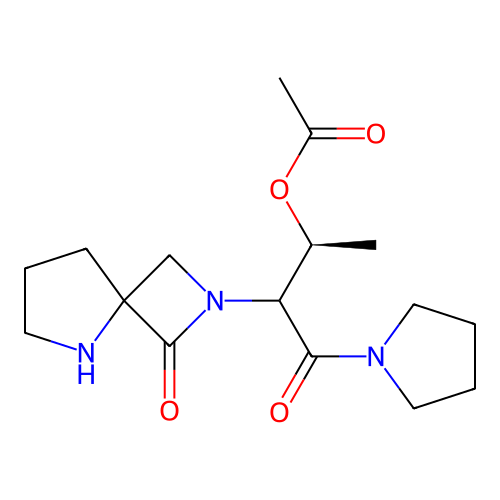
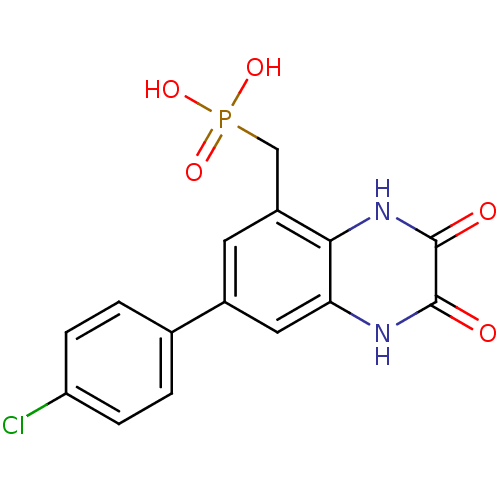
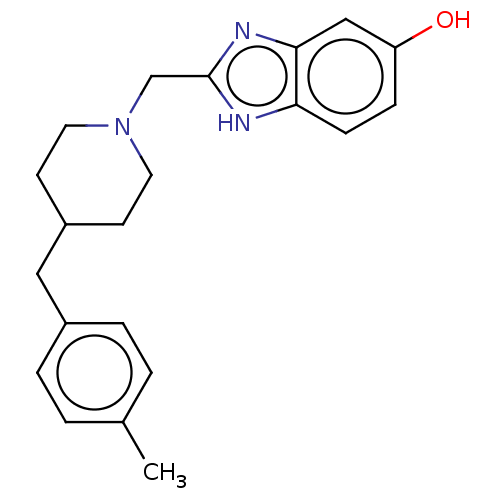
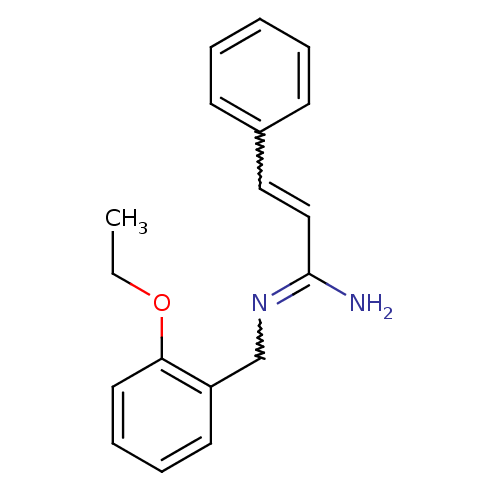
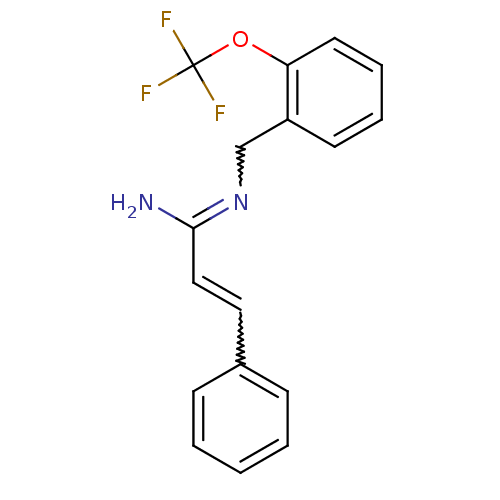
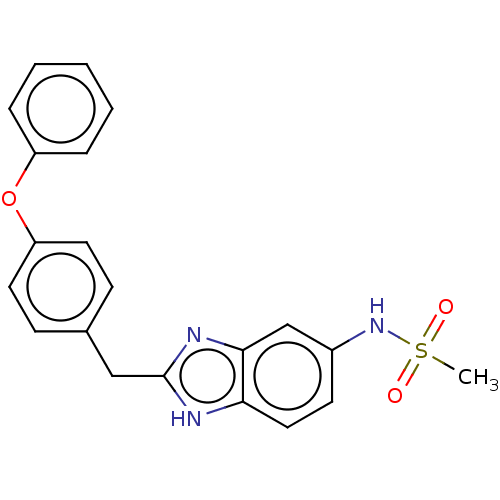
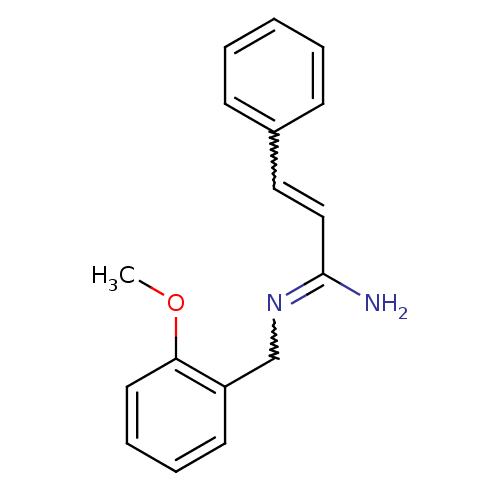
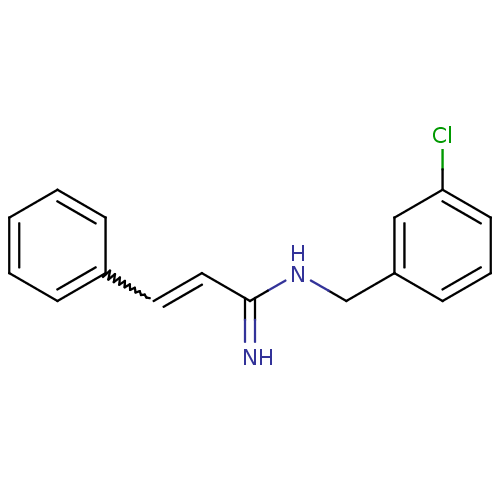
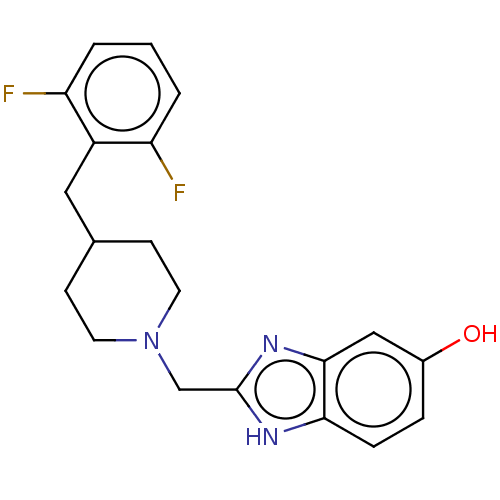
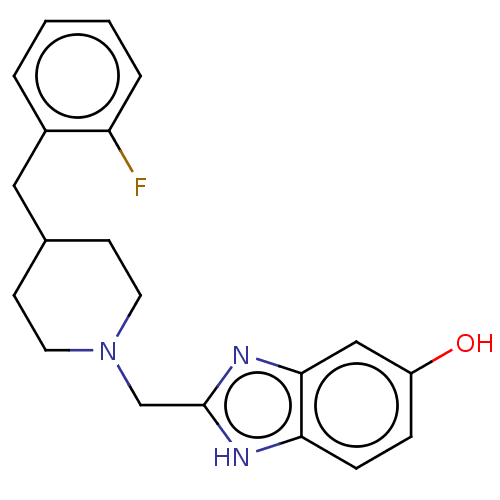
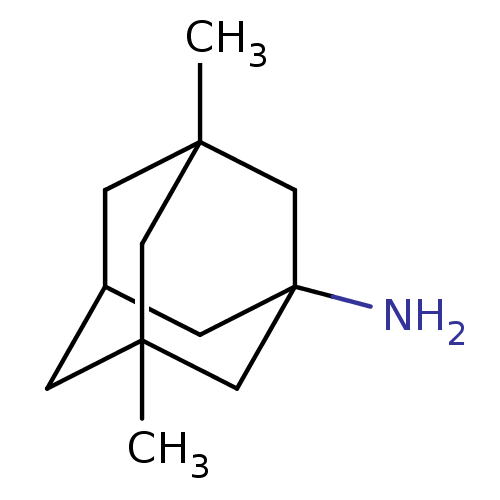
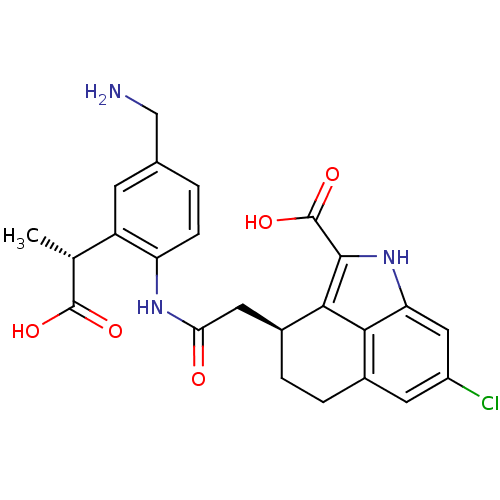
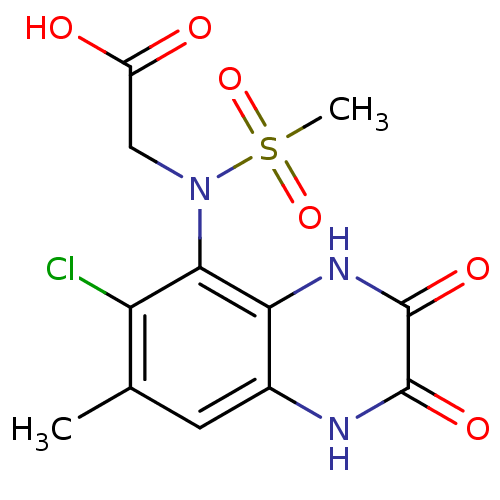
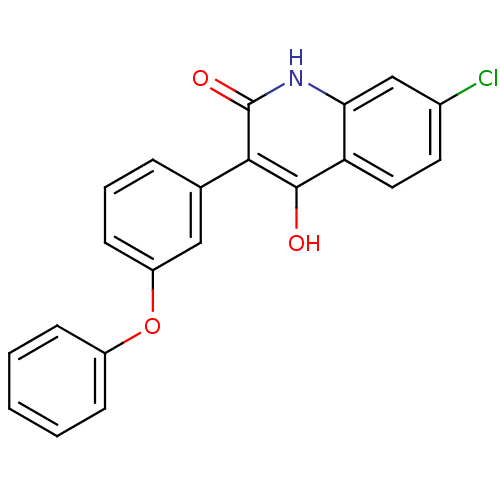
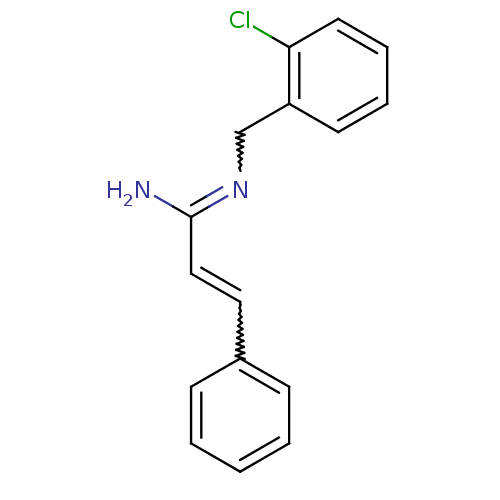
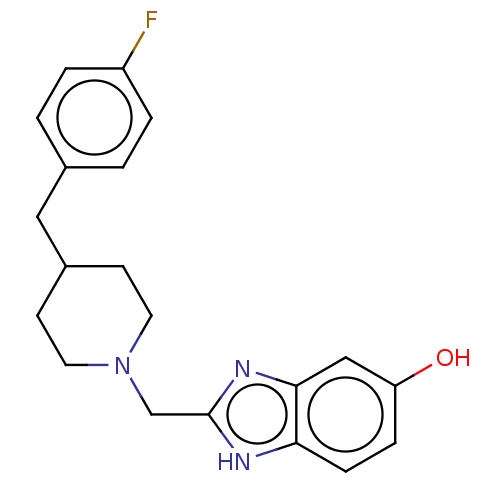
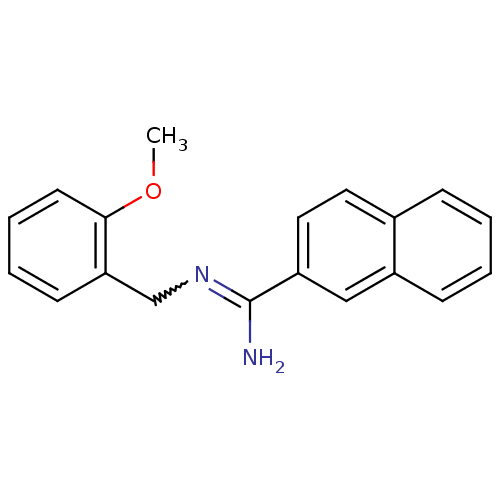
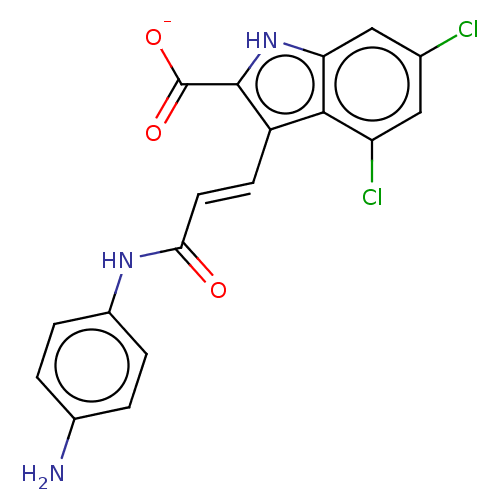
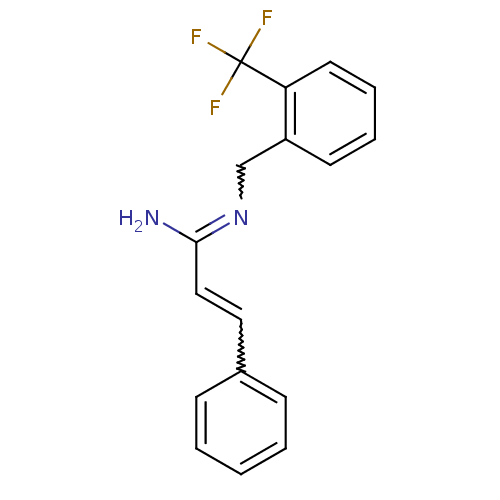
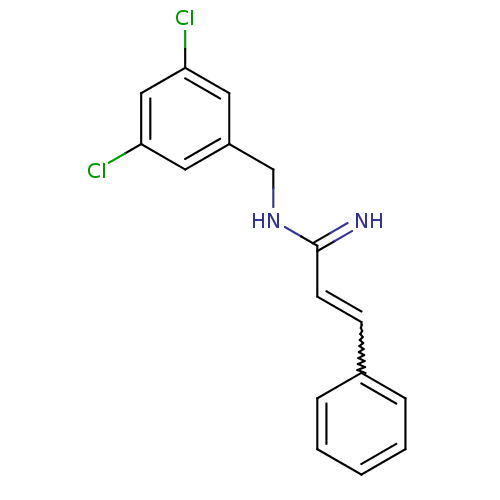
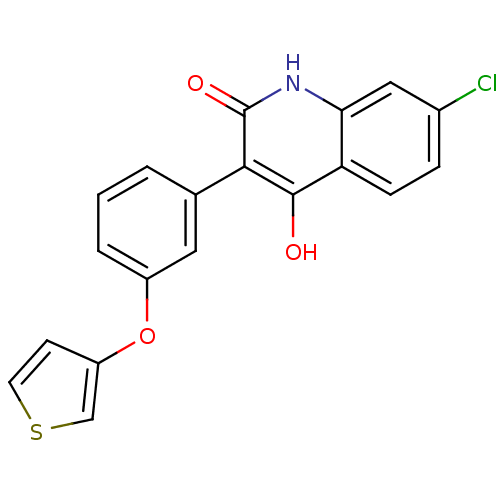
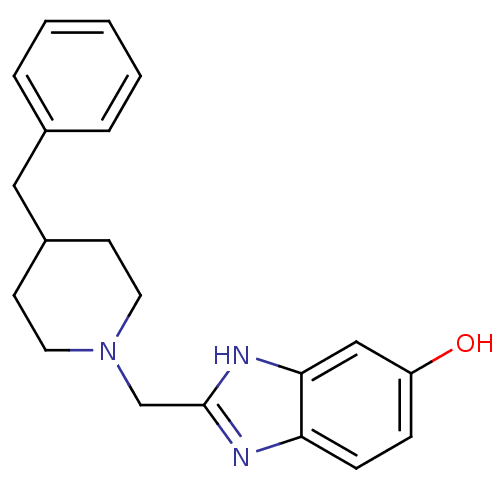
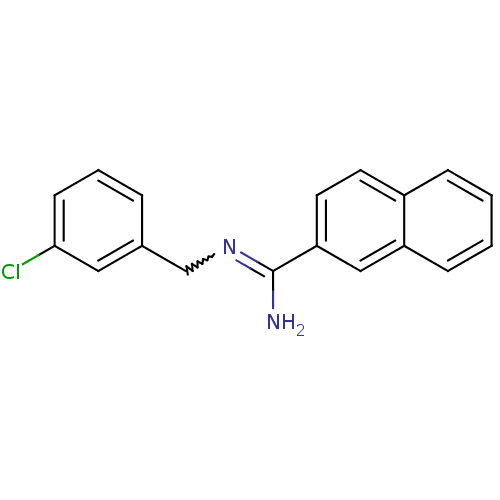
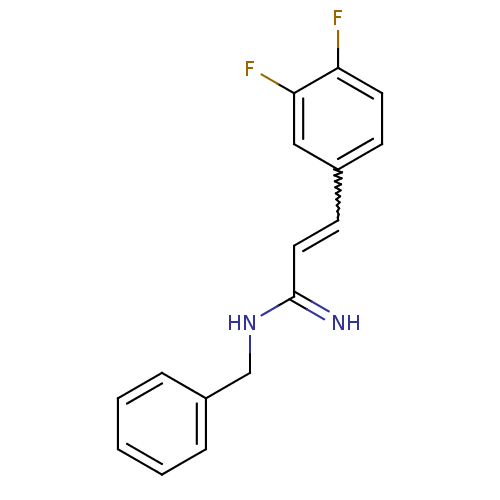
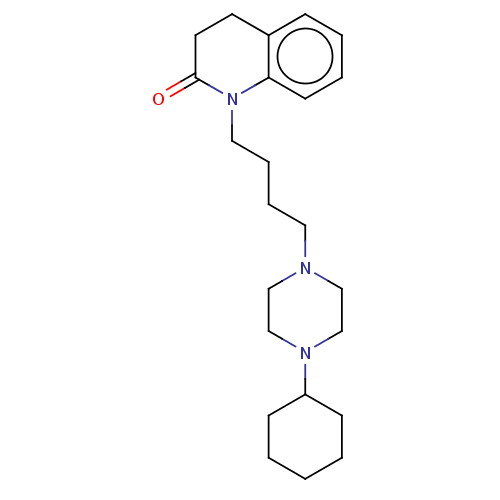
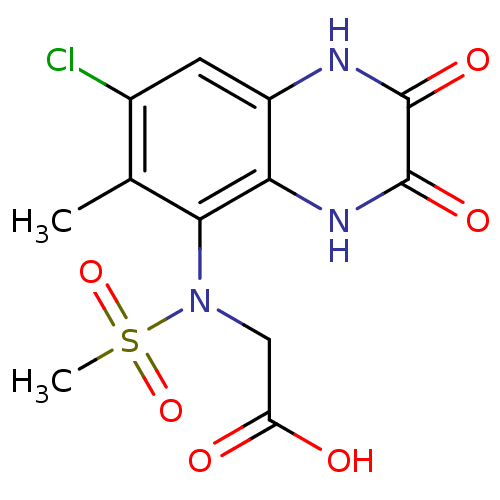
Report error Found 3666 Enz. Inhib. hit(s) with Target = 'Glutamate receptor ionotropic, NMDA 1'
