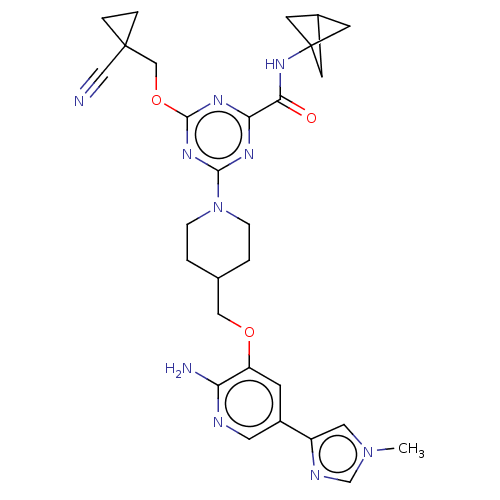
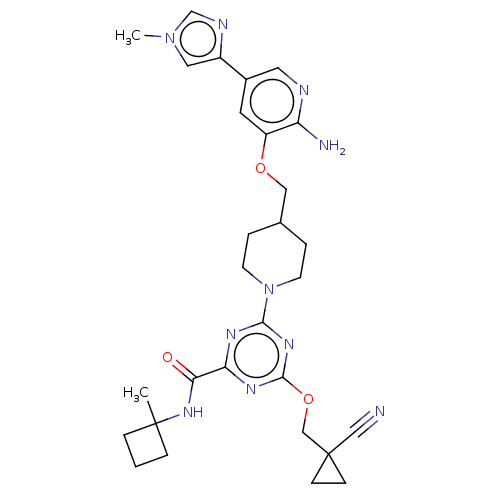
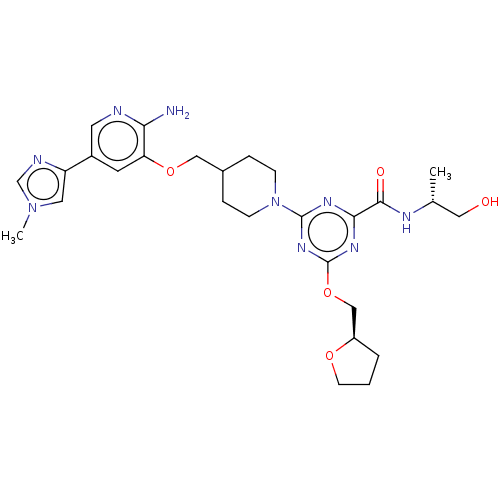
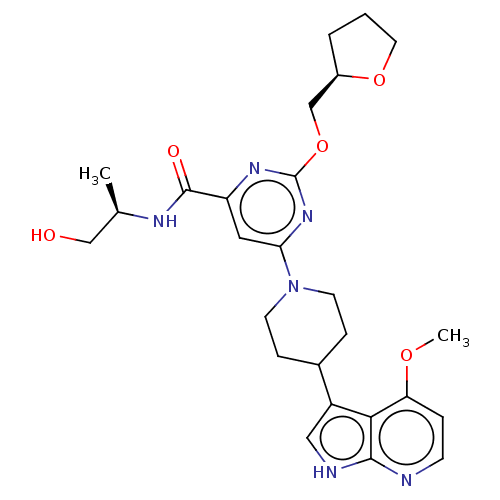
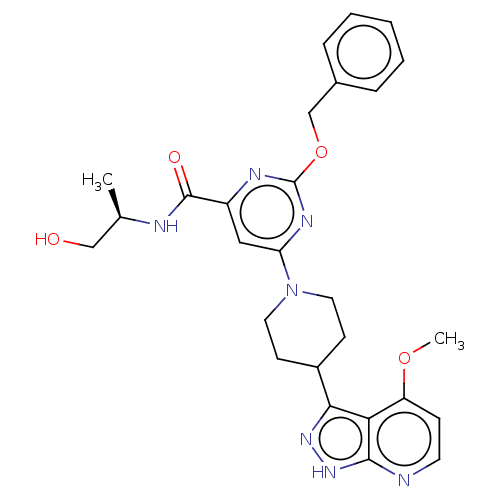
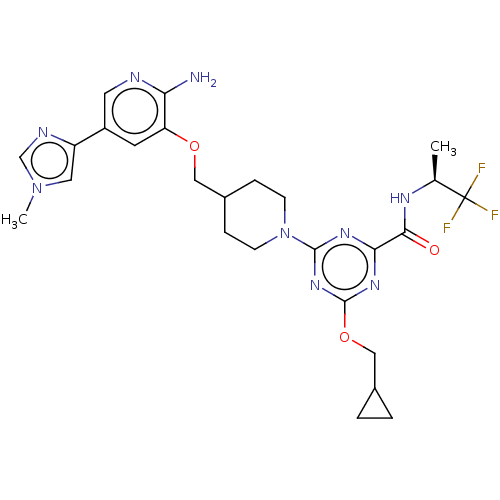
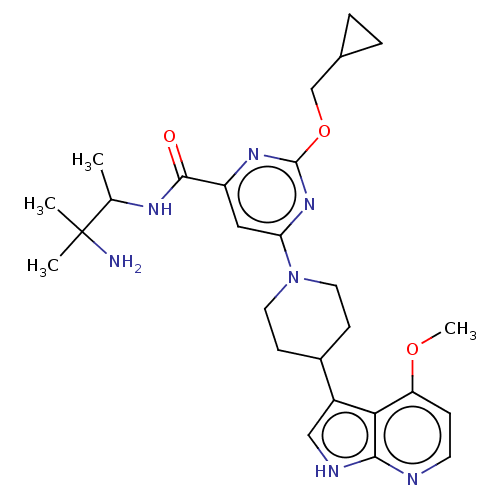
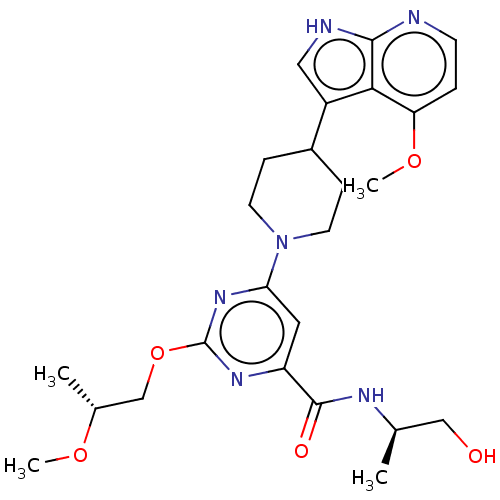
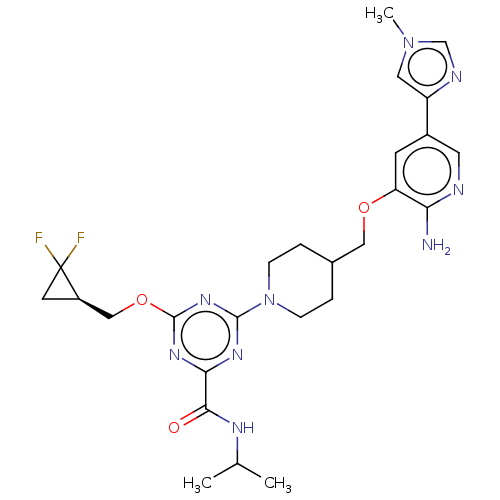
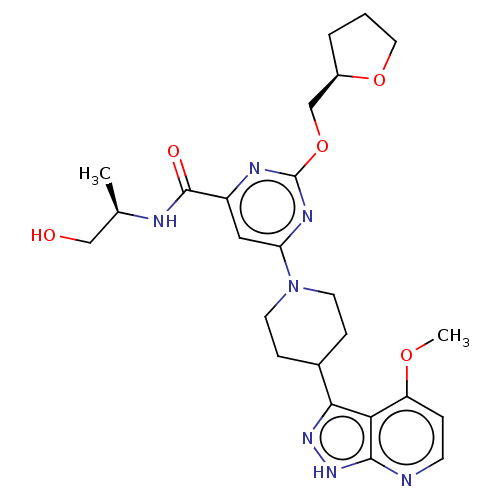
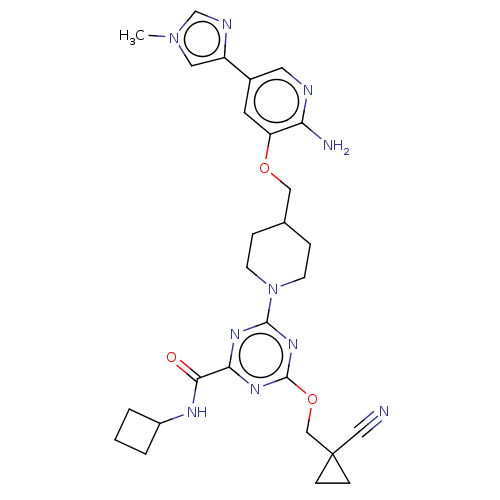
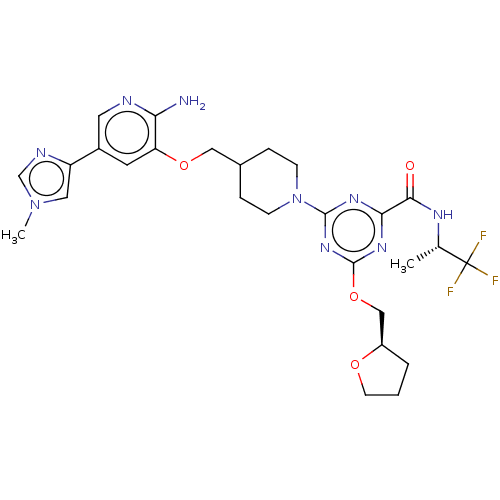
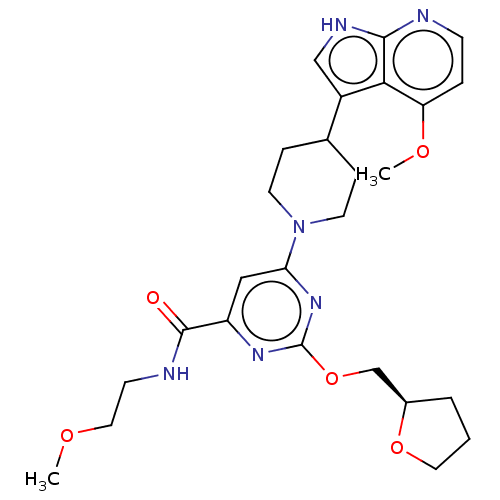
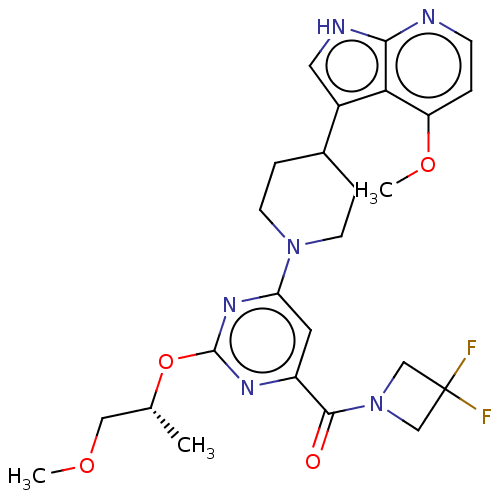
