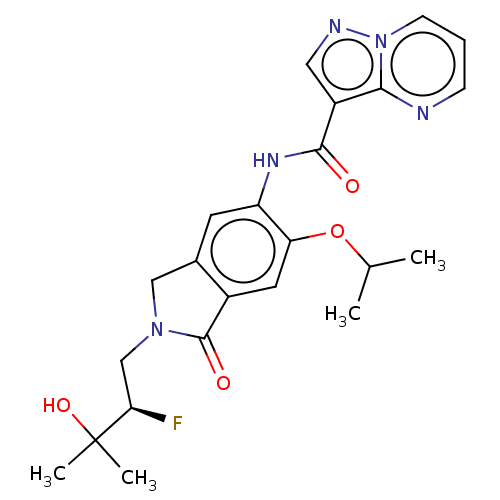
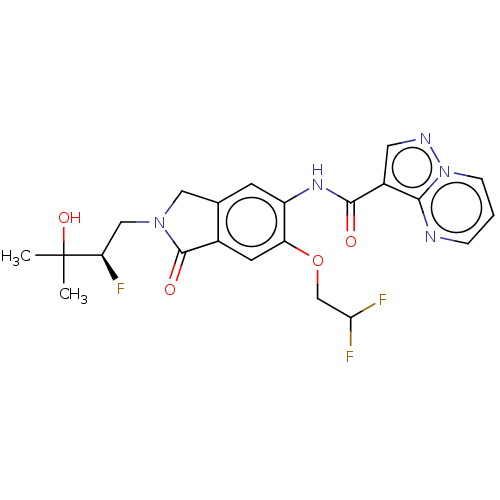
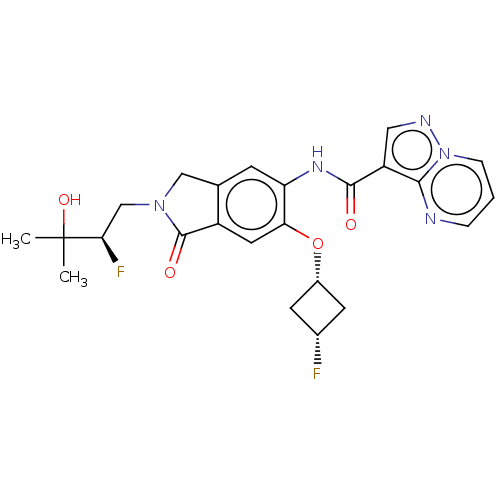
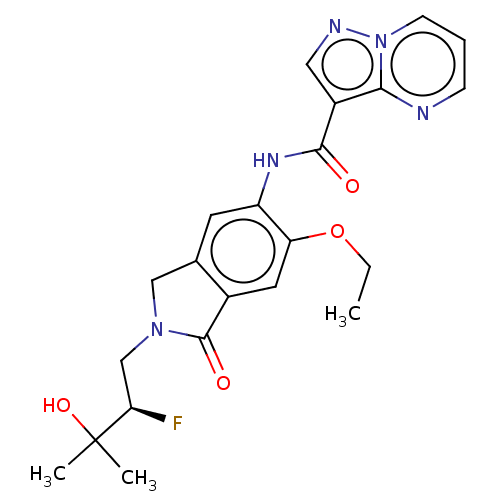
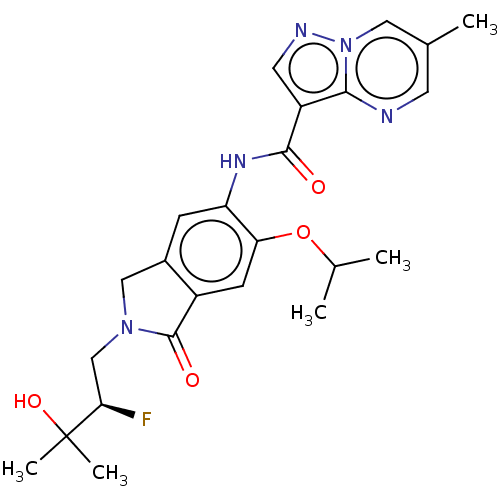
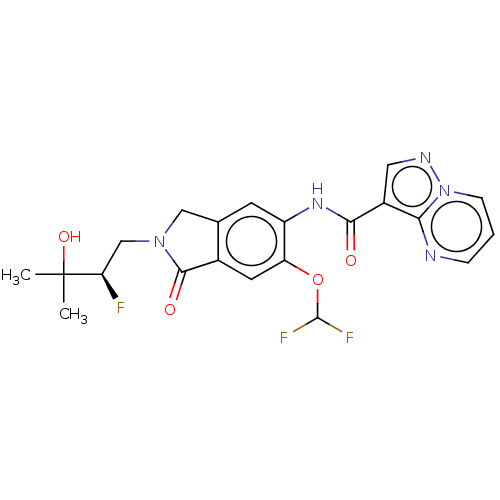
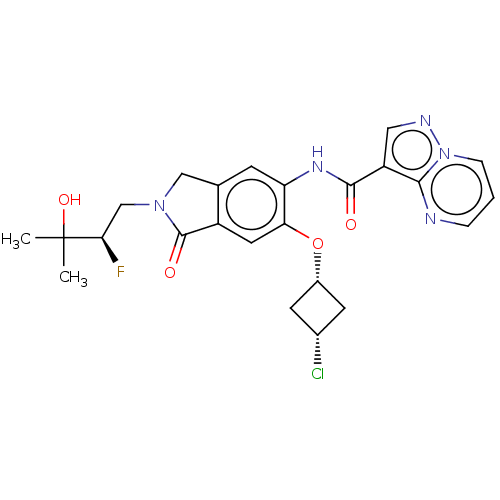
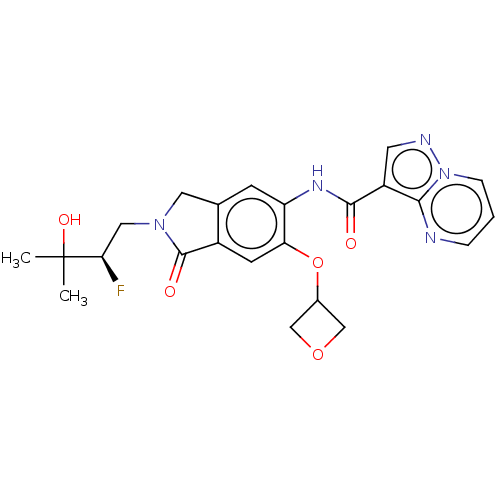
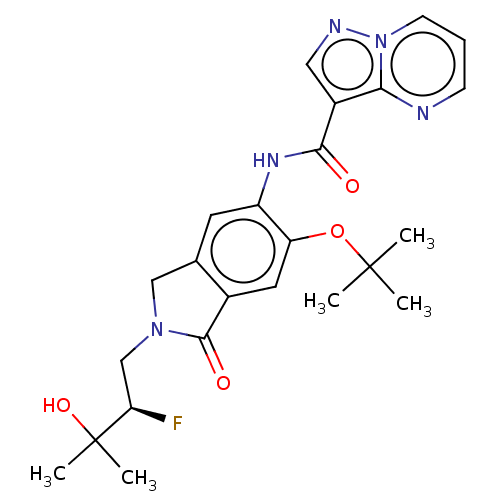
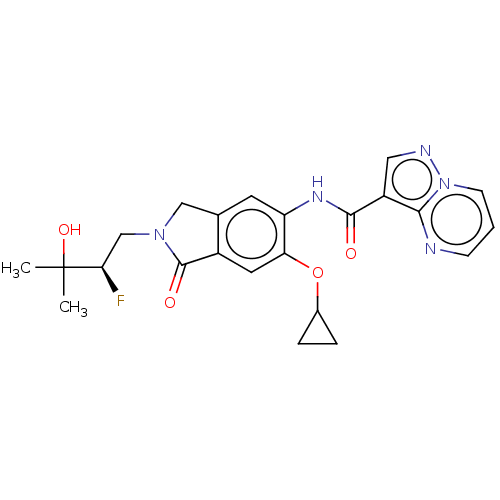
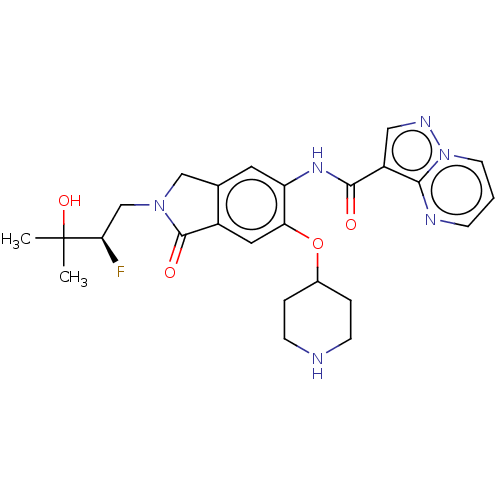
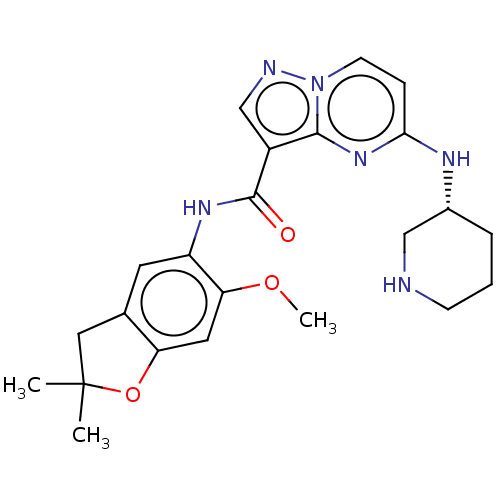
