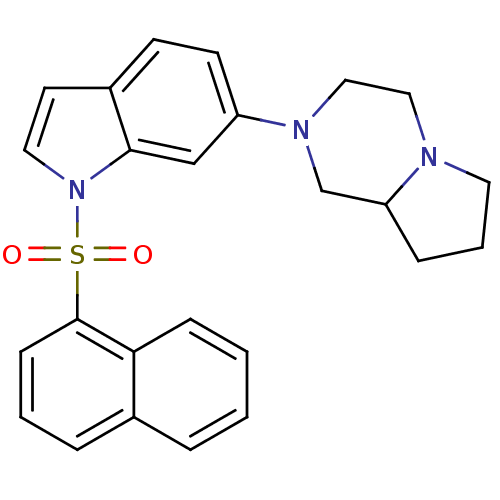
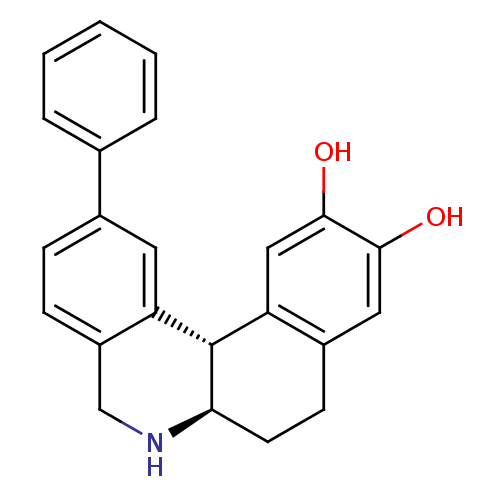
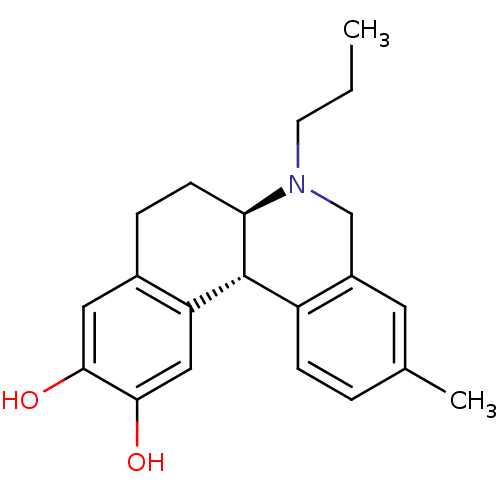
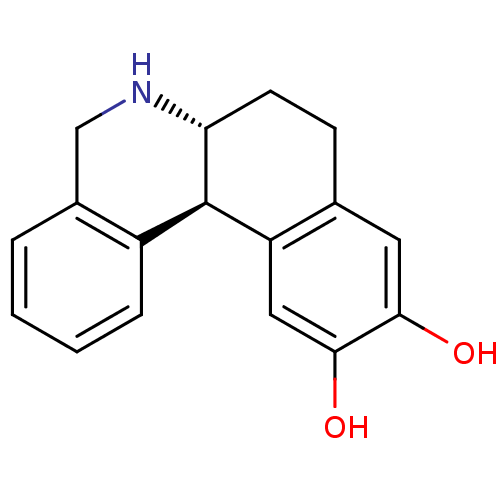
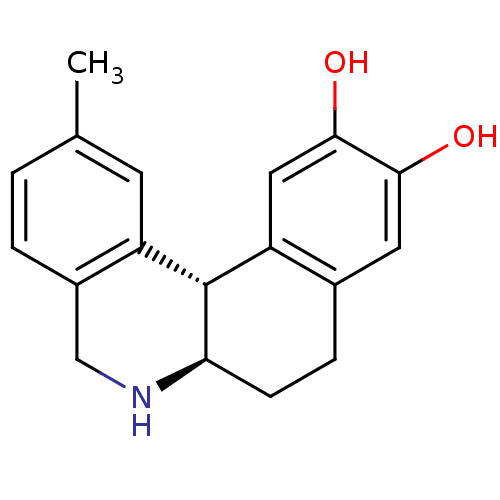
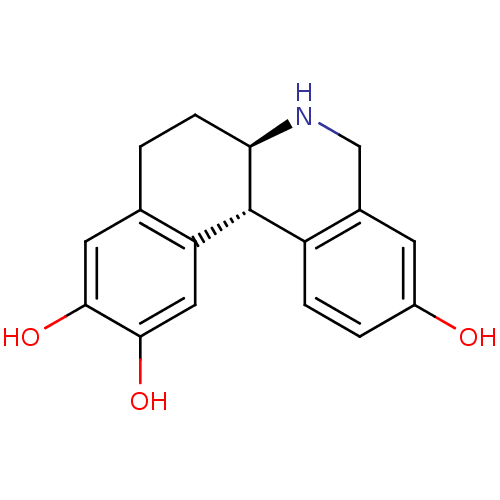
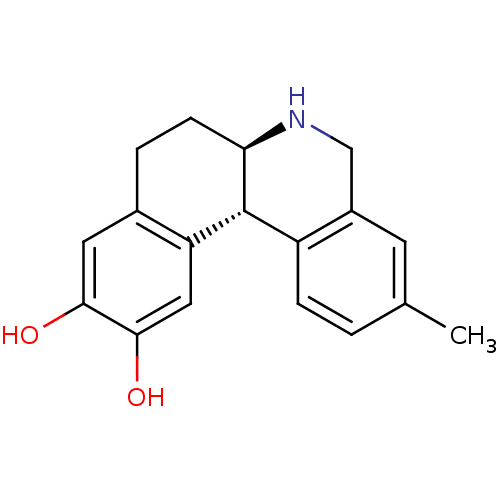
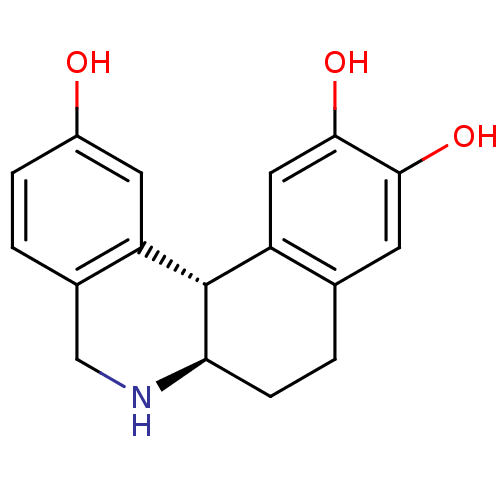
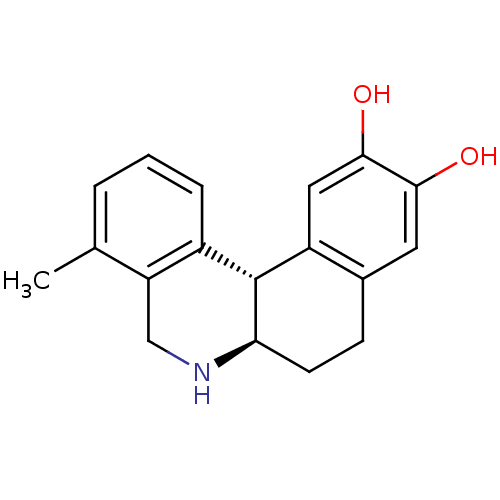
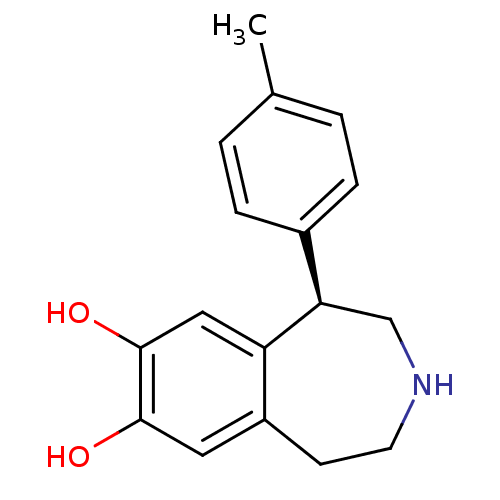
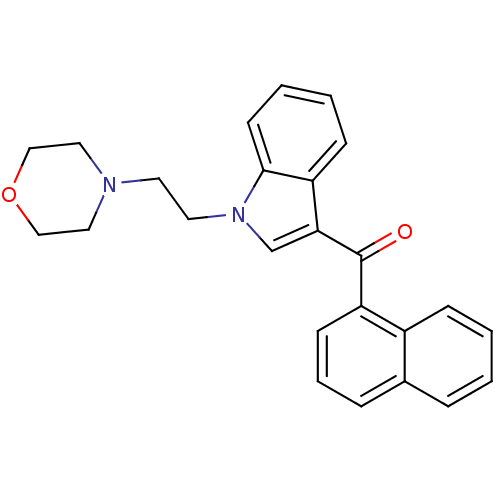
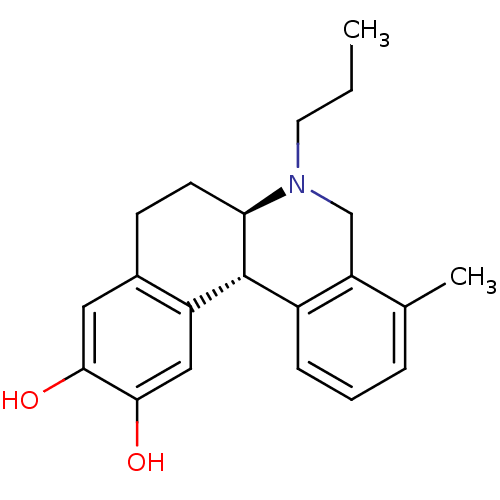
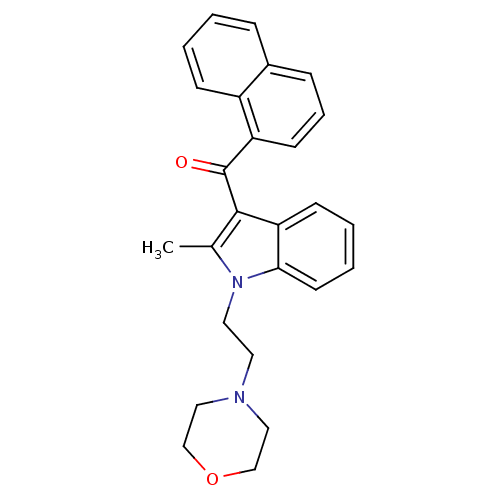
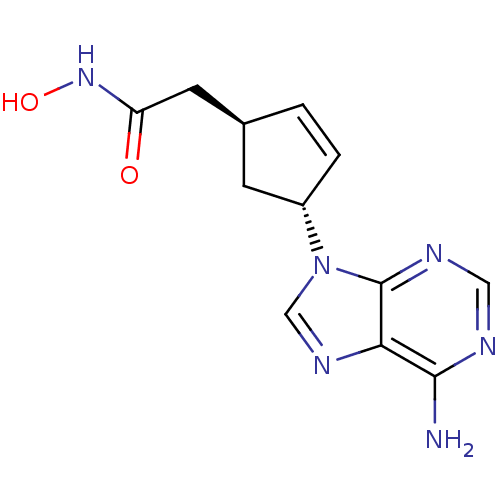
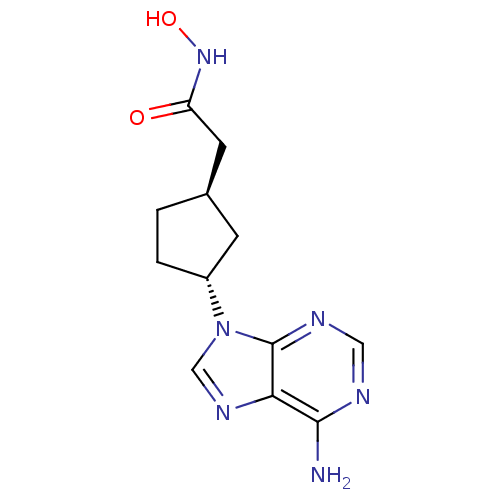
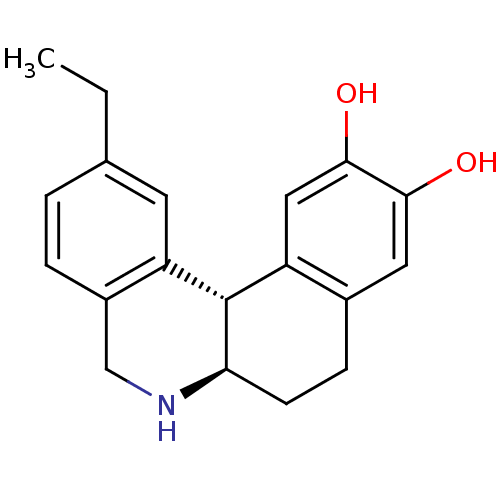
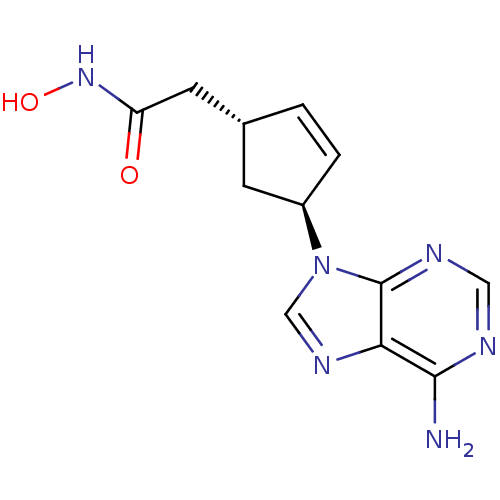
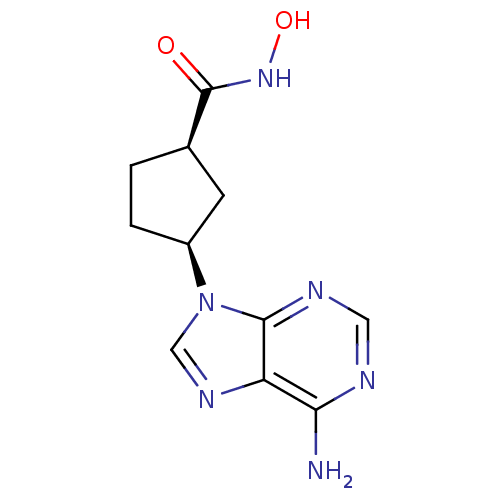
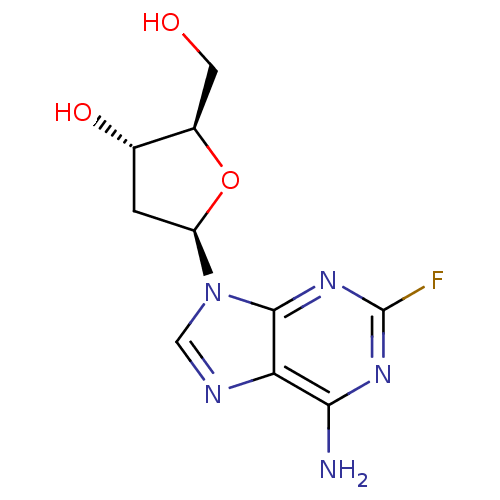
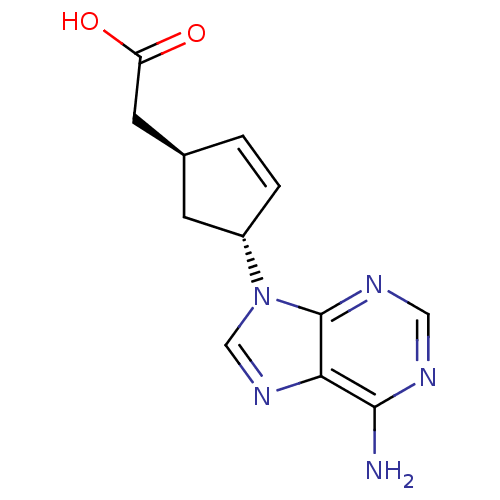
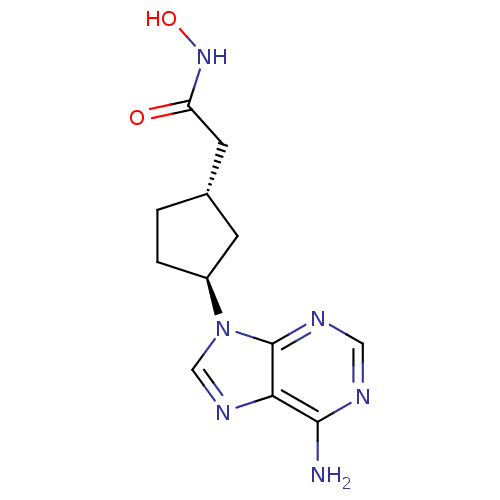
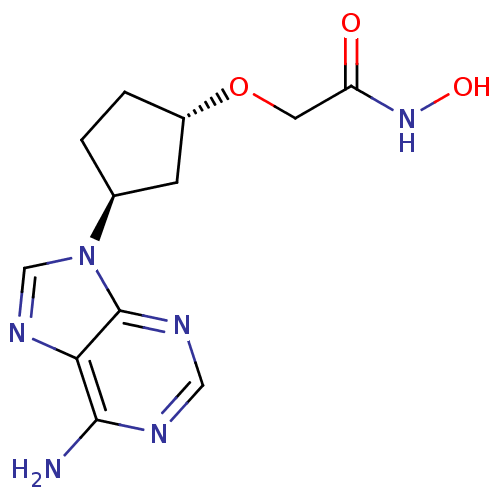
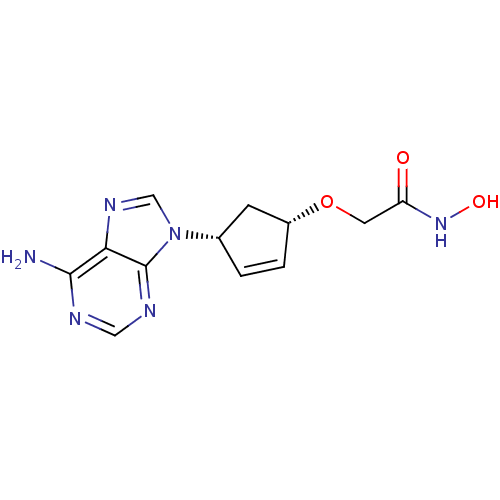
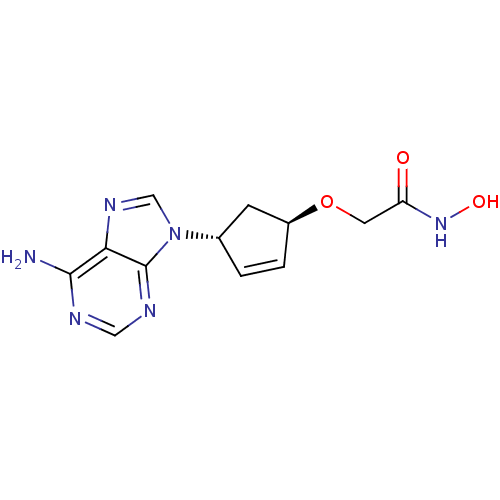