Affinity DataEC50: 0.000349nMAssay Description:Assays were conducted as described in Moskal et al. (Moskal, J. R., Kuo, A. G., Weiss, C., Wood, P. L., O'Connor Hanson, A., Kelso, S., Harris, R...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Inhibitory activity against Xenopus laevis oocyte expressing 1A/2A heteromeric human NMDA (hNMDA) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.400nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.600nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.680nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:Inhibitory activity against Xenopus laevis oocyte expressing 1A/2B heteromeric human NMDA (hNMDA) receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 0.720nMAssay Description:In vitro inhibition of Glu/Gly stimulated [Ca2+] influx in LtK-cells expressing the hNR1a/NR2B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.820nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 0.850nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataIC50: 0.950nMAssay Description:Antagonist activity at recombinant GluN1/GluN2A receptor (unknown origin) expressed in Xenopus laevis oocytes assessed as inhibition of glycine/gluta...More data for this Ligand-Target Pair
Affinity DataKi: 0.990nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 0.990nMAssay Description:Binding affinity to NR1/NR2B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Binding affinity for glycine site of N-methyl-D-aspartate glutamate receptor determined by displacement of [3H]- DCKA (5,7-dichlorokynurenic acid) in...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Antagonist activity at human NMDA NR1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Affinity for glycine site of N-methyl-D-aspartate glutamate receptor determined by displacement of tritium labeled selective glycine antagonist [3H]-...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Inhibition of [3H]glycine to glycine binding site, associated with the N-methyl-D-aspartate glutamate receptor 1 in crude synaptic membranes prepared...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMAssay Description:Inhibitory activity against Xenopus laevis oocyte expressing 1A/2A heteromeric human NMDA (hNMDA) receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Binding affinity to NR1/NR2B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:Inhibition of [3H]glycine to glycine binding site, associated with the N-methyl-D-aspartate glutamate receptor 1 in crude synaptic membranes prepared...