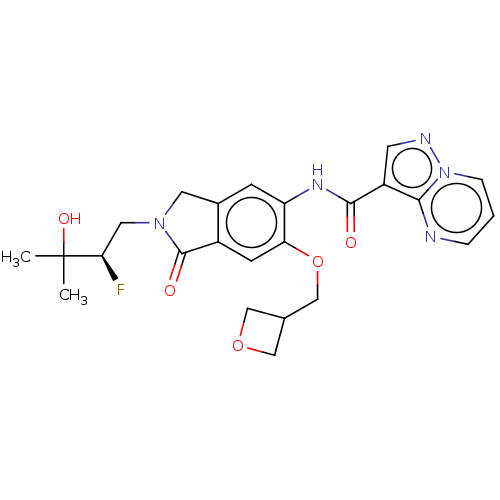
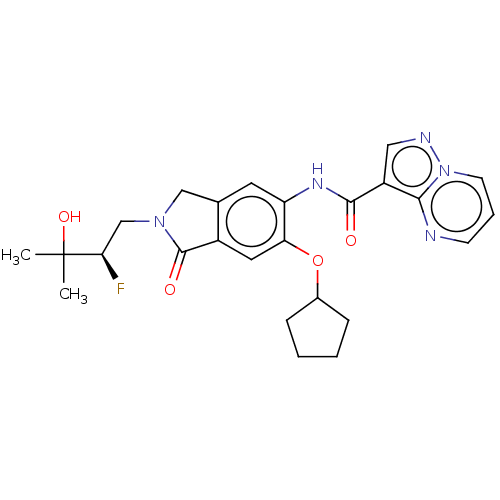
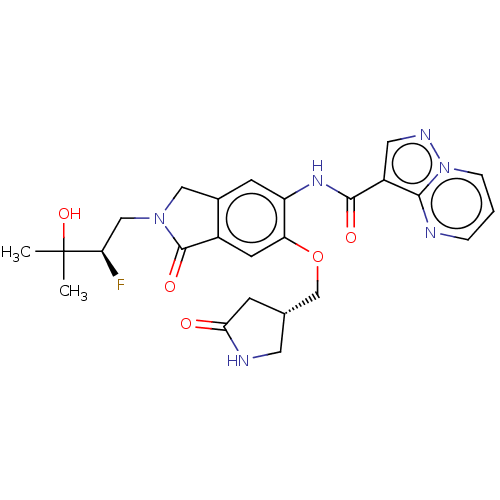
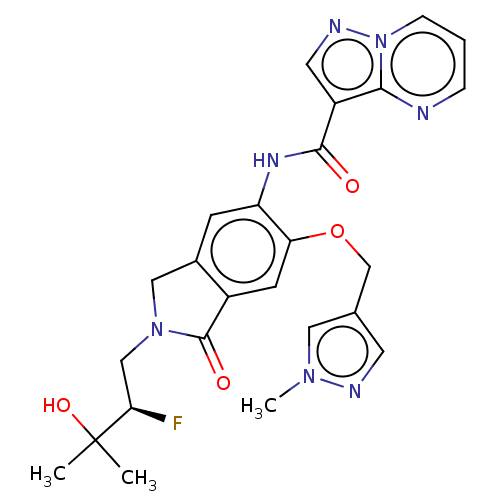
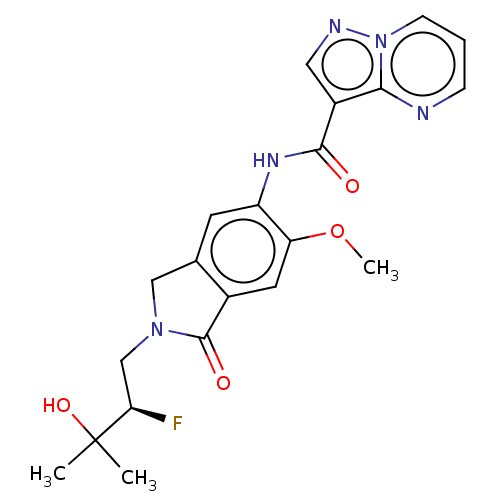
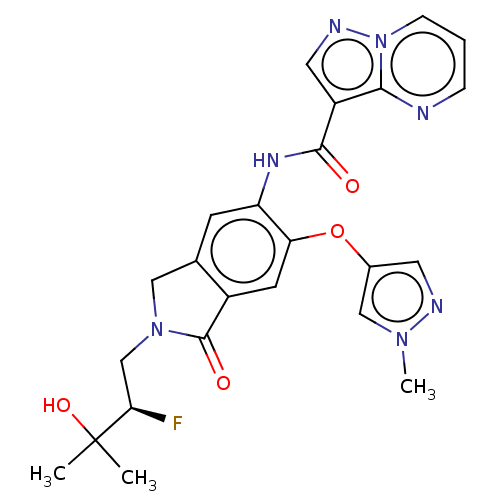
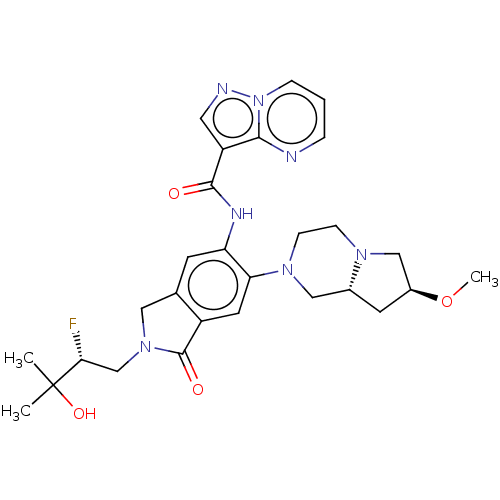
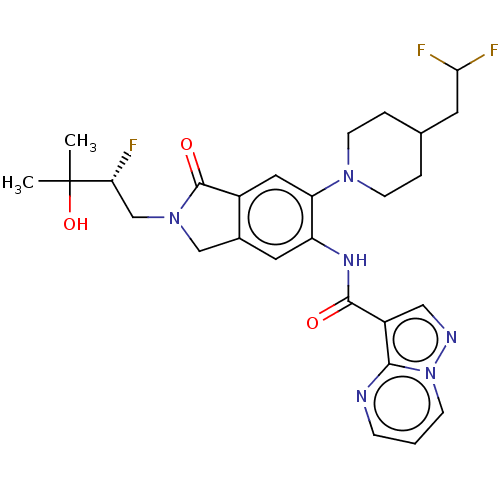
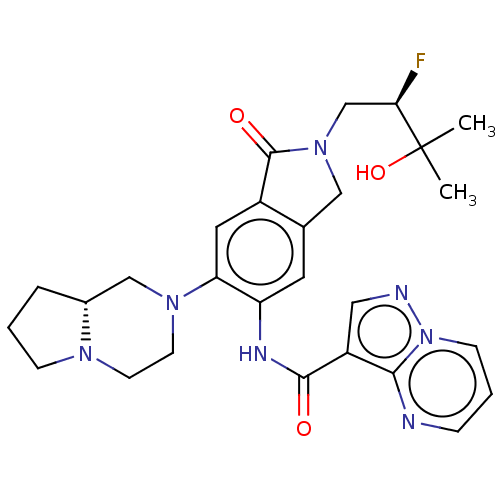
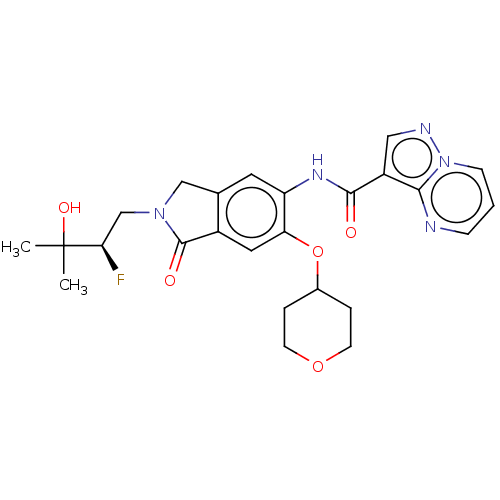
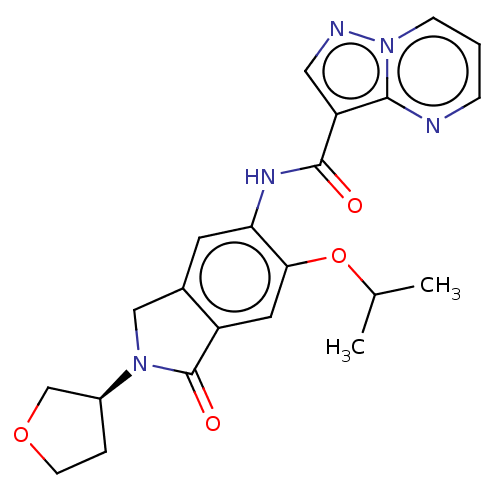
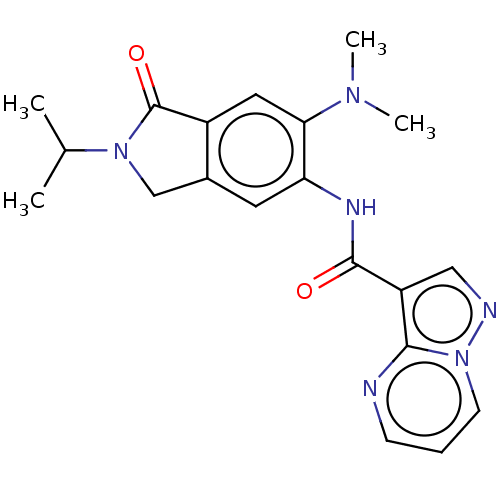