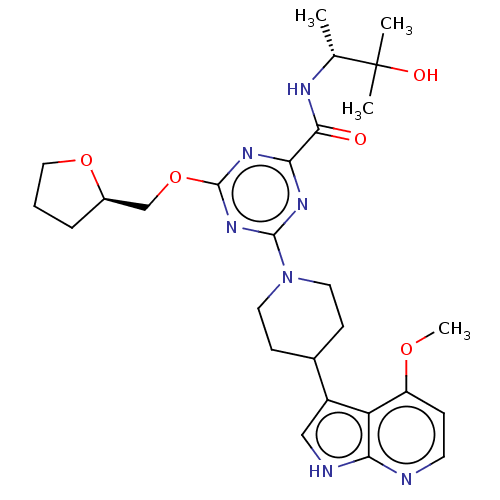
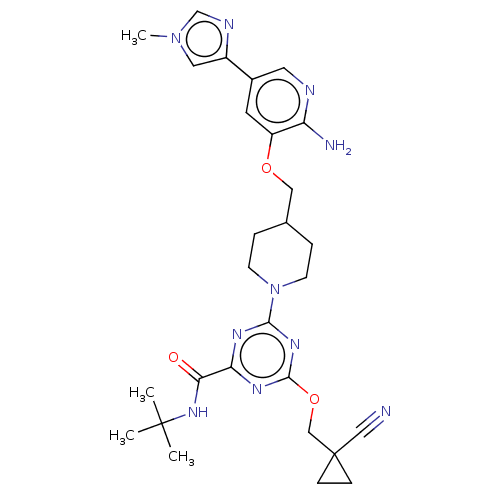
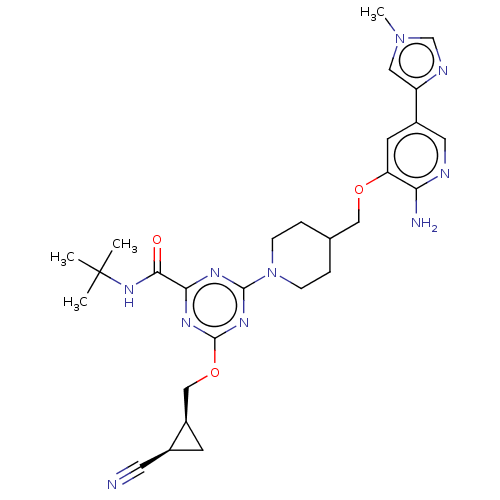
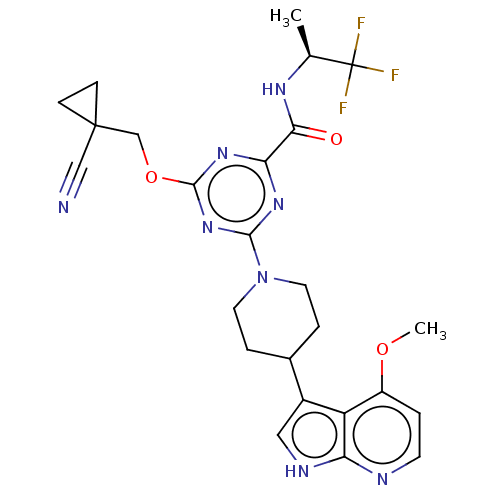
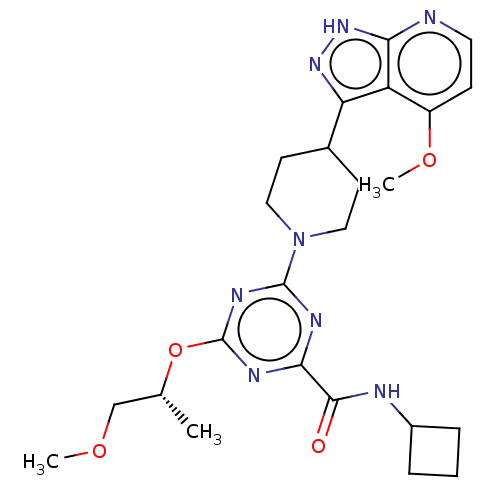
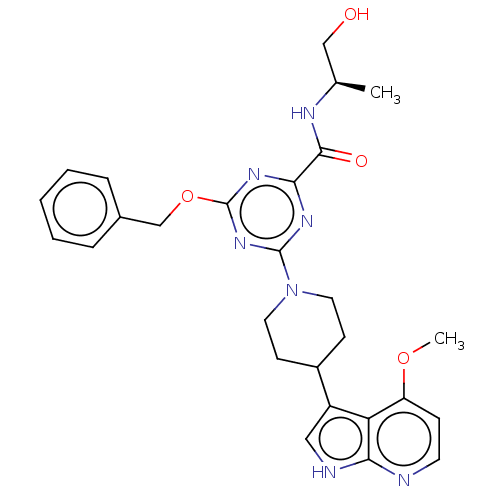
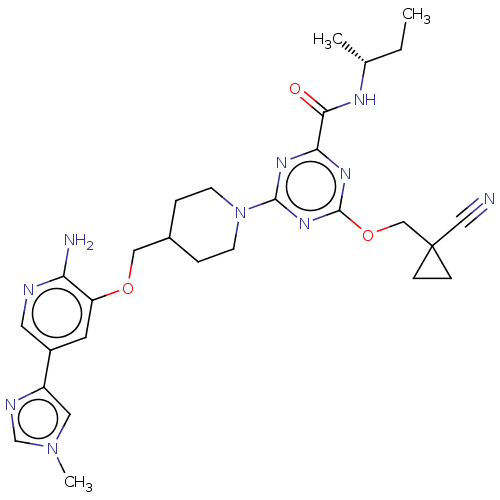
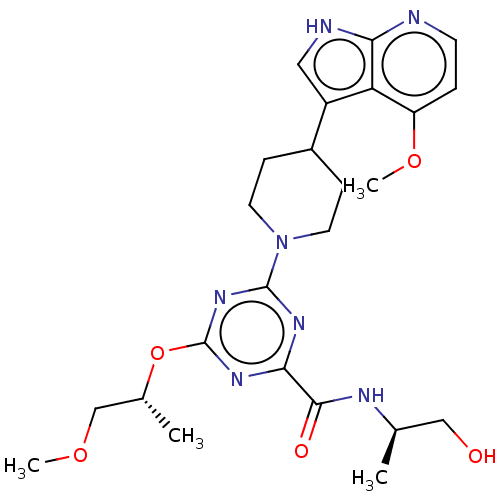
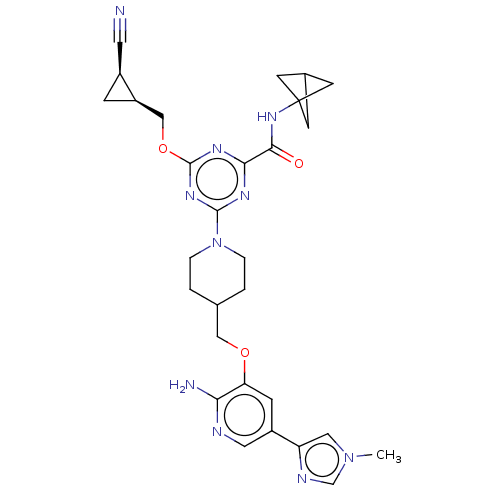
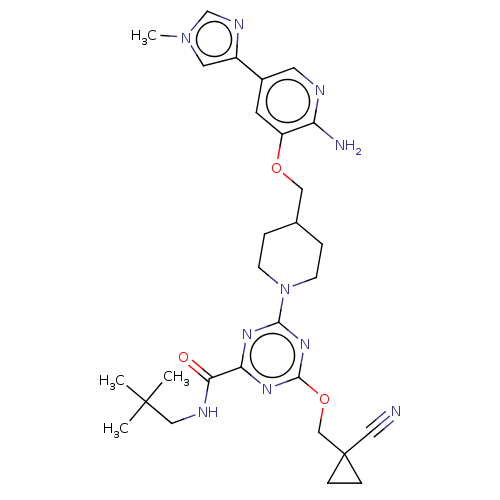
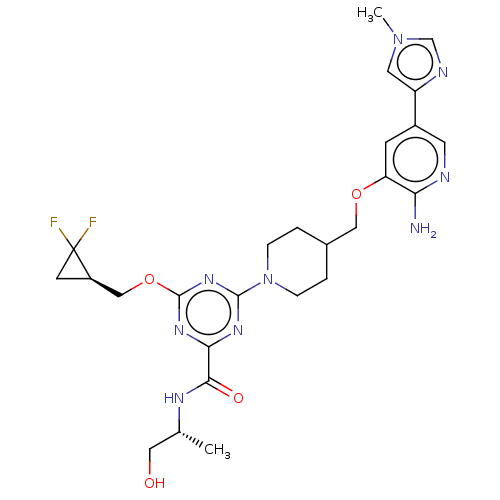
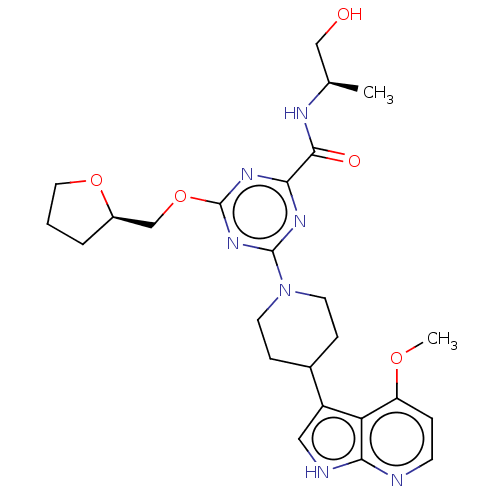
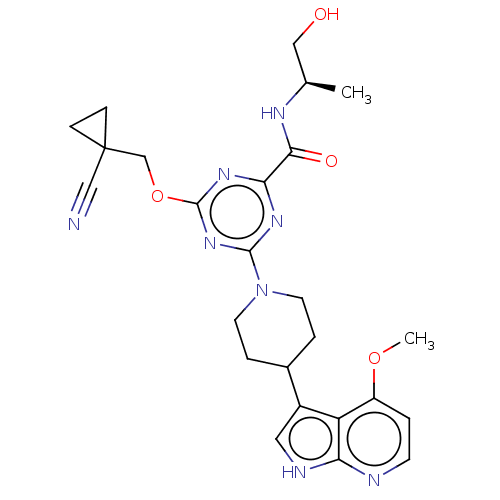