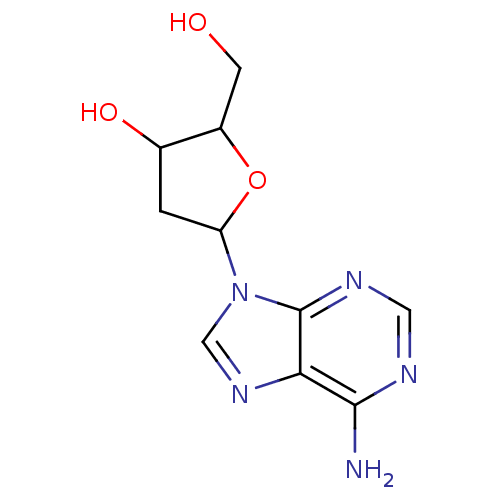
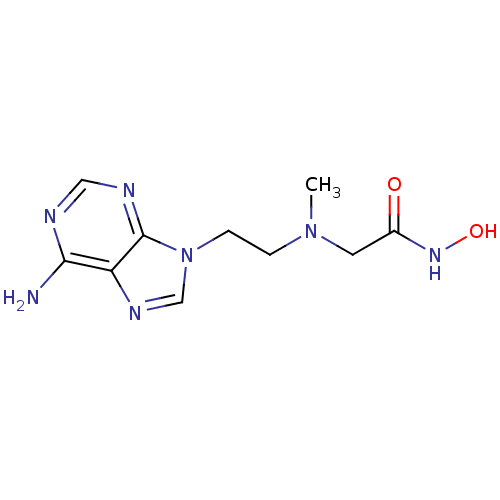
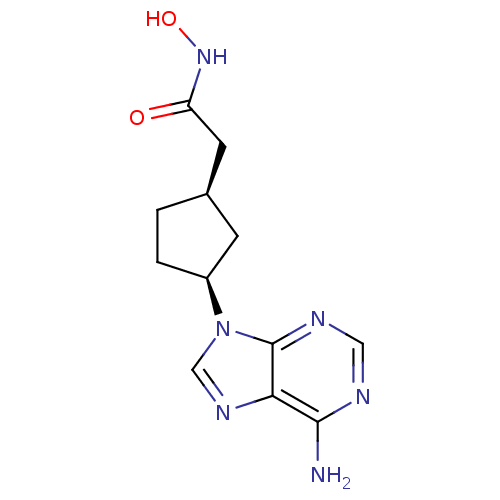
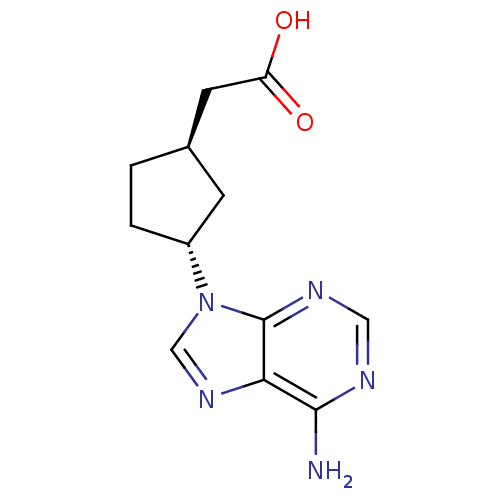
Report error Found 105 Enz. Inhib. hit(s) with Target = 'Adenylate cyclase type 5'
Affinity DataIC50: 7.20nMAssay Description:Antagonistic activity evaluated in adenylyl cyclase assayMore data for this Ligand-Target Pair
Affinity DataEC50: 8nMAssay Description:Ability of the Compound to activate Adenylate cyclase activity was measured by the conversion of [alpha-32P]ATP to 3'5'-cyclic AMPMore data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: >10nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 10nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 51nMAssay Description:Tested for effective dose agonist activity against adenylate cyclase in rat striatal membraneMore data for this Ligand-Target Pair
Affinity DataEC50: 60nMAssay Description:Effective concentration against adenylate cyclaseMore data for this Ligand-Target Pair
