Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Indoleamine 2,3-dioxygenase 1
Ligand
BDBM50538502
Substrate
n/a
Meas. Tech.
ChEMBL_1972941 (CHEMBL4605759)
IC50
32±n/a nM
Citation
 White, C; McGowan, MA; Zhou, H; Sciammetta, N; Fradera, X; Lim, J; Joshi, EM; Andrews, C; Nickbarg, EB; Cowley, P; Trewick, S; Augustin, M; von K�enig, K; Lesburg, CA; Otte, K; Knemeyer, I; Woo, H; Yu, W; Cheng, M; Spacciapoli, P; Geda, P; Song, X; Smotrov, N; Curran, P; Heo, MR; Abeywickrema, P; Miller, JR; Bennett, DJ; Han, Y Strategic Incorporation of Polarity in Heme-Displacing Inhibitors of Indoleamine-2,3-dioxygenase-1 (IDO1). ACS Med Chem Lett 11:550-557 (2020) [PubMed] Article
White, C; McGowan, MA; Zhou, H; Sciammetta, N; Fradera, X; Lim, J; Joshi, EM; Andrews, C; Nickbarg, EB; Cowley, P; Trewick, S; Augustin, M; von K�enig, K; Lesburg, CA; Otte, K; Knemeyer, I; Woo, H; Yu, W; Cheng, M; Spacciapoli, P; Geda, P; Song, X; Smotrov, N; Curran, P; Heo, MR; Abeywickrema, P; Miller, JR; Bennett, DJ; Han, Y Strategic Incorporation of Polarity in Heme-Displacing Inhibitors of Indoleamine-2,3-dioxygenase-1 (IDO1). ACS Med Chem Lett 11:550-557 (2020) [PubMed] Article More Info.:
Target
Name:
Indoleamine 2,3-dioxygenase 1
Synonyms:
I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase
Type:
Enzyme
Mol. Mass.:
45330.80
Organism:
Homo sapiens (Human)
Description:
P14902
Residue:
403
Sequence:
MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVEKLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLELPPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKVIPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGNPQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMPPAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQQPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
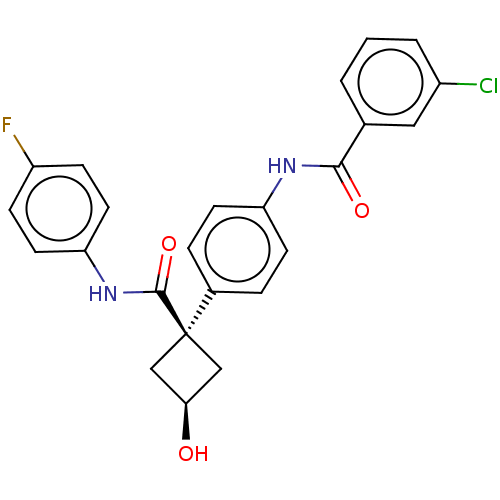
Inhibitor
Name:
BDBM50538502
Synonyms:
CHEMBL4643572
Type:
Small organic molecule
Emp. Form.:
C24H20ClFN2O3
Mol. Mass.:
438.879
SMILES:
O[C@H]1C[C@](C1)(C(=O)Nc1ccc(F)cc1)c1ccc(NC(=O)c2cccc(Cl)c2)cc1 |r,wD:3.5,1.0,(43.66,-3.66,;42.89,-5,;41.41,-5.4,;41.81,-6.89,;43.3,-6.49,;42.55,-8.24,;41.75,-9.56,;44.09,-8.28,;44.83,-9.63,;44.03,-10.94,;44.77,-12.29,;46.31,-12.32,;47.05,-13.67,;47.11,-11,;46.36,-9.65,;40.27,-6.86,;39.51,-5.51,;37.97,-5.48,;37.18,-6.82,;35.63,-6.8,;34.85,-8.12,;35.6,-9.46,;33.31,-8.1,;32.56,-6.76,;31.02,-6.74,;30.23,-8.06,;30.99,-9.41,;30.21,-10.74,;32.53,-9.42,;37.93,-8.16,;39.47,-8.18,)|