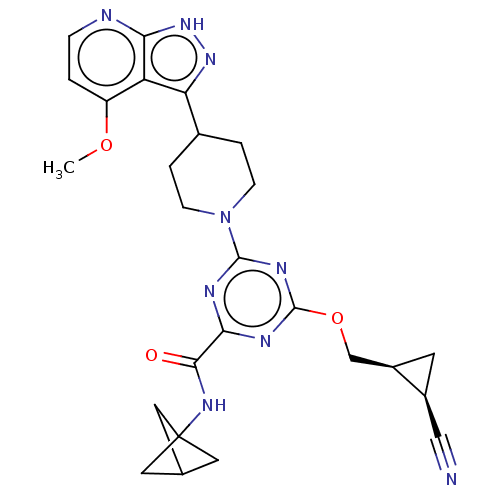
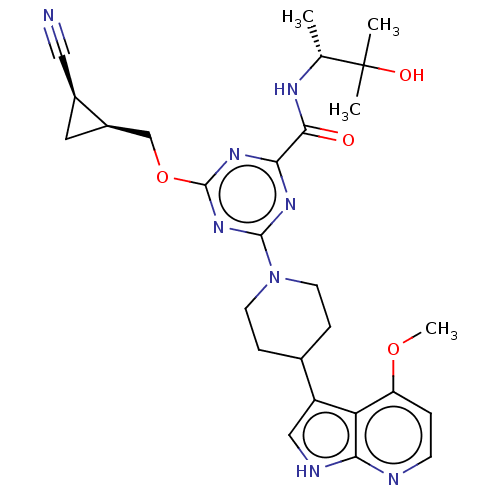
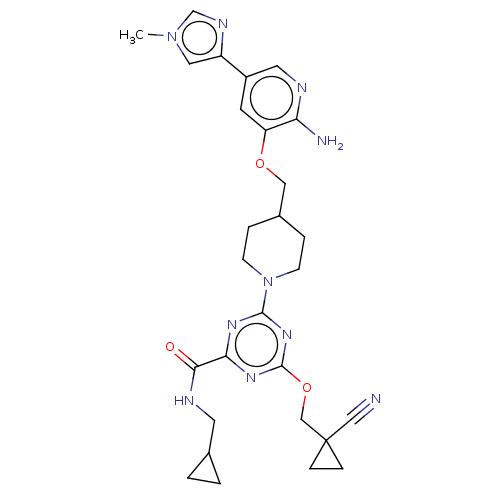
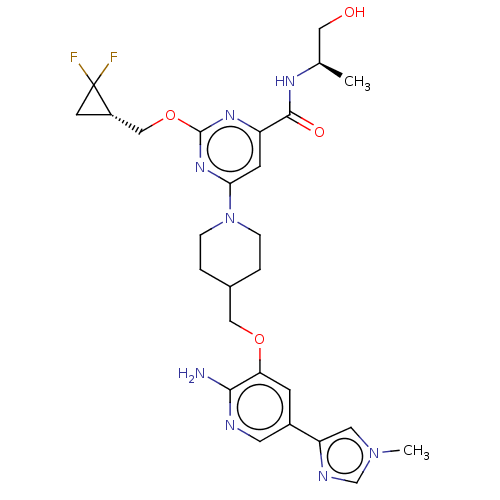
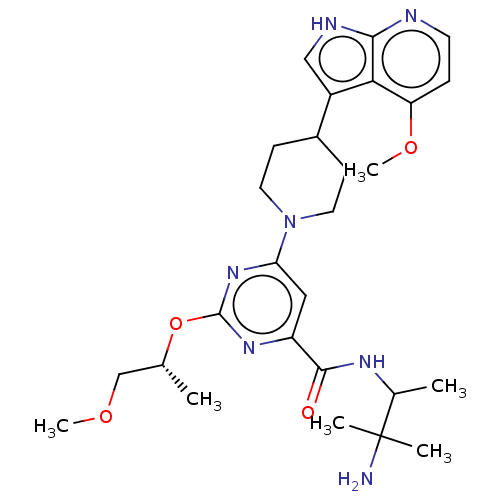
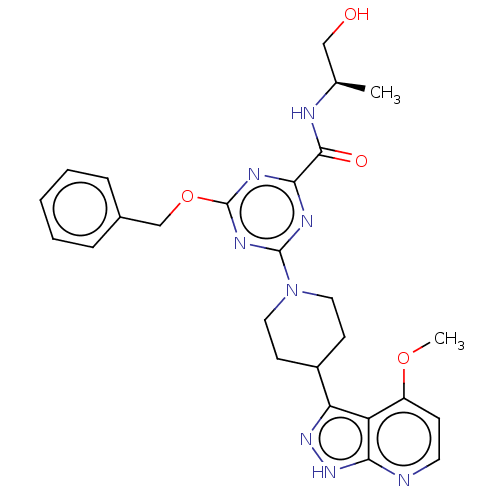
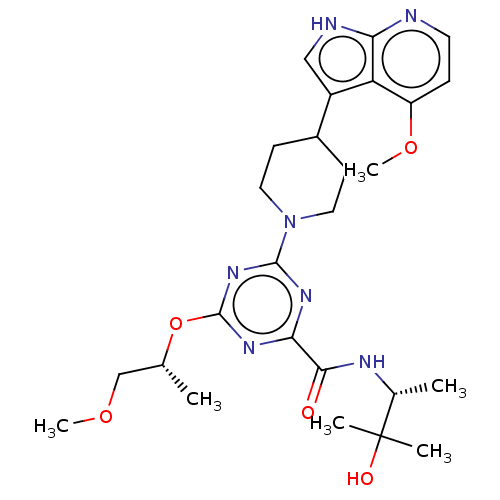
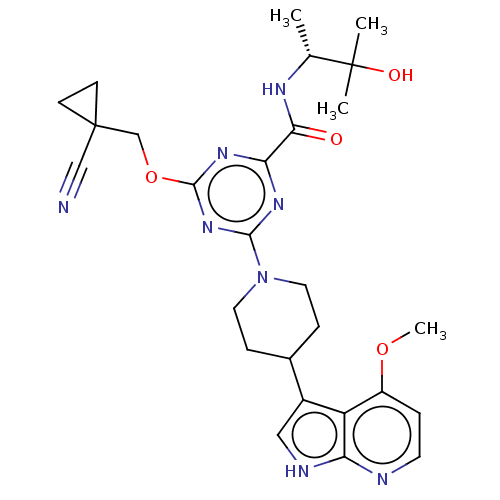
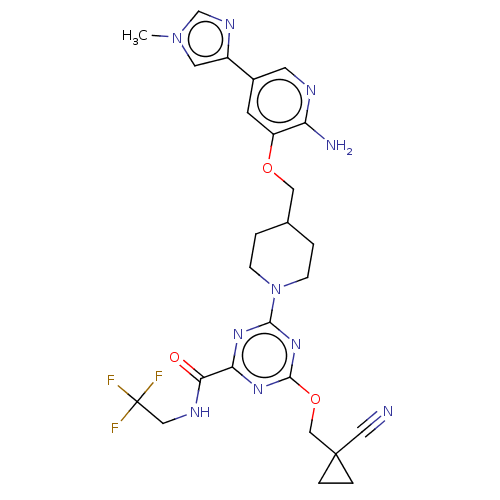
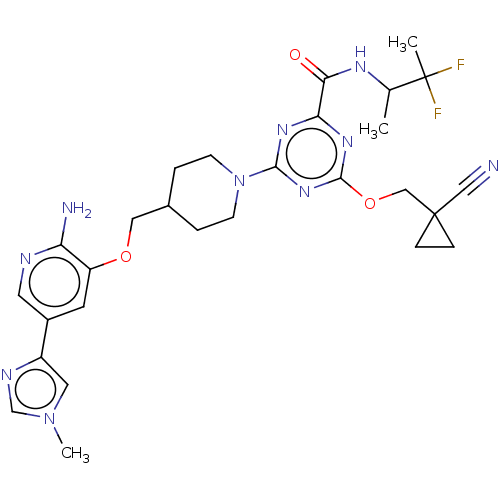
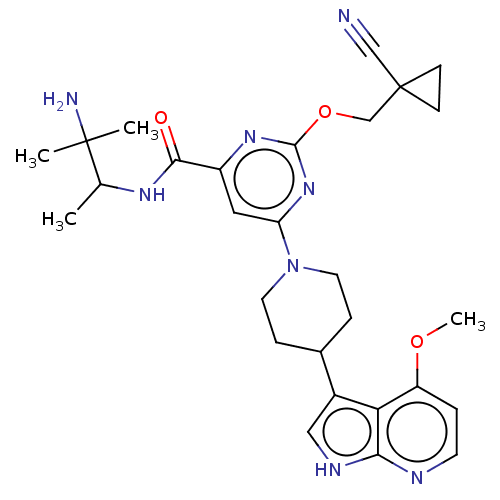
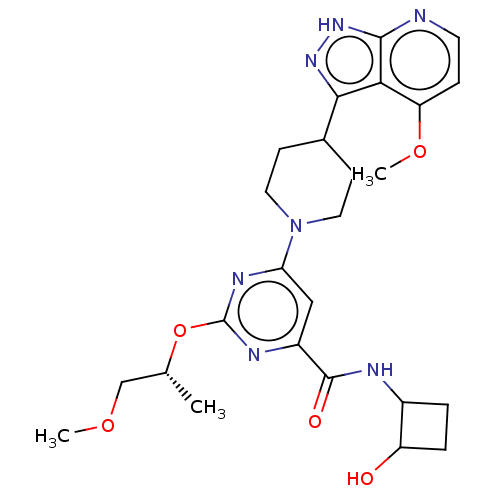
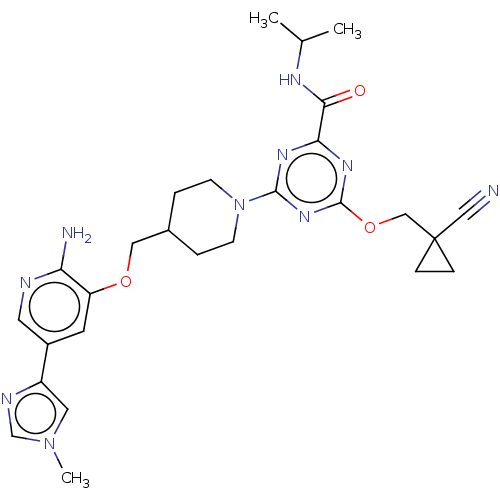
Report error Found 435 Enz. Inhib. hit(s) with all data for entry = 940