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:Antagonist activity at human NR1a/NR2B receptor expressed in mouse fibroblast LMTK cells assessed as inhibition of Glu/Gly induced Ca2+ influxMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Affinity measured by using [3H]5,7-dichlorokynurenic acid (DCKA) for the glycine binding site of NMDA receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:Inhibition of the binding of [3H]L-689,560 ([3H]-4) to the strychnine-insensitive glycine site on rat brain membranesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Affinity for glycine site of N-methyl-D-aspartate glutamate receptor determined by displacement of tritium labeled selective glycine antagonist [3H]-...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Displacement of [3H](E)-N1-(2-methoxybenzyl)cinnamamidine from human NR1a/NR2b receptor expressed in mouse Ltk cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Compound was evaluated for in vitro inhibition of [3H][(E)-N-(2-methoxybenzyl)cinnamamidine binding to human NR1a/NR2B receptors expressed in LtK-cel...More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Inhibition of [3H]glycine to glycine binding site, associated with the N-methyl-D-aspartate glutamate receptor 1 in crude synaptic membranes prepared...More data for this Ligand-Target Pair
Affinity DataKi: 1.70nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:Inhibitory activity against Xenopus laevis oocyte expressing 1A/2B heteromeric human NMDA (hNMDA) receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:In vitro inhibition of Glu/Gly stimulated [Ca2+] influx in LtK-cells expressing the hNR1a/NR2B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Displacement of [3H]ifenprodil binding to recombinant human NR1a/NR2B receptors expressed in L(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Modulation of recombinant human GluN1/GluN2B NMDA receptor expressed in mammalian cells assessed as inhibition of glutamate/glycine-induced intracell...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Binding affinity against NR1A/2B receptor in rat brains by [3H]ifenprodil displacement.More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:In vitro ability to displace [3H]L-689,560 binding to glycine site on the N-methyl-D-aspartate (NMDA) glutamate receptor 1 from rat cortical membrane...More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Displacement of [3H]ifenprodil from GluN1a/GluN2B (unknown origin) expressed in mouse L(tk-) cell membranes after 120 mins by solid scintillation cou...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Compound was evaluated for its ability to displace [3H]-L-689,560 from rat cortical membraneMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Inhibition of [3H]glycine to glycine binding site, associated with the N-methyl-D-aspartate glutamate receptor 1 in crude synaptic membranes prepared...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of NMDA-evoked increased intracellular [Ca2+] in cells expressing NR1/NR2B receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Antagonist activity at human NMDA NR1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Antagonist activity at human NMDA NR1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of the binding of [3H]L-689,560 ([3H]-4) to the strychnine-insensitive glycine site on rat brain membranesMore data for this Ligand-Target Pair