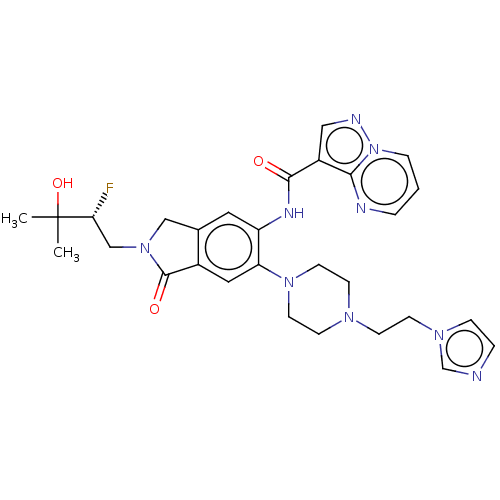
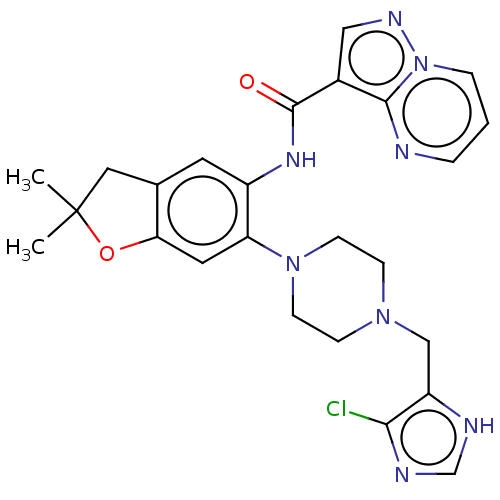
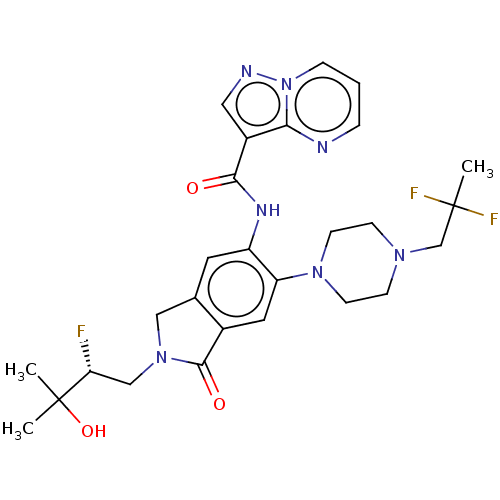
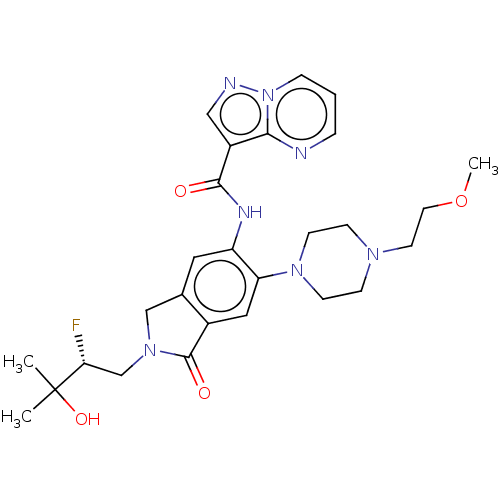
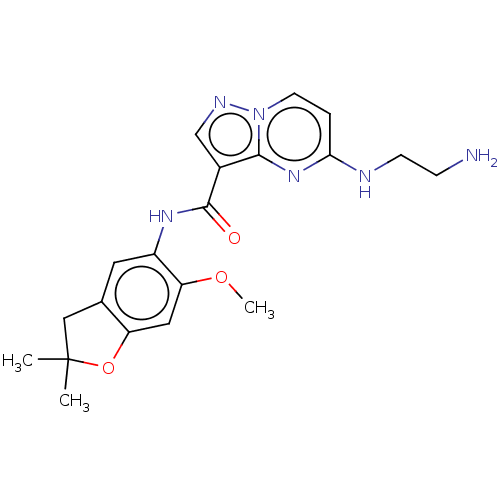
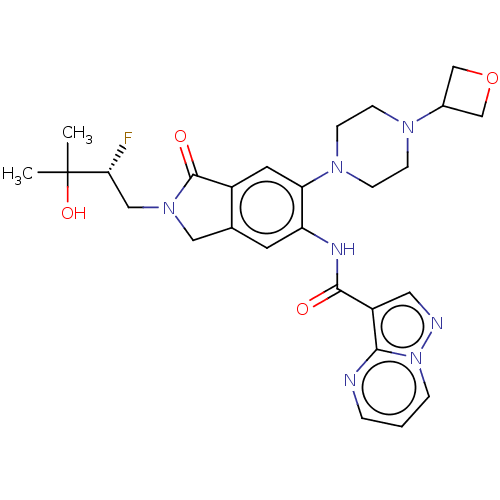
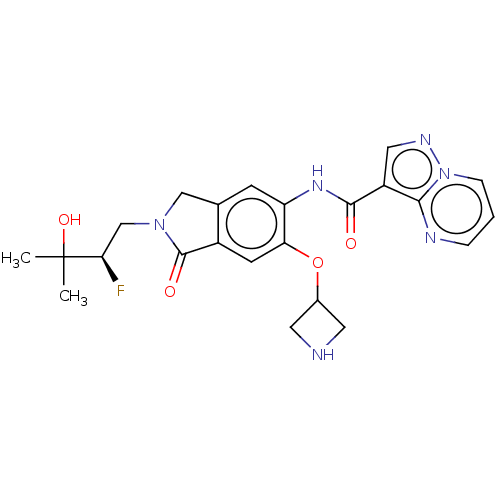
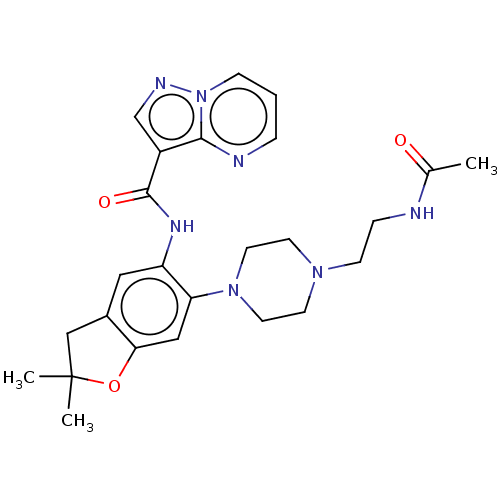
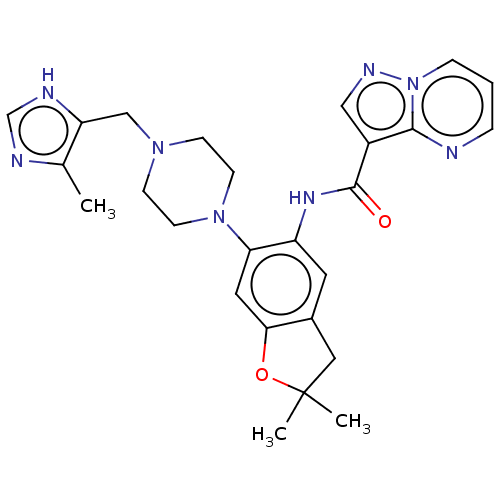
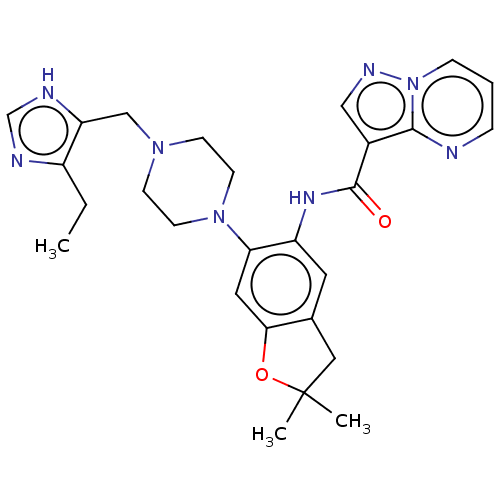
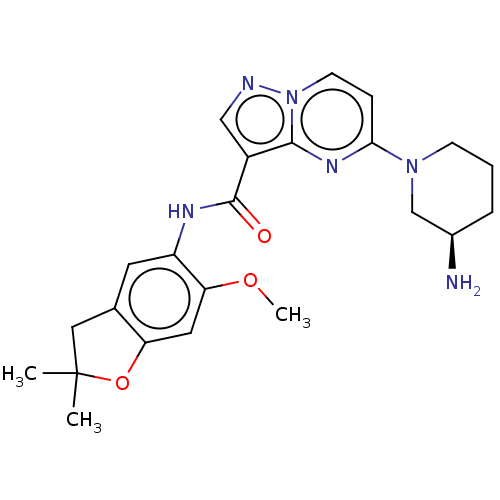
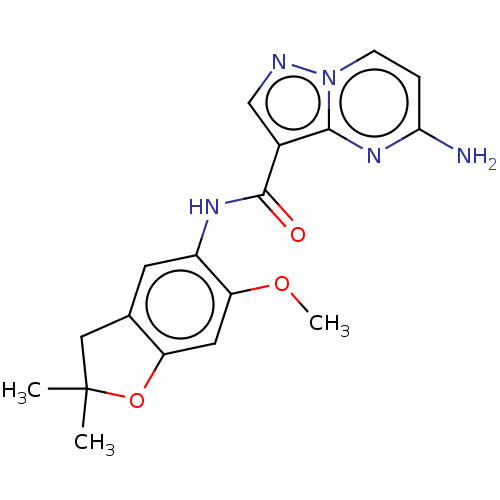