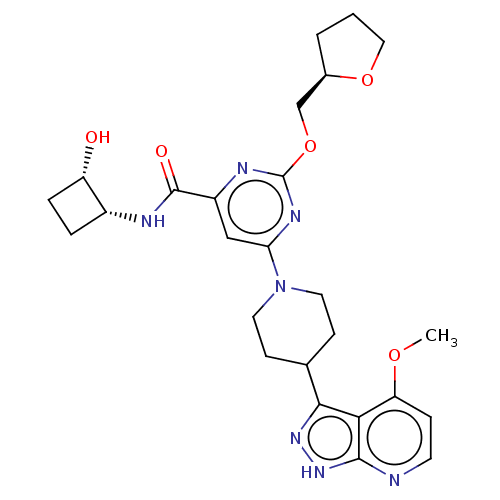
Affinity DataKi: 0.0100nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
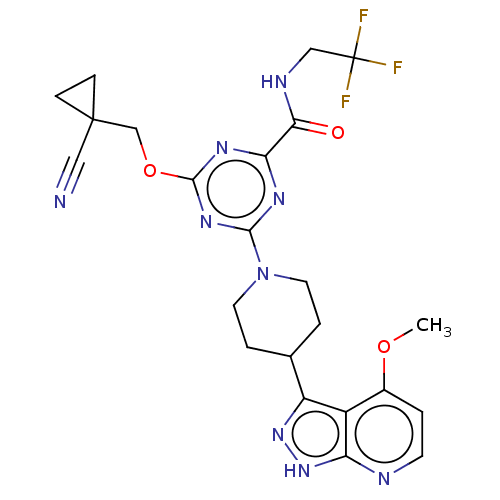
Affinity DataKi: 0.0120nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
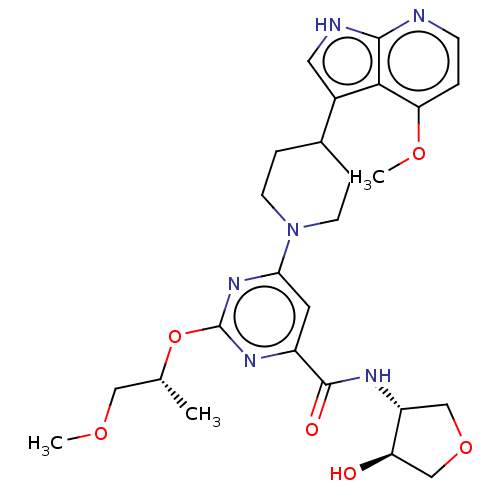
Affinity DataKi: 0.0120nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: <0.0130nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: <0.0130nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: <0.0130nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: <0.0130nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0140nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0150nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0150nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0180nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: <0.0180nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0190nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0200nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0200nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0200nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0210nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0210nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0210nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0250nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0260nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0280nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0280nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0280nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0290nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0300nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0320nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0320nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0330nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0340nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0350nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0370nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0380nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0380nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0390nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0400nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0400nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0410nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0480nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0480nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair
Affinity DataKi: 0.0490nMAssay Description:AXL enzyme inhibition (% inhibition, Kiapp and Ki values) by small molecule inhibitors was evaluated using a fluorescence-based microfluidic mobility...More data for this Ligand-Target Pair