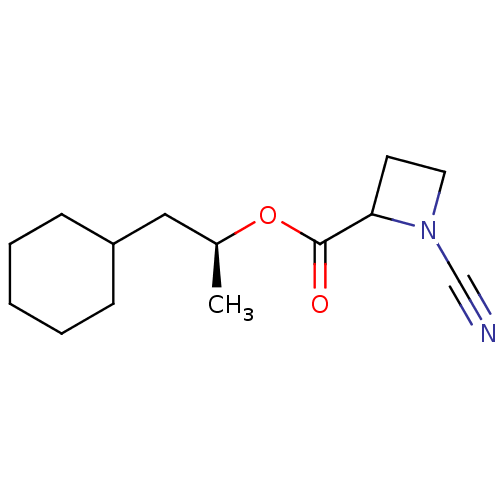
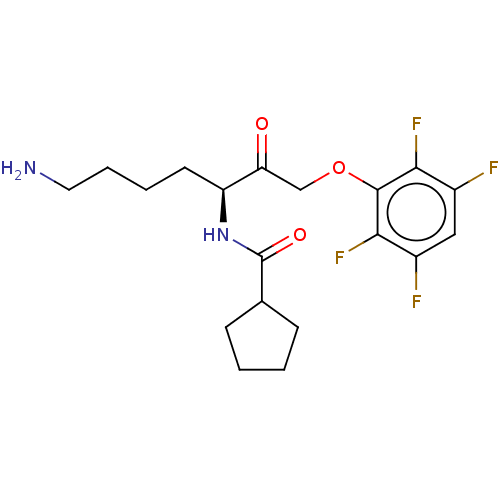
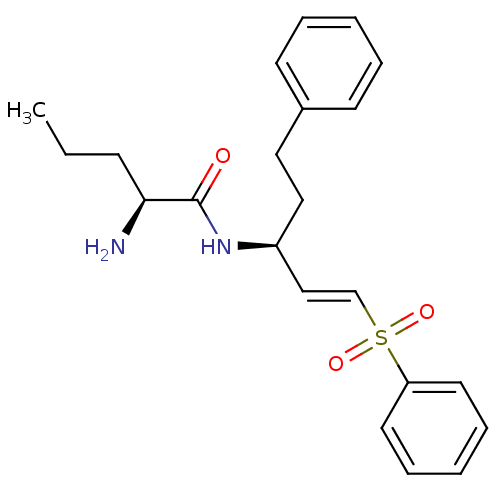
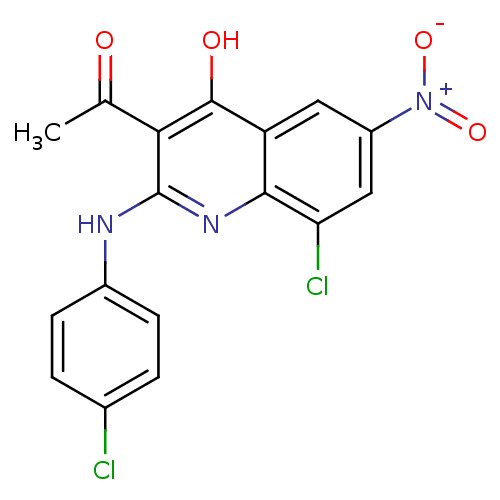
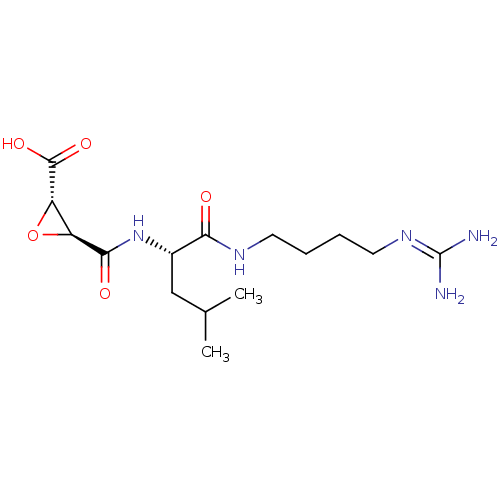
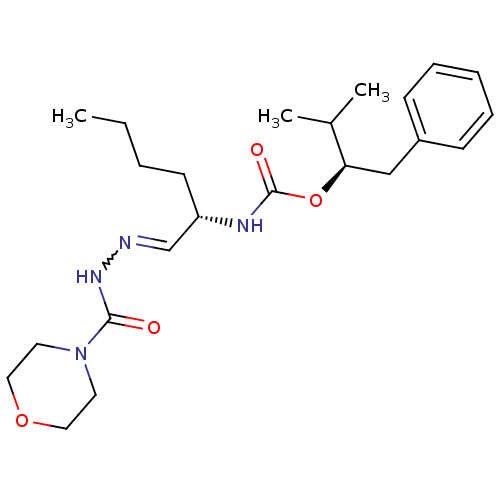
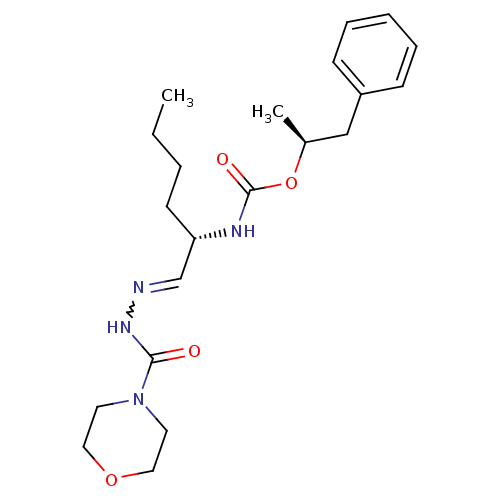
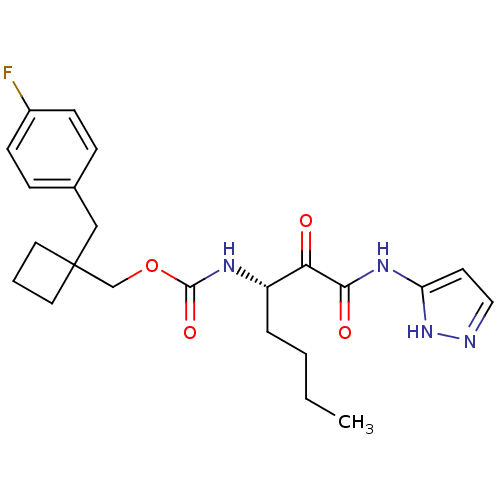
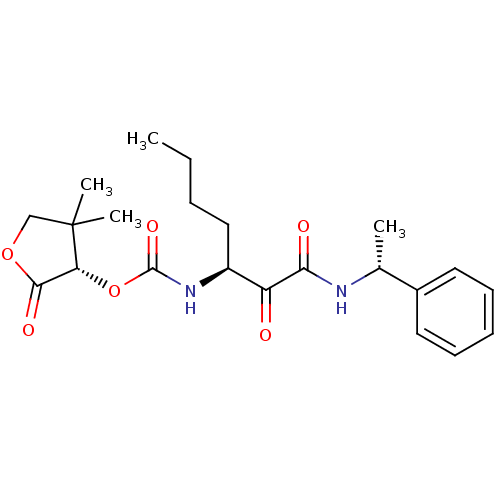
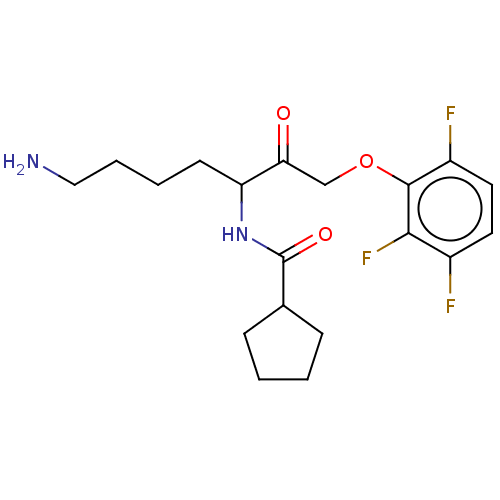
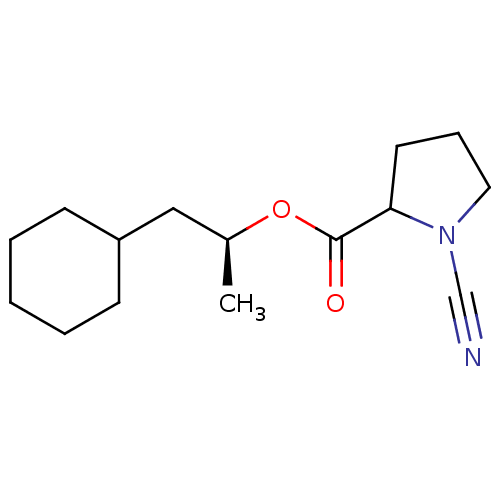
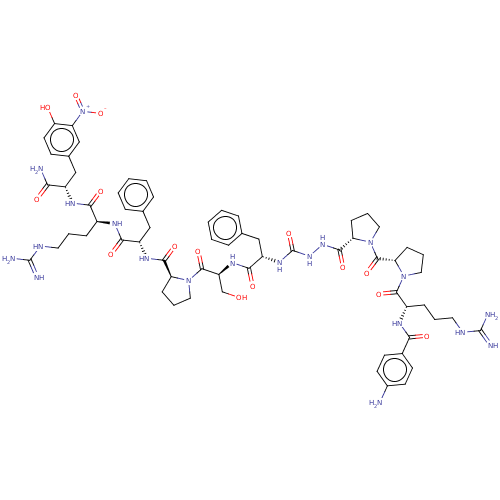
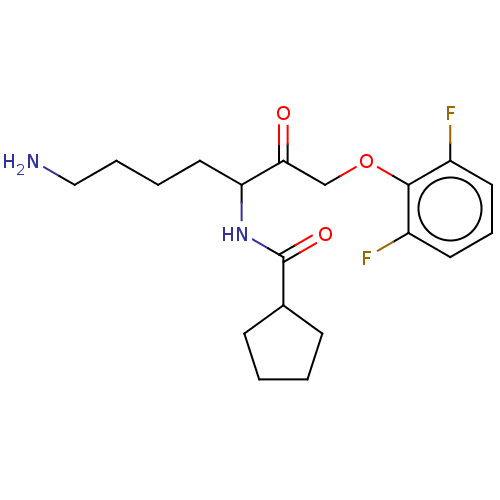
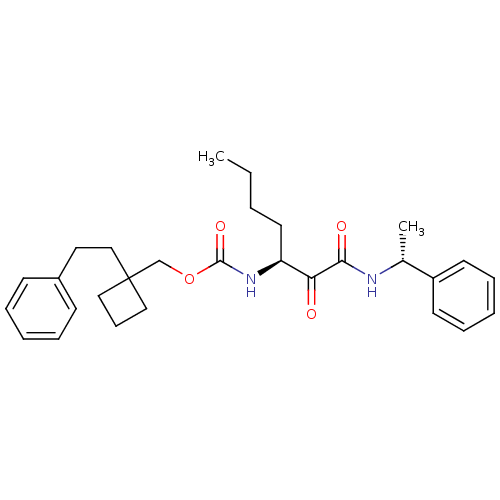
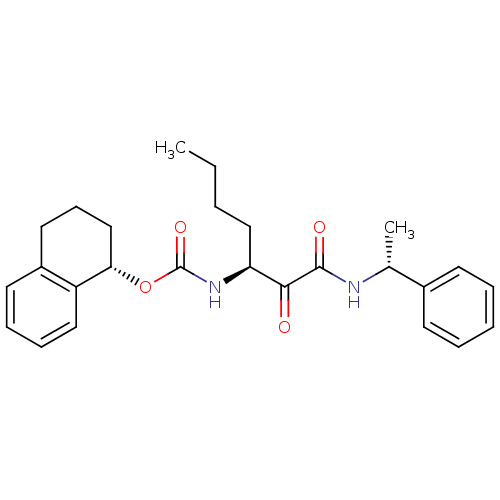
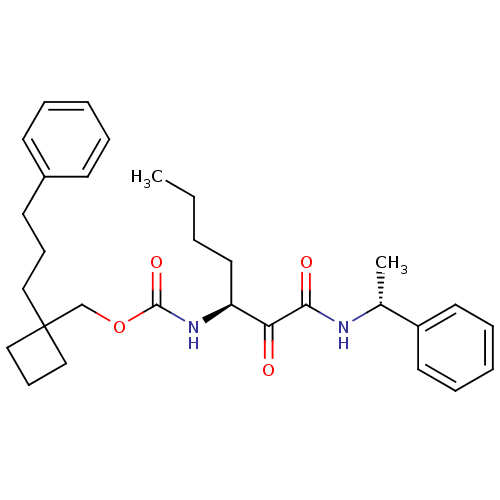
Report error Found 119 Enz. Inhib. hit(s) with Target = 'Pro-cathepsin H'
Affinity DataKi: 0.460nMAssay Description:Inhibition constant against human cathepsin H using L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Inhibition constant against human cathepsin H using L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of cathepsin HMore data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:Inhibition of recombinant human cathepsin H in a fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Inhibition of recombinant human cathepsin H in a fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Inhibitory concentration against human cathepsin H using 50 uM L-Arg-beta-naphthalamideMore data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 110nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataKi: 137nMAssay Description:Inhibition of human recombinant cathepsin H by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 145nMAssay Description:Inhibition of human liver cathepsin H after 30 mins by fluorometric end-point assayMore data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Inhibition of human recombinant cathepsin H after 10 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Inhibition of recombinant human cathepsin H in a fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 230nMAssay Description:Inhibition of recombinant human cathepsin H in a fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibitory concentration against human cathepsin H by fluorescence assay using 50 uM L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
Affinity DataIC50: 560nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 560nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataIC50: 560nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataIC50: 560nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 580nMAssay Description:Inhibitory concentration against human cathepsin H using 50 uM L-Arg-beta-naphthalamideMore data for this Ligand-Target Pair
Affinity DataKi: 1.30E+3nMAssay Description:Reversible competitive inhibition of human cathepsin H using fluorogenic AMC-derived peptide substrate assessed as reduction in residual activity pre...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Inhibition of human recombinant cathepsin H by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of cathepsins B, H, K, L, and S were measured in similar assays. Boc-Leu...More data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibitory concentration against human cathepsin H by fluorescence assay using 50 uM L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.65E+3nMAssay Description:Inhibition of human recombinant cathepsin HMore data for this Ligand-Target Pair
Affinity DataKi: 3.70E+3nMAssay Description:Inhibition of human recombinant cathepsin H by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibitory concentration against human cathepsin H by fluorescence assay using 50 uM L-Arg-b-naphthalamideMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+3nMAssay Description:Inhibition of 50 uM L-Arg-beta-naphthalamide binding to human cathepsin H in fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 6.59E+3nMAssay Description:Inhibition of human recombinant cathepsin HMore data for this Ligand-Target Pair
Affinity DataIC50: 6.60E+3nMpH: 5.5 T: 2°CAssay Description:Protease enzyme inhibition assay targeting diverse member of the cysteine protease families:cathepsin B, Z, and HMore data for this Ligand-Target Pair
