Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Proteasome subunit beta type-5
Ligand
BDBM50185621
Substrate
n/a
Meas. Tech.
ChEMBL_361596 (CHEMBL868170)
Ki
34000±n/a nM
Citation
 Basse, N; Papapostolou, D; Pagano, M; Reboud-Ravaux, M; Bernard, E; Felten, AS; Vanderesse, R Development of lipopeptides for inhibiting 20S proteasomes. Bioorg Med Chem Lett 16:3277-81 (2006) [PubMed] Article
Basse, N; Papapostolou, D; Pagano, M; Reboud-Ravaux, M; Bernard, E; Felten, AS; Vanderesse, R Development of lipopeptides for inhibiting 20S proteasomes. Bioorg Med Chem Lett 16:3277-81 (2006) [PubMed] Article More Info.:
Target
Name:
Proteasome subunit beta type-5
Synonyms:
DOA3 | PRE2 | PRG1 | PSB5_YEAST | Proteasome Macropain subunit PRE2
Type:
PROTEIN
Mol. Mass.:
31634.72
Organism:
Saccharomyces cerevisiae
Description:
ChEMBL_946840
Residue:
287
Sequence:
MQAIADSFSVPNRLVKELQYDNEQNLESDFVTGASQFQRLAPSLTVPPIASPQQFLRAHTDDSRNPDCKIKIAHGTTTLAFRFQGGIIVAVDSRATAGNWVASQTVKKVIEINPFLLGTMAGGAADCQFWETWLGSQCRLHELREKERISVAAASKILSNLVYQYKGAGLSMGTMICGYTRKEGPTIYYVDSDGTRLKGDIFCVGSGQTFAYGVLDSNYKWDLSVEDALYLGKRSILAAAHRDAYSGGSVNLYHVTEDGWIYHGNHDVGELFWKVKEEEGSFNNVIG
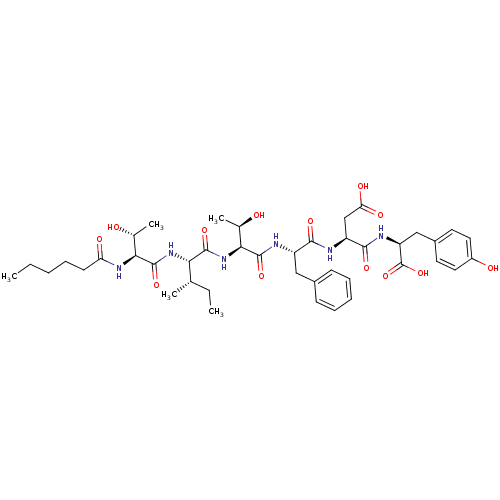
Inhibitor
Name:
BDBM50185621
Synonyms:
(S)-4-((S)-1-carboxy-2-(4-hydroxyphenyl)ethylamino)-3-((S)-2-((2S,3R)-2-((2S,3S)-2-((2S,3R)-2-hexanamido-3-hydroxybutanamido)-3-methylpentanamido)-3-hydroxybutanamido)-3-phenylpropanamido)-4-oxobutanoic acid | CHEMBL425955
Type:
Small organic molecule
Emp. Form.:
C42H60N6O13
Mol. Mass.:
856.9582
SMILES:
CCCCCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O