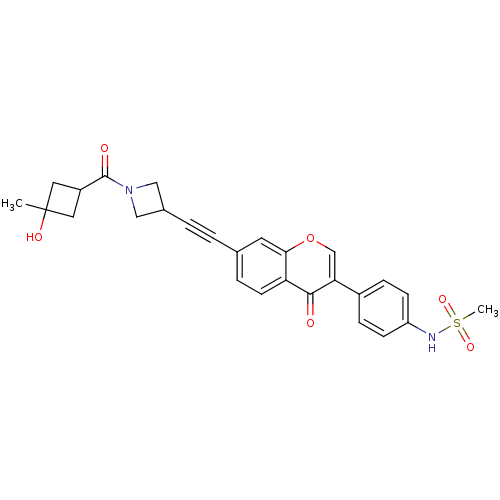
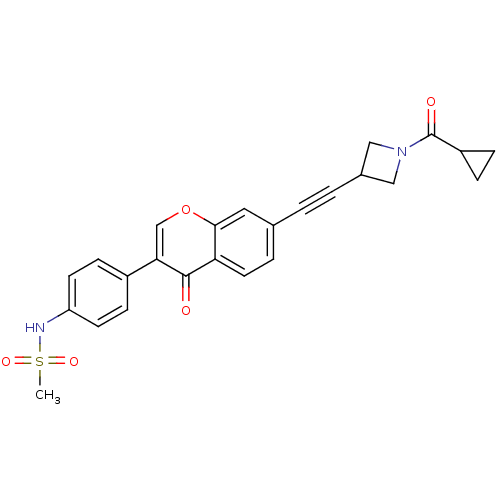
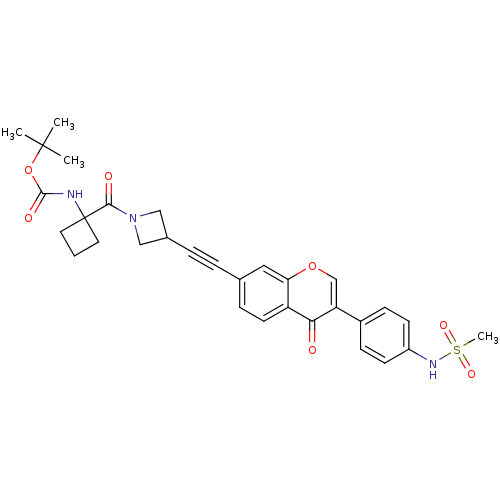
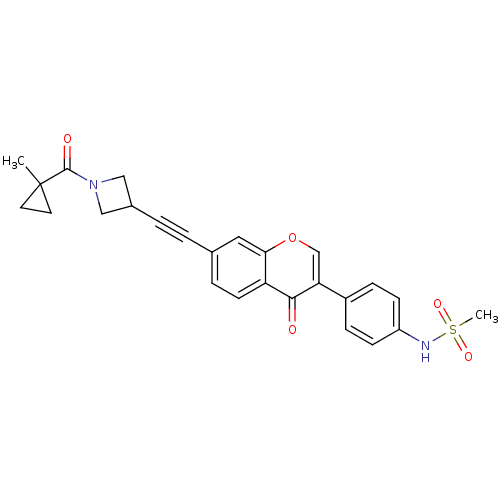
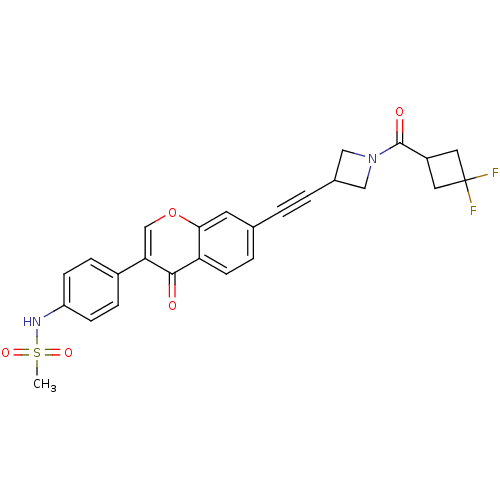
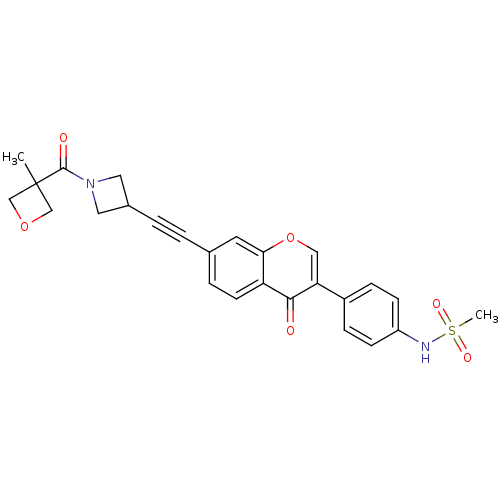
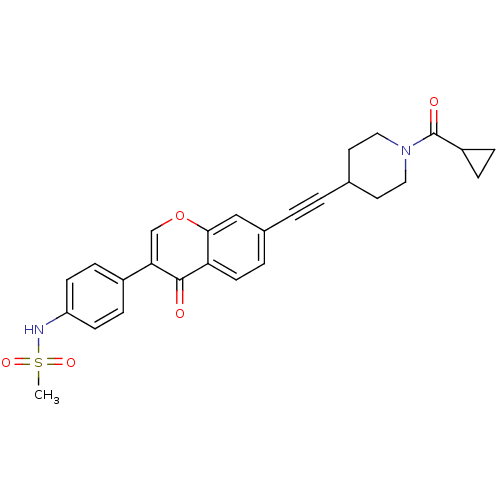
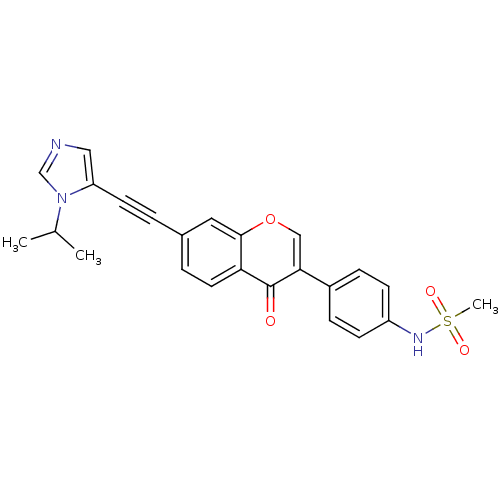
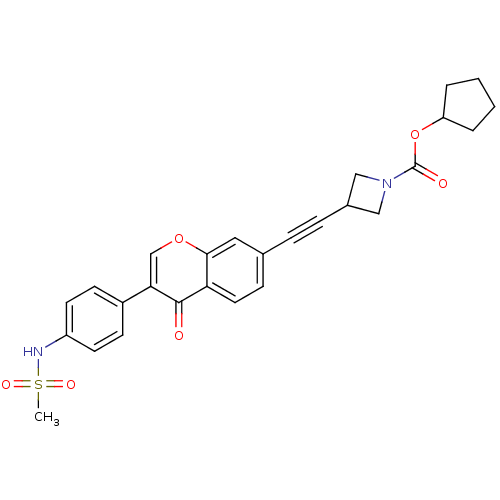
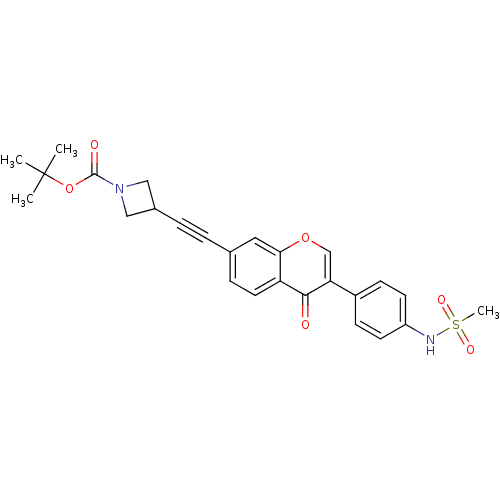
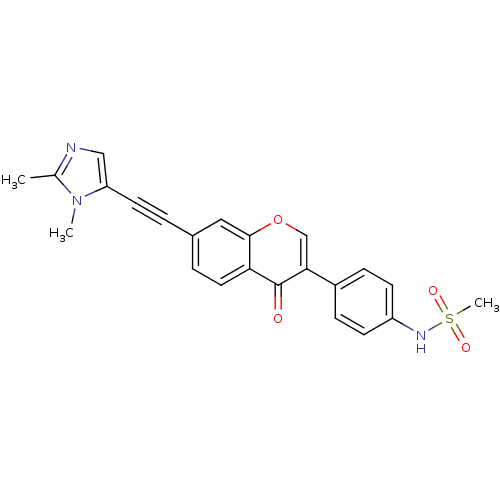
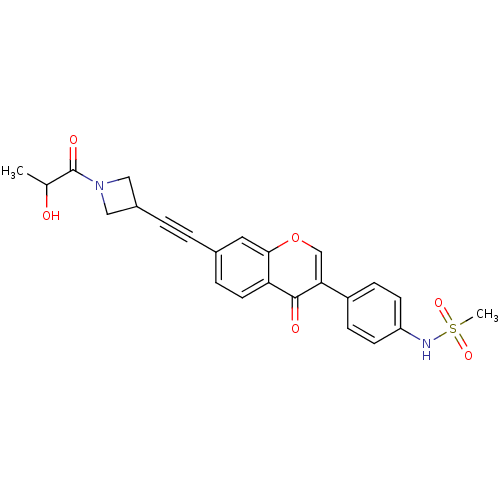
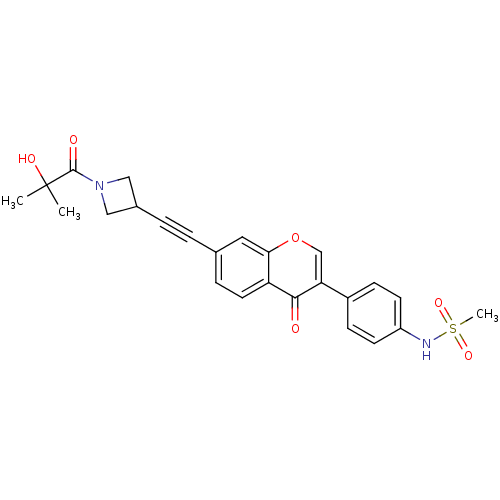
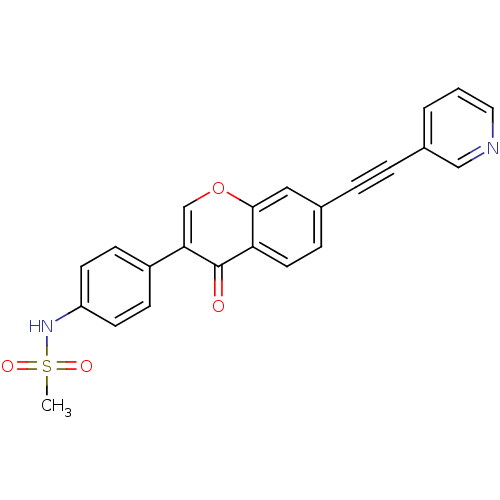
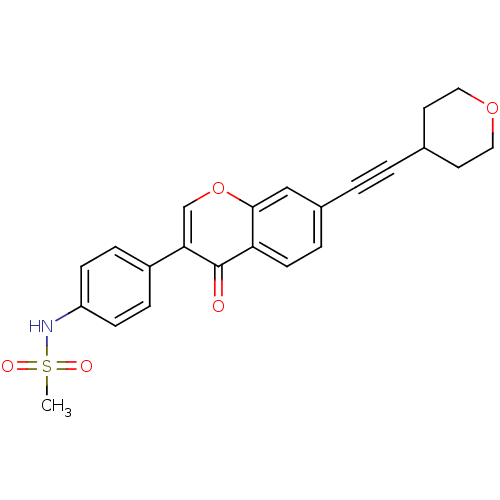
