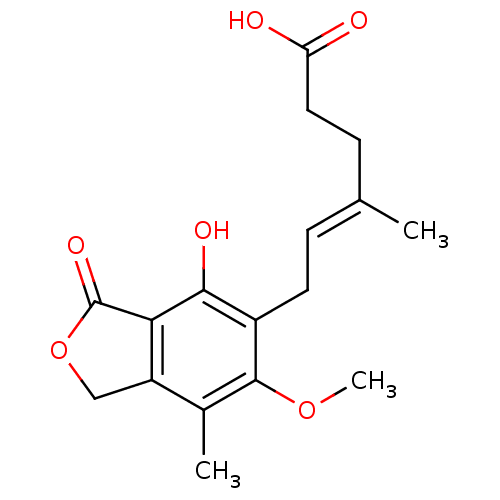
BDBM19264 (4E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3-dihydro-2-benzofuran-5-yl)-4-methylhex-4-enoic acid::(E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1H-2-benzofuran-5-yl)-4-methylhex-4-enoic acid::CHEMBL866::MPA (1)::Mycophenolic Acid (MPA)::Myfortic
SMILES COc1c(C)c2COC(=O)c2c(O)c1C\C=C(/C)CCC(O)=O
InChI Key InChIKey=HPNSFSBZBAHARI-RUDMXATFSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 22 hits for monomerid = 19264
Found 22 hits for monomerid = 19264
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 6nMAssay Description:Inhibitory activity against human Inosine-5'-monophosphate dehydrogenase 2 (IMPDH type II isoform); Range is 6-10 nMMore data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 7nMAssay Description:Inhibition of human IMPDH2 by SpectrophotometerMore data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 7nM ΔG°: -11.1kcal/molepH: 8.0 T: 2°CAssay Description:Inhibition assays were performed in duplicate using two different concentrations of human IMPDH type 2 (33 and 66 nM) and varying concentrations of i...More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Inhibition of human IMPDH 2More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Inhibition of human IMP dehydrogenase 2More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Inhibition of human IMPDH2More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 2(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Inhibitory activity against human Inosine-5'-monophosphate dehydrogenase 2 (IMPDH type II)More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 33nMAssay Description:Inhibitory activity against human Inosine-5'-monophosphate dehydrogenase 1 (IMPDH type I isoform); Range is 33-37 nMMore data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 33nM ΔG°: -10.2kcal/molepH: 8.0 T: 2°CAssay Description:Inhibition assays were performed in duplicate using two different concentrations of human IMPDH type 1 (87 and 155 nM) and varying concentrations of ...More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 33nMAssay Description:Inhibition of human IMPDH1 by SpectrophotometryMore data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibition of human IMPDH1More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibition of human IMPDH 1More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibitory activity against human Inosine-5'-monophosphate dehydrogenase 1 (IMPDH type I)More data for this Ligand-Target Pair
TargetInosine-5'-monophosphate dehydrogenase 1(Homo sapiens (Human))
Codon Pharmaceuticals
Curated by ChEMBL
Codon Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 40nMAssay Description:Inhibition of human IMP dehydrogenase 1More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 1.50E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in diabetic human kidney microsomes assessed as reduction in enzyme-mediated mycophenolic acid phenolic ...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 1.50E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in diabetic human liver microsomes assessed as reduction in enzyme-mediated mycophenolic acid phenolic 7...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 1.50E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in non-diabetic human liver microsomes assessed as reduction in enzyme-mediated mycophenolic acid phenol...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 1.50E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in non-diabetic human kidney microsomes assessed as reduction in enzyme-mediated mycophenolic acid pheno...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in non-diabetic human liver microsomes assessed as reduction in enzyme-mediated mycophenolic acid acyl g...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in non-diabetic human kidney microsomes assessed as reduction in enzyme-mediated mycophenolic acid acyl ...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in diabetic human liver microsomes assessed as reduction in enzyme-mediated mycophenolic acid acyl glucu...More data for this Ligand-Target Pair
TargetUDP-glucuronosyltransferase 2B7(Homo sapiens (Human))
University Of Rhode Island
Curated by ChEMBL
University Of Rhode Island
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:Uncompetitive substrate inhibition of UGT2B7 in diabetic human kidney microsomes assessed as reduction in enzyme-mediated mycophenolic acid acyl gluc...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)