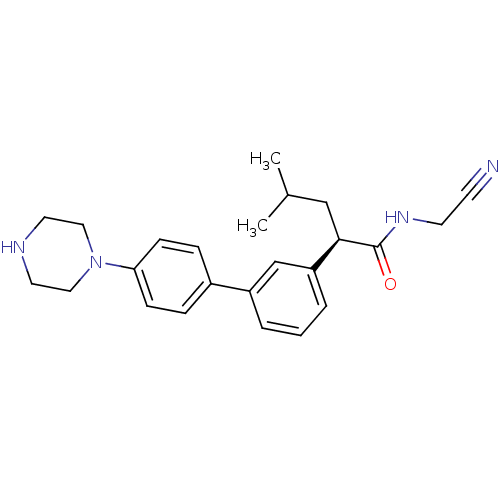
Affinity DataKi: 4.64E+3nMAssay Description:Inhibition of human cathepsin LMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nM EC50: 320nMpH: 5.5 T: 2°CAssay Description:Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.73E+3nMAssay Description:Inhibitory activity against cathepsin L from humanMore data for this Ligand-Target Pair
Affinity DataIC50: 3.73E+3nMAssay Description:Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.73E+3nMAssay Description:Inhibitory concentration against human cathepsin LMore data for this Ligand-Target Pair