Affinity DataKi: 7.00E+3nMAssay Description:Reversible competitive inhibition of human cathepsin H using fluorogenic AMC-derived peptide substrate assessed as reduction in residual activity pre...More data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMpH: 5.5 T: 2°CAssay Description:Protease enzyme inhibition assay targeting diverse member of the cysteine protease families:cathepsin B, Z, and HMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMpH: 5.5 T: 2°CAssay Description:Protease enzyme inhibition assay targeting diverse member of the cysteine protease families:cathepsin B, Z, and HMore data for this Ligand-Target Pair
Affinity DataIC50: 8.30E+3nMAssay Description:Inhibitory concentration against human cathepsin H using 50 uM L-Arg-beta-naphthalamideMore data for this Ligand-Target Pair
Affinity DataKi: 9.20E+3nMAssay Description:Inhibition of human liver cathepsin HMore data for this Ligand-Target Pair
Affinity DataKi: 9.20E+3nMAssay Description:Inhibition of human liver cathepsin HMore data for this Ligand-Target Pair
Affinity DataIC50: 9.80E+3nMAssay Description:Inhibition of human recombinant cathepsin H after 10 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:The capacities of compounds of the present invention to inhibit the activity of lysine gingipain were measured in a fluorogenic assay similar to thos...More data for this Ligand-Target Pair
Ligand InfoSimilars