Affinity DataEC50: 80nMAssay Description:Effective concentration required to stimulate Adenylate cyclaseMore data for this Ligand-Target Pair
Affinity DataEC50: 114nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 130nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 134nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 141nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 148nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 151nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataEC50: 210nMAssay Description:Effective concentration required to stimulate Adenylate cyclaseMore data for this Ligand-Target Pair
Affinity DataEC50: 275nMAssay Description:Compound was evaluated for inhibition of hormone stimulated Adenylate cyclase activity in N18TG2 membranes.More data for this Ligand-Target Pair
Affinity DataEC50: 333nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 370nMAssay Description:Compound was evaluated for inhibition of hormone stimulated Adenylate cyclase activity in N18TG2 membranes.More data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 600nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataEC50: 610nMAssay Description:Effective concentration as Adenylate cyclase activity was measured in rat homogenateMore data for this Ligand-Target Pair
Affinity DataEC50: 1.10E+3nMAssay Description:Compound was evaluated for inhibition of hormone stimulated Adenylate cyclase activity in N18TG2 membranes.More data for this Ligand-Target Pair
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Compound was evaluated for inhibition of adenylate cyclase from rat brainMore data for this Ligand-Target Pair
Affinity DataEC50: 3.00E+3nMAssay Description:Compound was evaluated for inhibition of hormone stimulated Adenylate cyclase activity in N18TG2 membranes.More data for this Ligand-Target Pair
Affinity DataIC50: 4.30E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 4.60E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 4.60E+3nMAssay Description:Compound was evaluated for inhibition of adenylate cyclase from rat brainMore data for this Ligand-Target Pair
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibition of recombinant human adenylate cyclase 5 expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibitory concentration against type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibitory concentration against type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibition of recombinant human adenylate cyclase 5 expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 8.20E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 9.40E+3nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 1.08E+4nMAssay Description:Inhibitory concentration against type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.08E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 1.08E+4nMAssay Description:Inhibitory concentration against Type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.36E+4nMAssay Description:Inhibitory concentration against type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.36E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 1.36E+4nMAssay Description:Inhibitory concentration against Type V Adenyl Cyclase enzymeMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Compound was evaluated for inhibition of adenylate cyclase from rat brainMore data for this Ligand-Target Pair
Affinity DataIC50: 1.53E+4nMAssay Description:Inhibition of recombinant human adenylate cyclase 5 expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 2.51E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 2.88E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair
Affinity DataIC50: 3.54E+4nMAssay Description:Inhibitory activity against recombinant human adenylate cyclase 5 expressed in HEK293 cells.More data for this Ligand-Target Pair