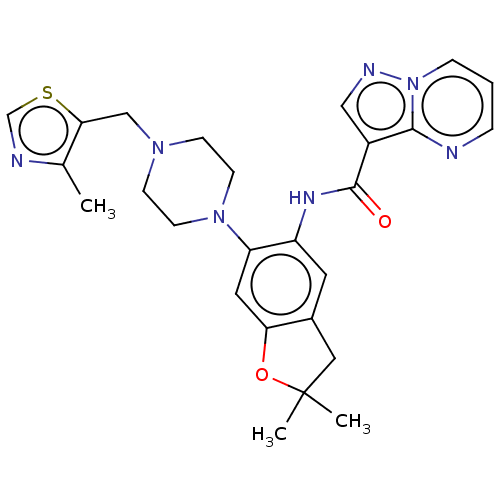
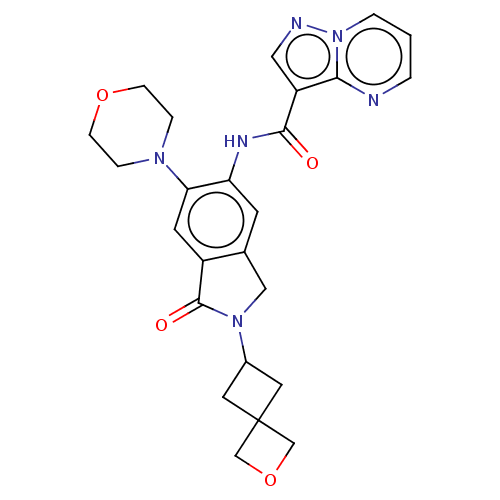
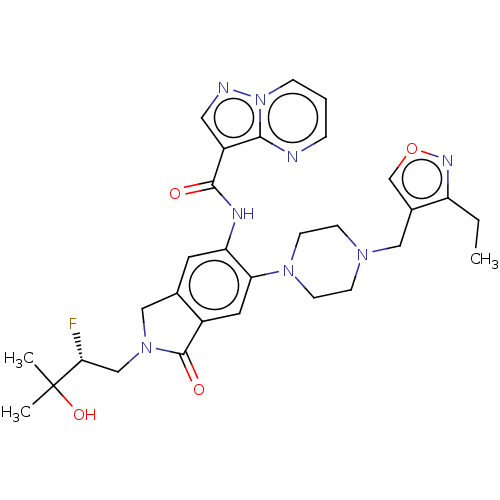
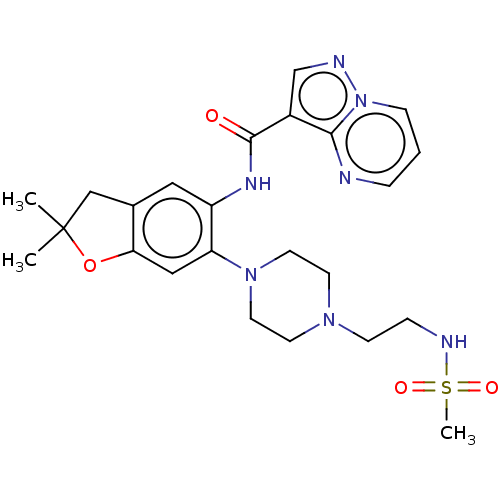
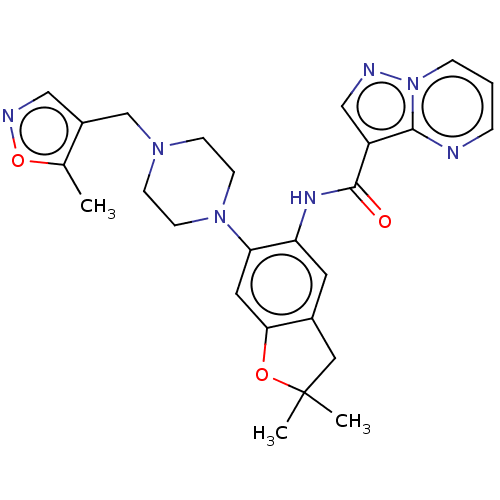
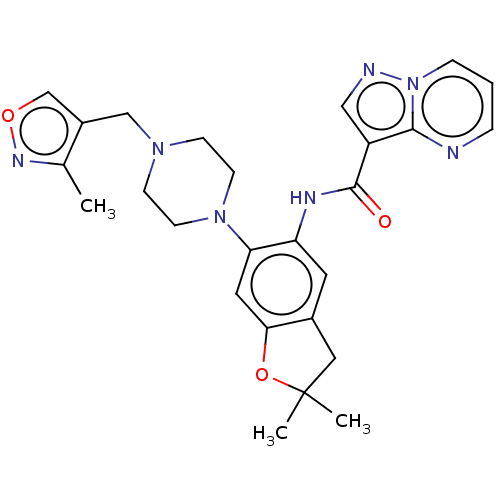
Report error Found 115 Enz. Inhib. hit(s) with all data for entry = 9966
Affinity DataKi: 0.170nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.260nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.280nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.370nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.420nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.490nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.510nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.590nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.610nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.630nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.630nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.670nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.700nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.710nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.870nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.970nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 0.970nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.30nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.80nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 2.10nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 2.30nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 2.5nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 2.60nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.20nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.20nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.40nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.80nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 3.90nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 4.10nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 4.60nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 4.60nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 4.90nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 5.10nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 5.20nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair
Affinity DataKi: 5.20nMAssay Description:The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over...More data for this Ligand-Target Pair