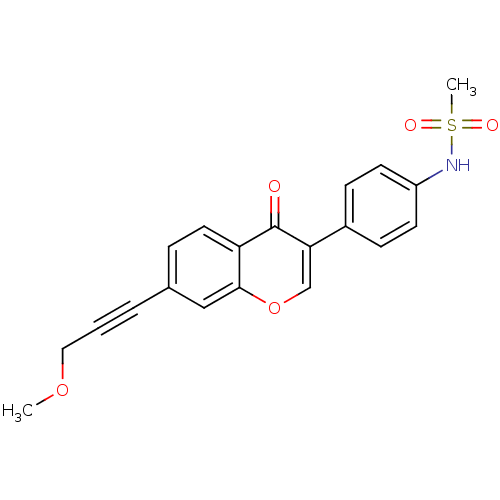
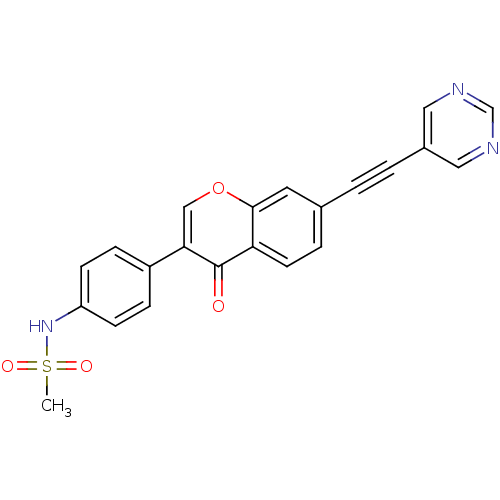
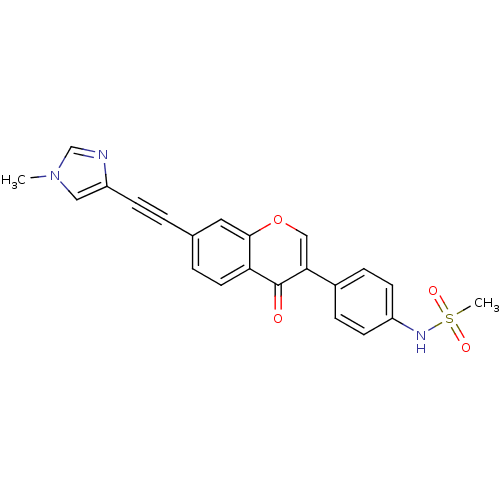
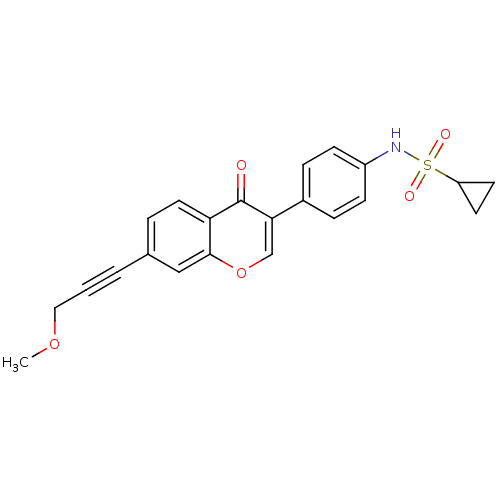
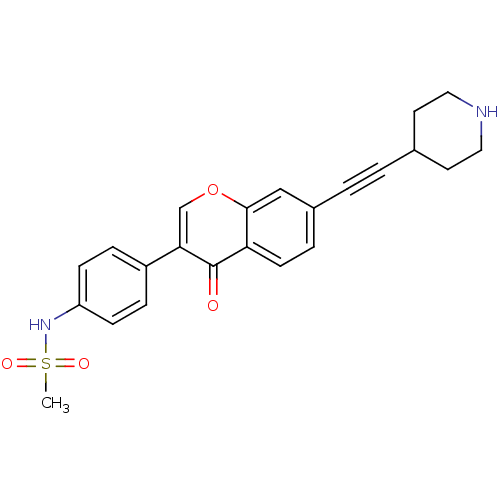
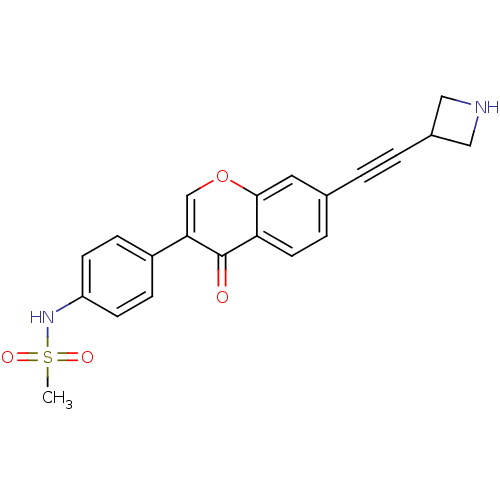
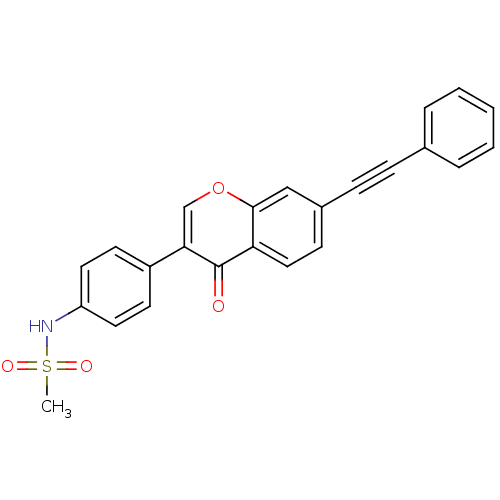
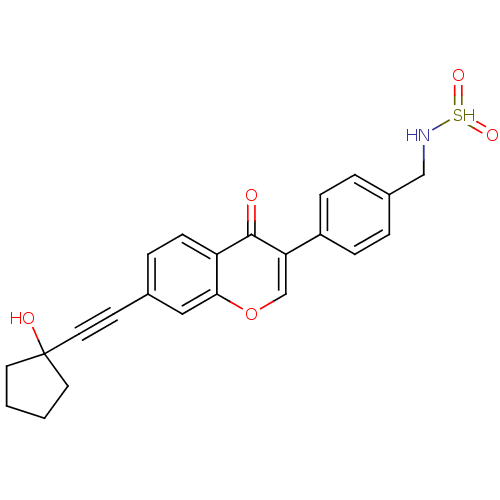
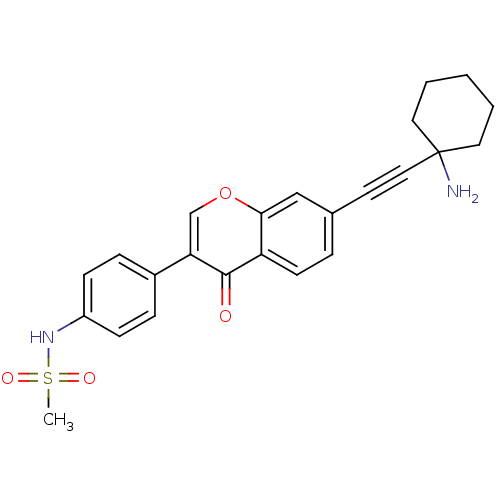
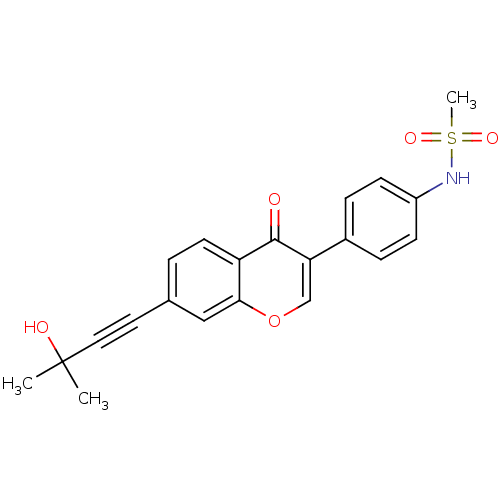
Report error Found 123 Enz. Inhib. hit(s) with all data for entry = 6228
Affinity DataIC50: 2.30nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 4.90nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.5nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 35nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 43nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 47nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 57nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 57.7nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 64nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 86nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 90nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 95nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 96nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 109nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 156nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 174nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 266nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 329nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 365nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 386nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 528nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.71E+3nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.18E+3nMpH: 7.4Assay Description:Standard ALDH2 reaction mixtures contained 150 uM formaldehyde, 2.5 mM NAD+, 10 mM MgCl2 and 10 nM recombinant human ALDH2 in 50 mM Hepes buffer, pH ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 4.69E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 8.65E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 9.10E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair
Affinity DataIC50: 9.10E+4nMpH: 7.5Assay Description:MAO assays included luminogenic MAO substrate, reaction buffers, Luciferin Detection and the reconstitution buffer with esterase. Standard MAO reacti...More data for this Ligand-Target Pair