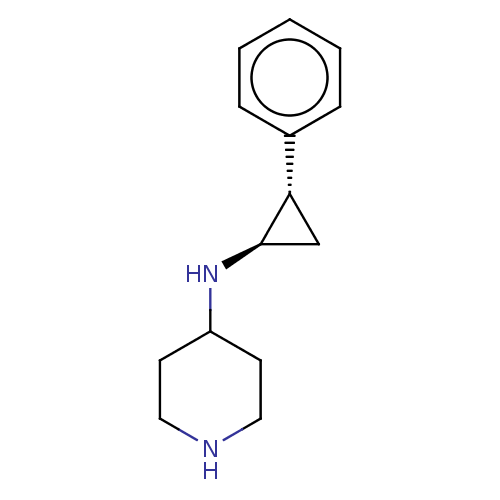
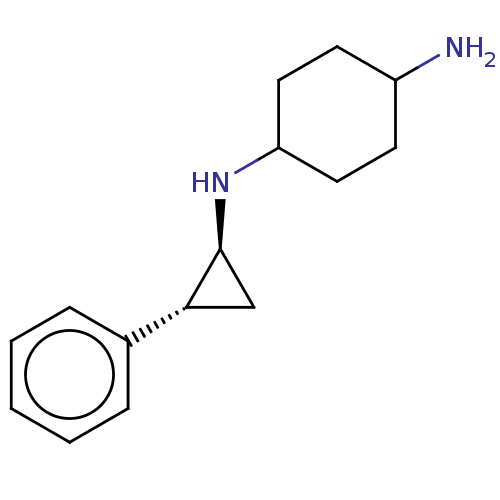


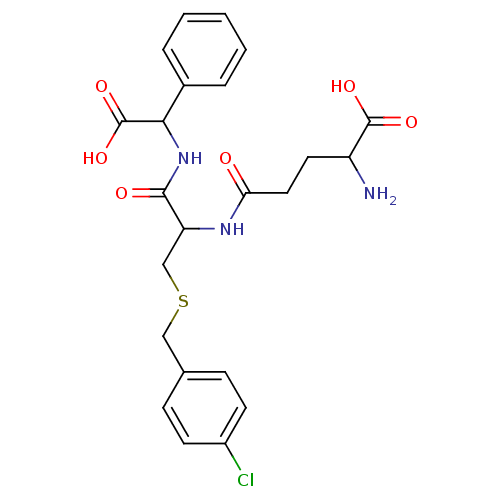


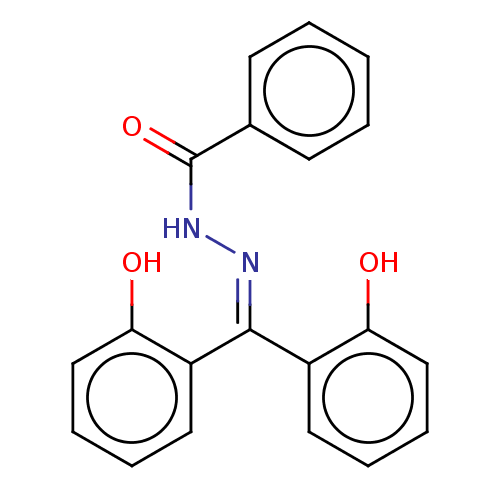

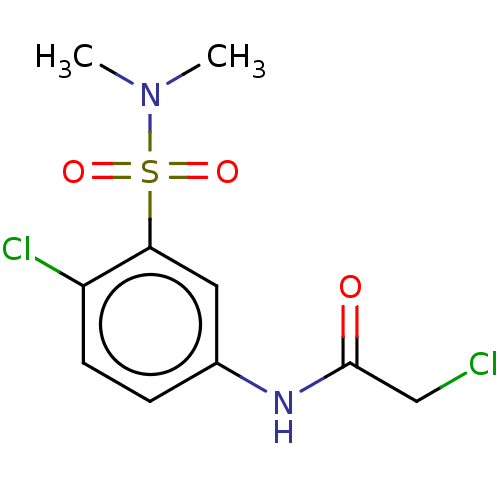
 3D Structure (crystal)
3D Structure (crystal)